About this course:
This course provides an overview of the various oral and intravenous (IV) cancer treatment modalities, including chemotherapy, targeted agents, biologic and immune-mediated therapies, hormonal treatments, and chemoprevention.
Course preview
Oncology Medication Management
An Overview of Oral and Intravenous Cancer Treatment Modalities
This course provides an overview of the various oral and intravenous (IV) cancer treatment modalities, including chemotherapy, targeted agents, biologic and immune-mediated therapies, hormonal treatments, and chemoprevention.
After this activity, learners will be prepared to:
- understand the difference between healthy and cancerous cell development, discuss primary and secondary cancer prevention strategies, and describe the goals of cancer therapy
- examine patterns in cancer drug resistance, recognize the most common side effects of chemotherapy, and discuss the clinical implications of each
- identify differences between cytotoxic chemotherapy and other types of cancer treatment
- identify the signs of chemotherapy hypersensitivity reactions and nursing interventions
- demonstrate understanding of the basic principles of safe handling, administration, storage, and disposal of cytotoxic medications and the proper personal protective equipment (PPE) required
- describe the structures of the immune system and differentiate between the innate and acquired immune system; how the immune system can be used to prevent, detect, and treat cancer
- differentiate between the side effects of chemotherapy, targeted therapies, immune-based therapies, and hormonal therapies, and their management strategies
- understand targeted therapies used in cancer treatment and the various mechanisms in which they work, in addition to the principles of chemoprevention and common chemopreventive agents
Cancer is a cluster of malignant diseases characterized by uncontrollable, abnormal cell growth; the ability to invade surrounding tissue and lymph nodes; and metastasize (spread) to distant locations within the body. The term “cancer” has evolved over several decades as biologic research has successfully enhanced the scientific understanding of cancer development and spread. However, cancer research remains profoundly driven toward answering the trillion-dollar question: how do we correct the abnormal mechanisms that occur at the cellular level to prevent, eradicate, and control disease? Scientific advancements and treatment breakthroughs have revolutionized the way cancer is managed, leading to innovative fields such as precision (or personalized) medicine, development of targeted therapies, and immunotherapy. Precision medicine uses the genomic profiling of a patient’s tumor to identify unique genetic mutations. This information allows healthcare providers (HCPs) to tailor cancer treatment to the patient’s tumor and select the most effective treatment. Targeted therapies block the growth and spread of cancer by interfering with specific genes, proteins, and blood vessels that allow cancer cells to replicate, grow, and spread. Immune-based therapies assist the immune system in identifying cancer cells and attacking them, as it would for any other infection, virus, or potential threat. Immunotherapy refers to medications like monoclonal antibodies, checkpoint inhibitors, cancer vaccines, and chimeric antigen receptor (CAR) T-Cell therapy. Nevertheless, cytotoxic chemotherapy remains the most prevalent treatment option and is still considered the standard of care and firstline treatment for many cancers (Nettina & Nelson-Tuttle, 2024; Yarbro et al., 2018).
The National Comprehensive Cancer Network (NCCN, n.d.-a) is an alliance of leading cancer centers and experts devoted to cancer care, research, and education. Through rigorous clinical trial research, data compiled across institutions, and annual expert panel reviews, the NCCN provides evidence-based guidelines for cancer treatment according to cancer type, pathology, genetics, staging, inheritance patterns, and other specific features. These guidelines are widely utilized in cancer care and guide medical decision-making throughout each patient’s disease trajectory. Despite the tremendous scientific advancements in identifying more specific and highly effective cancer treatments, drug resistance is a significant barrier to finding a cure for cancer. As a result, the question of how this disease is to be cured remains unclear and unrequited as the disease continues to expand and affect a significant proportion of the global population (NCCN, n.d.-a, n.d.-b; Yarbro et al., 2018).
According to the American Cancer Society (ACS, 2026a), more than 2.1 million new cancer diagnoses are expected in the United States in 2026, with approximately 626,000 cancer-related deaths. These numbers translate into nearly 1,700 deaths per day. Cancer was the second most common cause of death in the United States in 2024, exceeded only by heart disease. Nevertheless, substantial progress has been made in the last few decades, with cancer deaths dropping from 215.1 per 100,000 people in 1991 to 139.4 per 100,000 people in 2024, a decline of 35%. This reduction in death rates is attributable to advancements in early detection and treatment and a decrease in smoking rates. There are an estimated 18.6 million cancer survivors in the United States, representing about 5% of the population; this number is projected to increase to 22 million by 2035 (ACS, 2026a; Wagle et al., 2025; Xu et al., 2026).
Pathophysiology of Cancer
Cancer cells have distinct features compared to normal cells, such as their appearance under a microscope, growth, replication, and function. The cell cycle is a 5-stage process of cellular reproduction that occurs in both normal and cancerous cells. Gap 0 or G0 (quiescence) is the resting stage in which cells are temporarily out of the cell cycle. During this stage, all cellular activity continues except for reproduction. During Gap 1 or G1, ribonucleic acid (RNA) and protein synthesis occur. This stage is considered the gap between resting and DNA synthesis. Synthesis or S occurs when deoxyribonucleic acid (DNA) synthesis occurs, as cellular DNA is duplicated in preparation for division. Gap 2 or G2 encompasses further protein and RNA synthesis as the cell constructs the mitotic apparatus. Finally, Mitosis, or M, involves cellular division (Yarbro et al., 2018). Refer to Figure 1 for a depiction of the cell cycle.
Figure 1
The Cell Cycle and Checkpoints
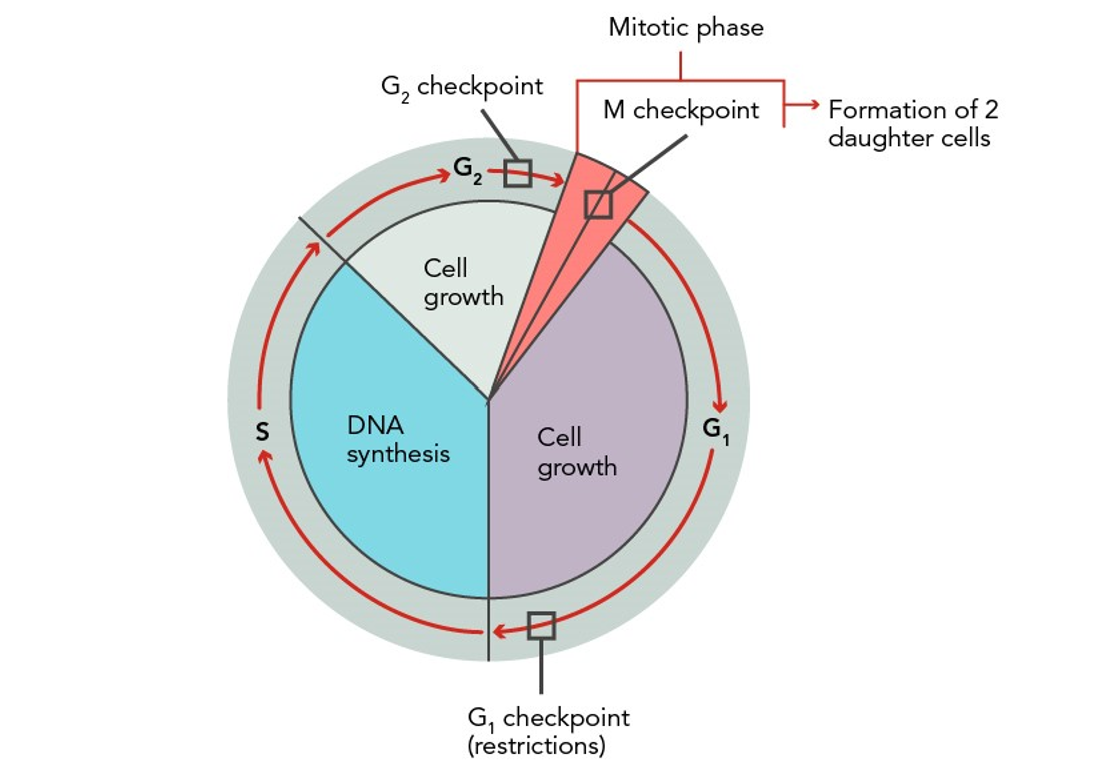
Cancer cells collect in an area, pile on top of one another, and create a malignant tumor. Normal, healthy cells reproduce in an organized, controlled, and orderly manner as they mature into cells that serve specific functions and have predetermined life spans. They undergo apoptosis, or programmed cell death, to help the body rid itself of unneeded cells. They do not divide when space or nutrients are limited and do not spread to other parts of the body. In contrast, cancer cells are less specialized and exhibit ...purchase below to continue the course Figure 2 Normal Versus Abnormal Cell Development All cancer is inherently genetic, as all cancer cells carry genetic mutations that lead to unregulated cell division and growth. However, it is critical to understand the distinction between genetic and inherited, as these terms are not synonymous. Inherited (or hereditary) cancer occurs when a damaged gene with a high cancer susceptibility is passed through generations within a family. In other words, a patient with hereditary cancer is born with a genetic mutation and predisposition to develop the disease. A classic example is the breast cancer genes (BRCA1 and BRCA2), which produce tumor-suppressor proteins that help repair damaged DNA, ensuring proper stability and function of cellular genetic material. When either of these genes are mutated, DNA damage may not be adequately restored, leaving cells vulnerable to additional genetic alterations that can lead to cancer. Inherited mutations in BRCA1/2 genes substantially increase the risk of breast and ovarian cancers and are associated with a heightened risk for several other cancers. A mutated BRCA1/2 gene can be inherited through a patient’s biological parents. Therefore, each child of a parent who carries the mutation has a 50% chance of inheriting the mutation. Despite the heightened media attention on inherited cancers and BRCA mutations following the 2013 New York Times op-ed spotlighting celebrity Angelina Jolie’s decision to have a preventive mastectomy after learning she carried the genetic mutation, most cancers are not inherited, and patients are often diagnosed without a family history (Liede et al., 2018; Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023). Metastases are secondary growths of cancer in another organ. It occurs after a cancerous cell detaches from the original tumor site, invades local tissue, and migrates through the lymphatics and blood vessels to another body area. Over time, the cancerous cell replicates in the new area, creating a secondary tumor site. The four most common sites where cancers metastasize are the liver, lung, bone, and central nervous system. A common misconception among patients with metastatic cancer is that they have developed a second cancer type, so vigilant patient and family education is often required to ensure accurate understanding. For example, a patient with breast cancer who develops metastases in the liver does not have liver cancer. Instead, the patient’s breast cancer cells have traveled to the liver, reproduced in the liver, and created a tumor, leading to a diagnosis of metastatic breast cancer or breast cancer with metastasis to the liver (Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023; Yarbro et al., 2018). Risk and Protective Factors While the definitive cause of cancer is not entirely understood, numerous factors increase the risk of the disease and are generally distributed between two categories: modifiable and nonmodifiable. Some theories postulate that cancer may occasionally occur due to spontaneous cell transformation when no causative agent is identified. Still, most researchers consider cancer development as a process resulting from cell damage induced by outside influences, called carcinogens. Carcinogens are substances, radiation, or exposures that can damage the genetic material (DNA) throughout a person’s lifetime, resulting in carcinogenesis or cancer formation. A few examples of carcinogens include tobacco, diesel exhaust, and ultraviolet (UV) radiation. Age is the most outstanding risk factor for cancer, as cancer incidence rises alongside age. Other risk factors include chemical exposure, poor nutrition, high-fat diets, obesity, sedentary lifestyles, and excessive alcohol intake. Although viruses are not considered carcinogens, they utilize carcinogenic mechanisms to transform human cells (ACS, 2023; Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023; Yarbro et al., 2018). Primary cancer prevention can thwart many cancers, involving the minimization of harmful exposures and reducing or avoiding unhealthy lifestyle behaviors. The ACS (2026a) researchers have determined that approximately 40% (850,000) of newly diagnosed cancers in the United States are potentially avoidable, as they are directly correlated with tobacco use, excess weight, alcohol consumption, and other modifiable behaviors. Tobacco is the single most significant cause of cancer-related deaths and is responsible for 86% of lung cancer deaths and 30% of all cancer deaths. Secondhand smoke results in more than 6,000 cases of lung cancer annually in patients who formerly or never smoked (ACS, 2026a). Skin cancers are primarily due to excessive sun and UV radiation exposure. While studies indicate that behavioral counseling on applying proper sunscreen and avoiding direct exposure improves sun protective behaviors, these studies do not indicate a decreased risk of sunburn or prevention of skin cancer (National Cancer Institute [NCI], 2025b, 2025c). Infections and viruses are associated with an increased risk of certain forms of cancer, such as the relationship between hepatitis B, hepatitis C, and hepatocellular cancer. Cancers related to human papillomavirus (HPV) can be prevented through behavioral and lifestyle changes and vaccination (ACS, 2023). According to the Centers for Disease Control and Prevention (CDC, 2024), HPV causes more than 11,000 cases of cervical cancer in the United States each year, and approximately 4,000 patients die annually. Additionally, HPV can cause cancer of the oropharynx, vagina, vulva, anus, and penis. Gardasil-9 is the currently approved HPV immunization that protects against over 90% of HPV-related cancers and genital warts. The CDC (2024) recommends two doses of the HPV vaccine at ages 11–12, but it can be started in children as young as 9 (CDC, 2024). Secondary cancer prevention involves partaking in screenings and testing to identify high-risk individuals who require increased surveillance compared to the general population. These measures can prevent cancer by identifying precancerous lesions and taking appropriate action before the cells develop into invasive cancer or by undergoing interventions, such as prophylactic mastectomy in an otherwise healthy patient with a BRCA mutation, to diminish the lifetime risk of breast cancer. Screenings allow for early cancer detection when it is still treatable or potentially curable. Examples of cancer screening tests include colonoscopy, sigmoidoscopy, fecal occult blood testing (FOBT), DNA fecal immunochemical testing (FIT; e.g., Cologuard), mammography, Papanicolaou testing (pap smear), prostate-specific antigen (PSA), and digital rectal exam (DRE; Viera, 2025). The US Preventive Services Task Force (USPSTF) now recommends cancer screening with a low-dose spiral computed tomography (CT) scan annually to detect curable stage I lung cancer in patients who meet designated criteria, such as: Goals of Cancer Therapy Cancer therapy has four main goals: prevention, cure, control, and palliation. While prevention and cure are relatively transparent in their definitions, control refers to extending a patient’s life when a cure is unlikely or impossible, by preventing the growth of new cancer cells and reducing the size and impact of the existing disease. Palliation focuses on comfort when cure and disease control cannot be achieved. Several key terms describe the types of therapy prescribed for cancer patients and are essential for HCPs to understand. Neoadjuvant therapy is given to shrink a tumor, so the primary treatment, usually surgical intervention, may not need to be as extensive. For instance, neoadjuvant chemotherapy is given to shrink a breast tumor so the surgeon can perform a lumpectomy instead of a mastectomy. Adjuvant therapy is given after the primary treatment and aims to prevent recurrence and reduce micro-metastases. For potentially curative treatment regimens, maximum tolerated doses of drugs are delivered on a specific schedule to achieve the greatest efficacy (i.e., the highest cancer cell kill rate). Chemotherapy may also be used for palliation. Palliative therapy aims to relieve or delay cancer symptoms, focusing on comfort, symptom management, and improved quality of life. Thus, palliative chemotherapy doses are often adjusted to minimize treatment-related toxicity. Chemoprevention uses specific pharmaceutical agents to prevent cancer in high-risk individuals. The most common example of chemoprevention is tamoxifen (Soltamox), an oral selective estrogen receptor modulator for individuals at high risk for developing breast cancer. Myeloablation is the obliteration of bone marrow in preparation for stem cell or bone marrow transplantation with high-dose/intensive chemotherapy (Hinkle et al., 2021; Rogers & Brashers, 2023; Yarbro et al., 2018). Drug Resistance Cancer can adapt and mutate over time, particularly in response to treatment. Drug resistance occurs when cancer stops responding to the prescribed treatment and starts to grow again. It poses a significant barrier to finding a cure for cancer as tumors develop the ability to resist the effects of chemotherapy agents. For instance, a patient may have an initial robust response to treatment for some time, before the treatment stops working. Drug failure in cancers attributed to resistance can have various causes, and multiple resistance mechanisms may occur. Intrinsic resistance occurs when cancer cells are inherently resistant to cancer drugs. In contrast, acquired resistance happens when cancer cells develop resistance after exposure to the drug due to the emergence of resistant cancer clones. In some cases, cancers can develop multiple drug resistance (MDR), resulting in minimal cell death and the growth of drug-resistant tumors. While there are several proposed rationales for cancer drug resistance, it is often due to a combination of issues. Some mechanisms include: Concern for drug resistance is the primary reason cancer drugs are often given in combination. Since cancer cells are selective in their division, replication, and spread mechanisms, combining therapies with varying mechanisms of action can reduce the incidence of developing resistance to a single drug. Furthermore, as cancer progresses, it increasingly develops mutations. As a result, if cancer becomes resistant to a drug or a group of drugs, it is much more likely to become resistant to other drugs. Therefore, following appropriate evidence-based treatment guidelines is essential, as the best possible treatment protocol should be administered first (Konieczkowski et al., 2018; Lei et al., 2023; Yarbro et al., 2018). Types of Therapies Chemotherapy Chemotherapy, also called cytotoxic or antineoplastic therapy, encompasses a group of high-risk, hazardous drugs that destroy cancer cells with as few effects on healthy cells as possible. Chemotherapy generally works by interfering with the normal cell cycle; impairing DNA synthesis and cell replication; and preventing cancer cells from dividing, multiplying, and forming new cancer cells. The growth and size of a tumor are a product of the proportion of cells actively dividing (growth fraction), the length of the cell cycle (doubling time), and the rate of cell loss. The higher the growth fraction is, the greater the chemotherapy response or cell kill. Tumor burden refers to the number of cancer cells, the size of the cancerous tumor, or the amount of cancer in the body. Cancers with smaller tumor burdens are usually more sensitive to chemotherapy because they are vascular. As a tumor grows, the growth rate slows due to the crowding of cells, poor vascularization from decreased blood flow, and limited nutrients. As a result, the number of cells actively dying decreases, and treatments become less effective. Factors outside the characteristics of the tumor that influence response to chemotherapy include the patient’s general state of health and physical status, including performance status, comorbidities, age, and prior cancer treatment. Generally, chemotherapy-naïve patients (i.e., those who have never received chemotherapy before) tend to tolerate and respond to treatment with less toxicity and greater efficacy than those who have received prior therapies (Nettina & Nelson-Tuttle, 2024; Yarbro et al., 2018). Chemotherapy drugs are distributed throughout the body by the bloodstream and can cause significant morbidity and mortality if not used correctly and cautiously. Chemotherapy may be used as a single agent or combined with other drugs. However, it is more commonly used in combination for greater efficacy against cancer and to reduce the potential for drug resistance. Chemotherapy dosing is based primarily on the patient’s body surface area (BSA). BSA is calculated by multiplying a patient’s weight (kilograms) by height (centimeters), dividing the product by 3600, and then taking the square root of that number to achieve the final metric (mg/m2). Other agents are dosed based on the area under the curve (AUC), which refers to the amount of drug exposure or the total drug concentration in plasma over time. While the most common route of chemotherapy administration is intravenous (IV), it may also be administered via other routes, including oral, subcutaneous (injection), intramuscular (injection), intrathecal (directly into the central nervous system), intravesicular (directly into the bladder by urinary catheter), or intraperitoneal (infused directly into the intra-abdominal cavity). IV chemotherapy is generally administered in hospitals, outpatient infusion clinics, or via continuous infusion pumps that patients wear at home (Amjad et al., 2023; Hinkle et al., 2021; Konieczkowski et al., 2018). Specialized education, preparation, and training are required for oncology nurses who administer chemotherapy and other hazardous cancer medications to ensure safe patient care. The Oncology Nursing Society (ONS) and Oncology Nursing Certification Corporation (ONCC) offer the ONS/ONCC Immunotherapy Certificate Course and provide up-to-date, evidence-based resources. The ONS also outlines competencies required for nurses to administer these agents, including in-depth knowledge of cancer medications and infusion therapy practices. Most accredited cancer centers and hospitals in the United States require oncology nurses to hold proper certification before administering these medications (ONS, n.d.). In addition, the American Society of Clinical Oncology’s (ASCO) Quality Oncology Practice Initiative (QOPI) certification program also requires participating hospitals, infusion centers, and healthcare provider practices to comply with safety standards for chemotherapy administration (ASCO, n.d.; ONS, 2018). Since chemotherapy can cause severe irritation, damage, and injury to the veins and subcutaneous tissue, many patients have a central venous catheter (i.e., port or mediport) placed; this small device is surgically implanted under the skin, usually in the chest wall, for direct access to the bloodstream. The port may be used to draw blood and infuse chemotherapy drugs. Some chemotherapy medications, such as vesicants, should be given only through a port as they are too caustic to be administered through a peripheral vein. Vesicants are drugs that can lead to severe soft tissue necrosis or the formation of blisters when they leak or infuse outside the vein and into the soft tissue (i.e., extravasation). The severity of chemotherapy extravasation varies according to the drug type, dose, and concentration. Initial signs and symptoms can include acute burning pain and swelling at the infusion site and become increasingly severe in the hours, days, and weeks following the initial injury. Patients may develop blisters, which usually begin within 3 to 5 days and may be followed by peeling or sloughing of the skin with invasion to and destruction of deeper structures. Tissue necrosis usually occurs within 2 to 3 weeks. In the most severe cases, damage can reach tendons, nerves, and joints, leading to functional and sensory impairment of the area, disfigurement, or loss of the limb entirely. Some examples of chemotherapy vesicants include doxorubicin (Doxil), dactinomycin (Cosmegen), mitomycin C (Mutamycin, Mitosol), and vinca alkaloids (Vinblastine, Vincristine, and Vinorelbine). Specific guidelines address the management of peripheral IV sites for chemotherapy, such as location, placement, monitoring parameters, and frequency of evaluations of blood return. All chemotherapy agents should be considered irritants and can cause inflammation, pain, or irritation. When administering chemotherapy through a peripheral IV site, the appearance and function of the site must be closely monitored before, during, and after the infusion, assessing for erythema, swelling, or loss of blood return. Patients should be counseled to report any pain, burning, or other abnormal sensations during the infusion (Amjad et al., 2023; Hinkle et al., 2021; Nettina & Nelson-Tuttle, 2024; Olsen et al., 2019). Most chemotherapeutic agents are broad in their attack, killing healthy and cancer cells. According to Olsen and colleagues (2019), agents are classified according to their biochemical activity, mechanism of action, and phase of action during the cell cycle. The cell cycle phases are divided into cell cycle–specific and cell cycle–nonspecific. Cell cycle–specific drugs exert cytotoxic effects on cells actively dividing at targeted stages within the cell cycle. These drugs are not active against cancer cells during the resting phase (G0) and are schedule-dependent; i.e., they are most effective if administered in divided doses or by continuous infusion. Continuous infusion may occur over 24 hours for up to 7 days. It is given primarily at a slower rate, allowing the drug to reach more cancer cells when actively dividing and amenable to cell death. In contrast, cell cycle–nonspecific drugs have a broader impact on cancer cells, as significant cytotoxic effects are exerted on cells at any phase within the cell cycle, including G0. These agents are considered dose-dependent and are most effective when administered by bolus, as the number of cells affected is directly proportional to the amount of drug given. Table 1 displays the classification of the most common chemotherapy agents according to their mechanism of action and effect on the cell cycle (Healy, 2024; Rogers & Brashers, 2023). Table 1 Classification of Chemotherapy Agents Category Chemotherapy Agents Mechanism of Action Effect on Cell Cycle Antimetabolites 5-fluorouracil (Adrucil), capecitabine (Xeloda) Alkylating agents Antitumor antibiotics Vinca alkaloids Taxanes (microtubule agents) Podophyllotoxins (Amjad et al., 2023; Hinkle et al., 2021) Since cancer cells divide rapidly, chemotherapy is primed to target rapidly dividing cells, impacting normal cells that divide quickly, such as the gastrointestinal (GI) tract, skin/hair cells, and bone marrow. The most common side effects include lowering blood counts, nausea, vomiting, diarrhea, hair loss, fatigue, and mucositis (refer to Table 2). In addition, several chemotherapy agents have dose-limiting toxicities or severe side effects that lead to dose reduction or treatment discontinuation. Dose-limiting toxicities require early recognition and intervention to minimize hospital admissions, readmissions, and infections and improve the patient’s survival. There is an extensive range of acute and long-term toxicities associated with chemotherapy agents. The comprehensive list extends well beyond the scope of this module; however, the most common side effects will be discussed (Amjad et al., 2023; Hinkle et al., 2021; Olsen et al., 2019; Rogers & Brashers, 2023). Table 2 Common Chemotherapy Side Effects System Side Effects Hematopoietic Integumentary Gastrointestinal Genitourinary Neurologic Cardiovascular Vascular Pulmonary Reproductive Endocrine Psychiatric Latent effects (Amjad et al., 2023; Hinkle et al., 2021; Olsen et al., 2019; Rogers & Brashers, 2023) Bone marrow suppression denotes a decline in the three major elements of the blood: neutropenia (white blood cells [WBCs]), anemia (red blood cells), and thrombocytopenia (platelets). Patients are most likely to experience their chemotherapy nadir (when the cell counts are at their lowest point) approximately 7–10 days after each chemotherapy dose. When the body’s natural immune defense is suppressed, the patient is considered neutropenic, and their ability to respond to everyday bacteria, viruses, or pathogens is impaired. As a result, the patient is highly susceptible to illness and bloodstream infections (bacteremia or sepsis). Neutropenia is defined by an absolute neutrophil count (ANC) of 1,500/mm3 or below and is a primary dose-limiting toxicity of chemotherapy. The risk for infection is heightened when the ANC drops below 500/mm3. The most common sign of infection in a neutropenic patient is a fever, and patients need to be monitored and treated promptly for fevers or other signs of infection. Febrile neutropenia is a medical emergency requiring prompt evaluation, workup, and empiric antibiotics. Patients should be counseled on ways to avoid infection, such as thorough handwashing, hygiene, and avoiding those with illness. Patients should also avoid eating raw meats, seafood, eggs, and unwashed vegetables when neutropenic due to the risk of acquiring foodborne illness (DeVita et al., 2023; Nettina & Nelson-Tuttle, 2024). Colony-stimulating factors such as filgrastim (Neupogen) or pegfilgrastim (Neulasta) may be given to prevent or treat neutropenia. These injectable agents stimulate the bone marrow to produce WBCs rapidly, thereby reducing the risk of infection and neutropenic fever. According to Olsen and colleagues (2019), there are specific parameters and indications for using a colony-stimulating agent, and these agents are usually well tolerated. The most common side effects include bone pain, particularly in the long bones, such as the hips and sternum, where bone marrow is naturally produced. Patients may use over-the-counter (OTC) pain relievers such as acetaminophen (Tylenol) or nonsteroidal anti-inflammatory drugs (e.g., ibuprofen [Motrin]) for comfort; however, most patients find that antihistamines (e.g., loratadine [Claritin]) provide superior relief from symptoms (Olsen et al., 2019). Critical Consideration: A fever greater than 101°F (38.3°C) in a patient with an ANC below 500/mm3 is a medical emergency requiring immediate intervention and administration of antibiotics. The patient is at risk of sepsis, which can be fatal if untreated. Antibiotics should be administered only after the appropriate blood, wound, and urine cultures have been obtained (Olsen et al., 2019). Anemia is a common consequence of chemotherapy and generally becomes more significant with each successive dose. In addition to low hemoglobin and hematocrit, patients may display signs and symptoms of pallor, fatigue, lethargy, chest pain, shortness of breath (SOB), and weakness. Some patients may benefit from oral iron supplements, folic acid, and an iron-rich diet. Others may require erythropoietin-stimulating agents such as epoetin alfa (Procrit, Epogen), darbepoetin alfa (Aranesp), or blood transfusions. Thrombocytopenia, or a low platelet count, is an effect of chemotherapy caused by suppressing megakaryocytes in the blood, impairing the body’s ability to form a blood clot, and heightening the risk of bleeding. Platelets stop bleeding by clumping and forming plaques in blood vessel injuries, such as cuts, lacerations, and other wounds. The risk of bleeding is present when a patient’s platelet count falls below 50,000/mm3, becomes a high risk if their count falls below 20,000/mm3, and becomes a critical risk if their count falls below 10,000/mm3. Patients may require platelet transfusions if their count drops below 20,000/mm3. Thrombocytopenia is particularly dangerous for patients on anticoagulation therapy. Patients should be monitored for easy bruising, petechiae, epistaxis, gum bleeding, hematuria, rectal bleeding, or wounds that do not heal. Patients should be counseled on strategies to prevent injury, such as avoiding shaving with razors, rectal suppositories, or participating in activities that increase the risk of injury (e.g., contact sports, skiing, and horseback riding). Patients and caregivers must also be counseled on the risk of hemorrhage, paying particular attention to any acute head injuries. For example, those involved in motor vehicle collisions or falls are at risk for subarachnoid or intracerebral hemorrhage. These patients always require emergent evaluation by a clinician and will often undergo radiographic imaging of the head to rule out bleeding, such as a CT scan (Brant et al., 2024; Nettina & Nelson-Tuttle, 2024; Olsen et al., 2019). The GI system is often impacted by chemotherapy, causing side effects such as nausea, vomiting, diarrhea, constipation, anorexia, and mucositis. These manifestations can lead to severe complications such as dehydration (fluid volume deficit), electrolyte disturbances, protein deficit, weight loss, and cachexia (muscle wasting). Anorexia, or loss of appetite, usually has a multifactorial etiology in patients with cancer receiving chemotherapy. Patients frequently report dysgeusia (absent or altered taste) since chemotherapy changes the reproduction of taste buds. Dysgeusia can lead to inadequate nutrition and protein intake, resulting in weight loss and cancer-provoked cachexia. Mucositis refers to inflammation and ulceration of the mucous membranes lining the mouth and other parts of the GI tract. This condition commonly presents with initial oral burning and discomfort, followed by the appearance of open sores and ulcers throughout the oral cavity, extending down the GI tract to the anus. Mucositis can significantly impair oral intake due to pain, thrush, and dysphagia (difficulty swallowing). Consistent oral hygiene with baking soda–based oral rinses and normal saline solution is critical for promoting healing, stimulating cell turnover, and avoiding infection. Patients should be encouraged to use oral agents to promote cleansing, debridement, and comfort, and alcohol-based mouthwashes should be avoided due to the potential for irritation. Painful mucositis can also be treated with oral solutions, including antifungal suspensions (i.e., nystatin [Mycostatin]) to treat thrush and lidocaine (Xylocaine; numbing analgesia) for comfort. Nurses must monitor for diarrhea in patients on chemotherapy, as life-threatening diarrhea has been reported with some chemotherapeutic agents, such as irinotecan (Camptosar). Irinotecan (Camptosar) has such a high incidence of severe diarrhea that it has been nicknamed “I ran to the can” by oncology personnel for decades. Diarrhea can be life-threatening due to associated fluid and electrolyte depletion, as electrolyte imbalance can induce life-threatening changes in cardiac function. In contrast, constipation can exacerbate existing anorexia and nausea and place patients at risk for bowel obstructions. Nurses should assess and monitor patients for changes in bowel habits before, during, and after each treatment (Brant et al., 2024; Nettina & Nelson-Tuttle, 2024; Olsen et al., 2019). Chemotherapy-induced nausea and vomiting patterns include acute, delayed, and anticipatory nausea. The researchers describe acute nausea as occurring 0–24 hours after chemotherapy administration, whereas delayed nausea often occurs several days after chemotherapy. Anticipatory nausea is a conditioned response generated from the repeated association between a treatment and prior nausea and vomiting episodes. It may be triggered by entering the hospital environment, smells, or sounds. Anticipatory nausea can and should be prevented with adequate antiemetic control. Prevention of nausea and vomiting is the goal, so the prescribed nausea management plan is directly correlated with the drug’s emetogenicity level (DeVita et al., 2023). Each chemotherapeutic agent is classified according to its emetogenic potential (risk for nausea), ranked low to high risk. Several major oncology groups have published consensus reports and evidence-based guidelines on preventing chemotherapy-induced emesis, such as the NCCN, ONS, and ASCO. Highly emetogenic drugs require enhanced antiemetic control with dual or triple therapy targeting various nausea receptors. Before starting any chemotherapy regimen, the nurse must thoroughly assess the patient’s prior experiences with nausea to tailor interventions to their needs. Identifying factors that stimulated nausea in the past can help prevent anticipatory nausea and improve the patient’s overall experience and outcome. Most chemotherapy regimens recommend premedicating with an appropriate antiemetic and prescribing an as-needed agent for home use. The most common antiemetic medications include first-generation serotonin (5-HT3) receptor antagonists (i.e., ondansetron [Zofran]), first-generation antipsychotics (i.e., prochlorperazine [Compazine]), and NK1 receptor antagonists (i.e., aprepitant [Emend]). Aprepitant (Emend, Cinvanti) must be prescribed with a 5-HT3 antagonist and may be used with a corticosteroid (i.e., dexamethasone [Decadron]), which potentiates its effects. For enhanced control of severe nausea and vomiting uncontrolled by the prior options, olanzapine (Zyprexa), a second-generation (i.e., atypical) antipsychotic that exerts its action primarily on dopamine and serotonin receptors, may be used. Nurses must teach patients and caregivers the importance of responding promptly to breakthrough nausea with as-needed medications before vomiting occurs. Patients should also be counseled on the benefits of consuming small, frequent, high-calorie meals instead of trying to consume three large meals daily. A four-drug combination is recommended by ASCO (Hesketh et al., 2020) for nausea/emesis prophylaxis in adults receiving highly emetic antineoplastic agents such as cisplatin (Platinol AQ) or anthracyclines. This regimen includes a 5-HT3 receptor antagonist, NK1 receptor antagonist, dexamethasone (Decadron), and olanzapine (Zyprexa) on day 1, followed by dexamethasone (Decadron) and olanzapine (Zyprexa) on days 2–4. Adults receiving moderate-risk medications (e.g., carboplatin [Kyxata]) or high-dose chemotherapy with bone marrow or stem-cell transplantation should be offered a two- or three-drug combination on day 1, including a 5-HT3 receptor antagonist, dexamethasone (Decadron), and possibly an NK1 receptor antagonist (optional). Similarly, patients receiving moderate to high-emetic-risk radiation therapy should be offered prophylactic 5-HT3 receptor antagonist with or without dexamethasone (Decadron) before each fraction and the following day. For low to minimal-emetic risk radiation therapy, breakthrough treatment with dexamethasone (Decadron), a 5-HT3 receptor antagonist, or a dopamine-receptor antagonist (olanzapine [Zyprexa]) should be offered. For antineoplastics that tend to cause delayed nausea (e.g., cyclophosphamide [Cytoxan], doxorubicin [Adriamycin, Doxil]), dexamethasone (Decadron) may be offered on days 2–3. Lorazepam (Ativan) can also be used as an adjunct medication. The evidence supporting the use of cannabinoids for prophylaxis is insufficient, but dronabinol (Marinol, Syndros) and nabilone (Cesamet) are still recommended for the treatment of nausea/vomiting. Pediatric patients receiving highly emetic antineoplastic agents should be offered a three-drug combination including a 5-HT3 receptor antagonist, dexamethasone (Decadron), and an NK1 receptor antagonist (aprepitant [Emend] or fosaprepitant [Focinvex]) if able. Those unable to receive dexamethasone (Decadron) should be offered a combination of palonosetron (Aloxi, Posfrea) and an NK1 receptor antagonist. Pediatric patients receiving moderately emetic antineoplastic agents should be offered a combination of a 5-HT3 receptor antagonist and dexamethasone (Decadron). Those unable to receive dexamethasone (Decadron) should be offered a 5-HT3 receptor antagonist and an NK1 receptor antagonist instead. Pediatric patients receiving low-emetic-risk antineoplastic agents should be offered ondansetron (Zofran) or granisetron (Kytril). For more evidence-based information, refer to ONS guidelines on controlling chemotherapy-induced nausea and vomiting (CINV) or NCCN guidelines for supportive care and antiemesis (DeVita et al., 2023; Hesketh et al., 2020; NCCN, n.d.-b; ONS, 2025). Alopecia, or hair loss, deserves special attention because it can cause significant emotional distress to patients. Chemotherapy-induced hair loss generally begins as hair thinning about 7–15 days after the first dose. It is due to damage to the dividing hair matrix cells, which causes the hair shaft to break at the follicular orifice or hair bulb. The degree of hair loss depends on the chemotherapy agent, the dose, and the administration schedule. Not all chemotherapy agents cause hair loss, so it is possible to undergo chemotherapy treatment without losing hair. However, certain regimens cause complete hair loss, specifically firstline chemotherapy for breast, ovarian, and endometrial cancers. Some agents that are more likely to cause total alopecia include paclitaxel (Taxol), bleomycin (Blenoxane), dactinomycin (Cosmegan), topotecan (Hycamtin), and docetaxel (Taxotere). In contrast, much less hair loss occurs with 5-fluorouracil (Adrucil), etoposide (Vepesid), gemcitabine (Gemzar), methotrexate (Trexall), and ifosfamide (Ifex). Patients should be counseled that their hair will begin to regrow within a few weeks following the cessation of chemotherapy, as permanent alopecia following chemotherapy is rare (Olsen et al., 2019). Novel treatments such as scalp hypothermia (i.e., “cold caps” or “scalp cooling”) worn during the infusion of certain alopecia-inducing chemotherapies have demonstrated efficacy in reducing alopecia. There is the strongest evidence of scalp cooling reducing hair loss in individuals being treated for breast cancer, but it also appears to be effective in other solid tumors, such as ovarian and prostate cancer. Options include manual (unregulated) cold caps and two FDA-approved scalp cooling systems, Dignicap and Paxman. Scalp cooling works by constricting the blood vessels of the scalp, reducing the amount of chemotherapy medicine that reaches the hair follicles. The cold is postulated to decrease the activity of the hair follicles, which slows cell division and makes the follicles less affected by chemotherapy. A meta-analysis of 10 studies indicated a 66% reduction in the risk of chemotherapy-induced alopecia. Unfortunately, cooling cap treatment is not available at all infusion centers and may not be covered by insurance, although it was reassigned for payment with the Centers for Medicare and Medicaid Services (CMS) using the current procedural technology (CPT) code 06627. Cold caps may cost between $1,500 and $3,000 per patient, depending on the number of treatment cycles. Some institutions offer financial support, and grants are available from the Hair to Stay Foundation and Sharsheret. The nonprofit organization, The Rapunzel Project, also helps people access scalp cooling treatment(ACS, 2026b; Rugo & van den Hurk, 2024). Chemotherapy-induced peripheral neuropathy (CIPN) can be a severe side effect commonly associated with specific chemotherapeutic agents such as the platinum agents (cisplatin [Kemoplat], oxaliplatin [Eloxatin]), the taxanes (paclitaxel [Onxol, Taxol], docetaxel [Taxotere]), vinca alkaloids (vincristine [Oncovin], vinblastine [Velban]), and bortezomib (Velcade). CIPN results in symptoms of paresthesia (numbness and tingling), weakness, and burning pain. CIPN initially affects the most distal points of the body, such as the fingertips and toes, and progressively moves proximally toward the midline as the damage progresses. Symptoms are usually symmetric, and in severe cases, patients may lose the sensation of the fingers, hands, toes, and feet altogether, leading to dysfunction in the ability to grasp or hold items and gait disturbance, including imbalance and falls. CIPN is dose-dependent and progressive during treatment but can also have a “coasting phenomenon” after treatment ends, with symptoms becoming more prominent after discontinuing the offending agent. The presence of pain, sensory changes, and weakness during treatment generally leads to chemotherapy dose reductions, changes in treatment protocols, or termination of the therapeutic agent entirely. The morbidity associated with CIPN can lead to pronounced alterations in quality of life and loss of independence with activities of daily living. Although its etiology is not well established or understood, CIPN can be severe, debilitating, and dose-limiting toxicity of certain chemotherapies (Lustberg, 2026). Currently, no medications or supplements have been shown to prevent CIPN definitively. Exercising regularly, cryotherapy, compression therapy, and treating preexisting medical conditions (e.g., vitamin D deficiency) may reduce the risk of CIPN. The evidence on acupuncture and glucocorticoids is mixed, so this is typically not recommended prophylactically. Management of CIPN is equally complex, and effective treatment options are limited. Pharmacologic treatment focuses on symptom relief, even though many agents are not consistently effective. OTC pain medications, menthol creams, capsaicin cream, or lidocaine patches may be used for comfort. Current recommendations do not suggest the use of gabapentin (Neurontin) or other antiseizure medications to treat or prevent CIPN. Some patients may find relief from selective serotonin-norepinephrine reuptake inhibitors (SNRIs) such as duloxetine (Cymbalta) or other antidepressants. Exercise and acupuncture may also be helpful for patients with CIPN, although the evidence is not robust. An electrocutaneous device called a scrambler is also being studied for the treatment of CIPN. Improvement in function and resolution of symptoms often occurs gradually over time, but in some cases, nerve damage may be permanent (Lustberg, 2026). Chemotherapy-induced cardiotoxicity is a severe complication that limits the use of specific chemotherapy agents and may include life-threatening dysrhythmias, conduction disturbances, cardiomyopathies, pericarditis or myocarditis, and pericardial effusions. Anthracyclines, such as doxorubicin (Adriamycin), daunorubicin (Cerubidine), epirubicin (Ellence), and idarubicin (Idamycin), are some of the most prominent cardiac offenders. Acute cardiotoxicities that develop during treatment or immediately after are typically reversible and manageable. However, chronic cardiotoxicities, such as weakened heart muscle and congestive heart failure (CHF), may manifest for long periods, up to decades after treatment ends. Chronic cardiotoxicity substantially affects overall morbidity and mortality and requires long-term management. The cumulative dose of doxorubicin (Adriamycin) is essential to monitor as it can dictate the potential for future cardiotoxicity. The cumulative dose should never exceed 500 mg/m2, or the risk of CHF will rise significantly. Nurses must remain vigilant when administering cardiotoxic chemotherapy agents to ensure cumulative doses do not exceed 500 mg/m2. Patients must undergo a baseline cardiac evaluation with an echocardiogram or a multigated acquisition (MUGA) scan to assess their left ventricular ejection fraction (LVEF). LVEF is subsequently monitored at specific intervals during treatment and as clinically indicated if symptoms develop. Signs and symptoms of cardiac dysfunction may include dyspnea, peripheral edema, chest pain (angina), and lightheadedness. Early detection and immediate proper medication can reverse the condition or minimize long-term effects (Olsen et al., 2019). Chemotherapy may induce reproductive alterations in all patients, including sexual dysfunction, loss of libido, and psychological effects associated with distorted body image. For instance, surgical intervention with orchiectomy (removal of the testis in males with testicular cancer) or mastectomy (removal of breasts in females with breast cancer) may negatively impact a patient’s self-image and, in turn, impair sexual health and intimate relationships. Side effects of chemotherapy can induce other body image distortions secondary to alopecia, vaginal dryness from hormonal therapy, and erectile dysfunction. For females, chemotherapy can cause amenorrhea (permanent or temporary loss of menses), damage the ovarian follicles, or cause ovarian fibrosis. It can also induce premature ovarian failure, leading to infertility and menopause, with associated hot flashes, night sweats, and weight gain. Males may experience low sperm counts, poor sperm motility, sterility, and sexual dysfunction. Nurses are responsible for ensuring patients are educated honestly and openly about the potential effects of chemotherapy, including the possibility of fertility loss. Patients or couples may desire fertility preservation for future family planning with cryopreservation of embryos or sperm (Brant et al., 2024; Liede et al., 2018). Patients must be educated on safe sex practices while undergoing cancer treatment. Patients and their partners must understand the importance of taking precautions to avoid pregnancy while receiving treatment, as chemotherapy can induce fetal harm and congenital disabilities if the fetus is exposed during certain stages of development. Females must be educated that it is still possible to conceive despite not having regular menstrual cycles during chemotherapy, so proper precautions must be taken to avoid pregnancy during treatment. Patients should be advised to abstain from sexual intercourse within 24–48 hours after chemotherapy administration due to the known presence of chemotherapy in bodily fluids. Patients should use a barrier method, such as a condom, to prevent unnecessary exposure to partners via bodily fluids. Finally, breastfeeding is generally contraindicated during chemotherapy due to drug excretion in breast milk (Brant et al., 2024; Olsen et al., 2019). Hypersensitivity Reactions A hypersensitivity reaction (HSR) occurs when a foreign substance overstimulates the immune system, which creates antibodies that trigger an immune response. Some chemotherapeutic agents commonly associated with HSRs include paclitaxel (Taxol), L-asparaginase (Erwinaze), and bleomycin (Blenoxane). HSRs can occur during the initial chemotherapy infusion or after subsequent administrations of the same agent. Most HSRs arise during the first 15 minutes of the infusion, but reactions can happen outside this time frame. Initial signs and symptoms can include hives, urticaria, pruritus, swelling, back pain, facial flushing, rhinitis, abdominal cramping, chills, and anxiety. An HSR may suddenly progress to life-threatening hypotension, bronchospasm, angioedema (swelling of the oral cavity, lips, or tongue), and anaphylaxis. In these cases, epinephrine 0.1–0.5 mg (1:10,000 solution for adult patients) may need to be administered by IV push or subcutaneous injection until emergency personnel arrive. Premedicating patients with corticosteroids, antihistamines, acetaminophen, and histamine 2-receptor antagonists can reduce the risk of HSRs. Oncology nurses must remain vigilant for signs of HSR and ensure they are prepared to intervene immediately. If an HSR is suspected, the infusion must be stopped immediately, and the patient must be monitored closely. Additional nursing interventions include notifying the provider, monitoring vital signs, applying supplemental oxygen, and administering normal saline and other emergency medications as indicated per institution policy or provider order. Nurses should become familiar with their institution’s specific chemotherapy hypersensitivity protocols and guidelines for further information and instruction (Nettina & Nelson-Tuttle, 2024). Safety and Exposure Cytotoxic drugs can also be hazardous to HCPs, so adhering to hazardous drug handling standards is critical to minimize occupational exposure. Exposure to hazardous medications is linked to increased risks for several types of malignancies, and exposure can occur through various sources, including workplace surface contamination. According to the 2019 ASCO safe handling of hazardous drugs standards, HCPs must wear appropriate PPE whenever there is a risk of chemotherapy being released into the environment, such as when preparing or mixing chemotherapy, spiking/priming IV tubing, administering the drug, and handling body fluids or chemotherapy spills. These guidelines also describe hazardous drug handling as posing reproductive risks, so HCPs who are pregnant, breastfeeding, or trying to conceive must notify their employer. These individuals should not be handling hazardous medications such as chemotherapy (Celano et al., 2019; Olsen et al., 2019; ONS, 2018). The guidelines indicate chemotherapy medications must be mixed, spiked, and primed under an approved filtered hood to reduce the risk of aerosolized exposure. Gloves tested for use with hazardous drugs are required, and the reuse of gloves is prohibited. HCPs should wear disposable, lint-free gowns made of low-permeability fabric when administering chemotherapy. Spill kits should be available where chemotherapy is stored, prepared, and distributed. Gloves and gowns should be discarded in leak-proof containers marked as contaminated or hazardous waste. Linens or clothes contaminated with chemotherapy or bodily fluids from patients who have received chemotherapy within 48 hours should be contained in specially marked hazardous waste bags. If any chemotherapy spills on clothes in the clinic, clothing should be thrown away or double-bagged in a plastic bag sealed for transport home. The clothing must be washed separately in hot water with regular detergent. The ONS has two standards that address the education of HCPs who administer and care for patients receiving chemotherapy, biotherapy, and immunotherapy agents. The standards support RNs as the minimum appropriately licensed professionals who can administer chemotherapy and biotherapy. They recommend educational requirements for HCPs, regardless of treatment indications, clinical settings, routes of administration, and patient population. Due to the unique safety considerations of these drugs, specialized education is needed for all individuals who administer chemotherapy and other anticancer agents. The ONS offers online courses for initial didactic preparation and knowledge maintenance for HCPs administering chemotherapy and immune-based treatments. However, each institution or practice must determine how to assess nursing competence in performing various chemotherapy-related skills (Celano et al., 2019; Olsen et al., 2019; ONS, 2018; Siegel et al., 2024). Immunotherapy Immunotherapy, or biologic therapy, stimulates the body’s immune system to fight cancer. Immunotherapy has emerged as a vital cancer treatment modality, and intensive research regarding its clinical efficacy is ongoing. Dr. William Coley discovered the connection between cancer and the immune system over 100 years ago. He was deemed the father of immunotherapy after discovering that malignant tumors disappear in patients who contracted acute streptococcal infections. Immunotherapy produces antitumor effects through the action of natural host defense mechanisms. Since the immune system’s primary function is to detect and eliminate foreign pathogens, immunotherapy strives to modify the patient’s immune defenses to become more sensitive to cancer cells by learning how to identify, attack, and kill them to promote tumor regression. Checkpoint inhibitors have been developed to enable the immune system to effectively harness disease-fighting immune cells (T-cells) against cancer. These are systemic treatments, and they work differently from chemotherapy as they are precise in their action. Some forms of immunotherapy are so highly specialized that they only target a single receptor on the surface of tumor cells. Immunotherapies are frequently combined with other treatment modalities. Several immunotherapies are currently used to treat cancer, including monoclonal antibodies, immune checkpoint inhibitors, CAR T-cell therapy, cancer vaccines, cytokines, and immunomodulators. While immunotherapy represents a promising cancer treatment approach, it is still not equally effective for all cancers at this stage in development (ACS, 2025c; Nettina & Nelson-Tuttle, 2024; Sengupta, 2017; Shoushtari & Johnson, 2024; Yarbro et al., 2018). Overview of the Immune System The immune system is a collection of cells, tissues, and organs that work together to defend the body against attacks by “foreign” invaders such as microbes, viruses, parasites, or other pathogens. The immune system strives to prevent invasion and protect against illness and infection by seeking out and destroying pathogens. The key to a healthy immune system is its ability to distinguish between the body’s cells (“self”) and foreign cells (“nonself”). The immune system cells launch an attack when they encounter anything that appears foreign. Any substance capable of triggering an immune response is called an antigen. An antigen can be a microbe, such as a virus or bacteria; all antigens carry marker molecules identifying them as foreign. The organs of the immune system are positioned throughout the body. They are called lymphoid organs because they house lymphocytes, the small WBCs that are the key performers of the adaptive immune system. There are two main types of lymphocytes: B-cells and T-cells. B-cells work by secreting antibodies into the body’s bloodstream and lymphatic system, which then attack antigens circulating in the bloodstream. T-cells attack the target cells (the infected or cancerous cells). An antigen matches an antibody, much like a key fits a lock (Hinkle et al., 2021; Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023). Mounting an Immune Response There are two main types of immune responses: innate (or nonspecific) and acquired (or adaptive) immunity. Innate immunity, also known as natural immunity, is present from birth. It is considered the first line of defense against pathogens and is a nonspecific form of immunity that is activated immediately and rapidly in response to invading pathogens. Innate immunity is always present in the body, prepared to attack pathogens, and does not generate immunologic memory. In other words, the innate immune system has no memory of prior pathogens and responds nonspecifically to each attack. It includes physical barriers (skin and mucous membranes), mechanical barriers (coughing and sneezing), chemical barriers (tears and sweat), inflammatory responses (production of specialized WBCs such as neutrophils, macrophages, and monocytes), complement activation, and the production of natural killer (NK) cells (large granular lymphocytes). An example of innate immunity includes the development of redness and swelling around a wound caused by lymphocytes invading the wound, working to keep microbes out and heal the injury before further damage or infection occurs (DeVita et al., 2023; Hinkle et al., 2021; Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023). Adaptive immunity is the second line of defense and is highly specific, as it responds individually to every pathogen it encounters. Mediated by T-cells and B-cells, the adaptive immune system responds if invading pathogens breach innate immune mechanisms. The adaptive immune system has immunologic memory and specificity, meaning it “remembers” prior attacks and can develop a repeat-specified response. An example of adaptive immunity is a vaccination, which leads to the creation of antibodies against a virus to prevent the acquisition of the illness later. If that virus tries to invade the body, the adaptive immune system is activated to produce additional antibodies quickly to address the infection and prevent illness. The acquired immune system responds more slowly than the innate immune system. There are three types of adaptive immunity: humoral immunity, cell-mediated immunity, and T-regulatory cells. Humoral immunity is mediated primarily by B-cells and results in the production of immunoglobulins (Igs). T-cells and their cytokine products mediate cell-mediated immunity. It does not involve an antibody but instead includes cytotoxic T-cells (usually CD8) and helper T-cells (usually CD4). T-regulatory cells, also known as suppressor T-cells, display the markers CD4 and CD25 and limit the activity of other immune effector cells. Ultimately, their primary role is to prevent damage to normal tissues and constrain the inflammatory response that can occur with infections (DeVita et al., 2023; Hinkle et al., 2021; Nettina & Nelson-Tuttle, 2024; Rogers & Brashers, 2023). When immune surveillance fails, cancerous tumors form. There are various schools of thought on the exact etiology of tumor escape mechanisms, but they broadly encompass the following (Miliotou & Papadopoulou, 2018; Sasikumar & Ramachandra, 2018): Immune Checkpoint Inhibitors Immune checkpoint inhibitors work by blocking the receptors that cancer cells use to inactivate T-cells. T-cells may better differentiate between healthy and cancer cells when this signal is blocked, augmenting the immune system’s response to cancer. Checkpoint inhibitors are presently categorized as programmed cell death-1 (PD-1)/PD-ligand 1 (PD-L1) inhibitors and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) inhibitors. By blocking tumor cells from inactivating T-cells, these medications allow the T-cells to remain active and fight the tumor cells (NCI, 2022; Shoushtari & Johnson, 2024). PD-1 Inhibitors/PD-L1 Inhibitors PD-1 is a transmembrane protein expressed on the surface of circulating T-cells, B-cells, and NK-cells and is used to recognize “self” antigens from “non-self.” PD-1 helps keep the immune response working properly. When PD-1 attaches to PD-L1, an inhibitory ligand expressed on some normal and cancer cells, it acts as an “off switch” to keep them from attacking other cells in the body. The binding of PD-1 to PD-L1 signals the T-cell to leave the neighboring cells alone, including cancer cells. Some cancer cells have large amounts of PD-L1, which helps them evade immune attacks. PD-1 and PD-L1 inhibitors are monoclonal antibodies developed to prevent the formation of this complex. When administered, these drugs block PD-1 or PD-L1, disrupting the immune system’s brakes, allowing T cells to target cancer cells. Monoclonal antibodies targeting PD-1 and PD-L1 inhibitors are approved for various cancers and indications such as melanoma, lung cancer, and solid tumors with microsatellite instability-high (MSI-H) mutations, and mismatch repair-deficient (dMMR) mutations. The approval of pembrolizumab (Keytruda) in 2015 for MSI-H or dMMR solid tumors was the first time a drug had been approved based on a biomarker test and not an anatomic tumor site, which ignited a shift in the cancer treatment trajectory. Since 2015, the indications of pembrolizumab (Keytruda) have expanded to numerous cancers such as biliary tract cancer, bladder cancer, colorectal cancer, endometrial carcinoma, hepatocellular carcinoma, lung cancer, renal cell carcinoma, triple-negative breast cancer, recurrent or metastatic head and neck squamous cell carcinoma, Hodgkin lymphoma, gastric or esophageal cancer, primary mediastinal B-cell lymphoma, melanoma, and recurrent or metastatic cervical cancer. Aside from pembrolizumab (Keytruda), examples of PD-1 inhibitors include nivolumab (Opdivo), cemiplimab (Libtayo), dostarlimab (Jemperli), penpulimab (Verzenio), retifanlimab (Zynyz), tislelizumab (Tevimbra), and toripalimab (Loqtorzi). Examples of PD-L1 inhibitors include atezolizumab (Tecentriq), avelumab (Bavencio), cosibelimab (Unloxcyt), and durvalumab (Imfinzi; ACS, 2025b; NCI, 2022; Shoushtari & Johnson, 2024; UpToDate Lexidrug, n.d.-c). PD-1/PD-L1 inhibitors are generally well tolerated; however, patients can experience severe and possibly life-threatening autoimmune-related adverse effects. Although an autoimmune reaction can affect any organ system, the most common reactions include colitis, hepatitis, endocrinopathies (thyroid and adrenals), pneumonitis, and skin rash. The most common side effects include fatigue, nausea, anorexia, cough, diarrhea, skin rash, and itching (NCI, 2022; Postow & Johnson, 2025; UpToDate Lexidrug, n.d.-c). Chimeric Antigen Receptor (CAR) T-Cell Therapy CAR T-cell therapy is a highly complex and cutting-edge form of immunotherapy that has rapidly expanded in recent years. T-cells are isolated from patients and genetically engineered using an inactive virus to express a CAR that recognizes a tumor-specific antigen. Millions of these modified T-cells, now called CAR T-cells, are grown and injected back into the patient to identify, attach to, and eventually induce cancer cell death (Laubach, 2025; Leukemia & Lymphoma Society [LLS], n.d.). The FDA has approved the following drugs used as CAR T-cell therapy: CAR T-cell therapy poses the risk of multiple potential toxicities that HCPs must be prepared to recognize and respond to quickly. Two significant immune-related adverse effects of CAR T-cell therapy are cytokine release syndrome (CRS) and neurotoxicity. Cytokines are small protein molecules generally activated by a stimulus and induce a response by binding to specific receptors, affecting the growth and differentiation of WBCs. They also regulate immune and inflammatory responses by enhancing cytotoxic activity, secreting additional mediators, and amplifying the immune response when stimulated. CRS is characterized by fever, nausea, vomiting, diarrhea, tachycardia, hypotension, tachypnea, and rash. These signs and symptoms occur approximately 1–14 days after the treatment and are a by-product of cell lysis. As cancer cells die, they release cytokines into the body, stimulating these manifestations. Some patients may require hospitalization for supportive care, but most are managed in the outpatient setting. Tocilizumab (Actemra) has been approved for use in patients over the age of 2 with CAR T-cell-induced life-threatening CRS. Complications of CRS may include renal dysfunction requiring dialysis, acute respiratory distress syndrome (ARDS), or a decline in cardiac ejection fraction (EF) or stroke volume (SV). A rare but severe effect reported with these medications is disseminated intravascular coagulation (DIC), a potentially life-threatening condition in which blood clots form in small blood vessels throughout the body in combination with associated bleeding. These blood clots can block blood flow and induce harm to major organs such as the kidneys and liver. Neurotoxicity may manifest as delirium, confusion, seizures, tremors, encephalopathy, incontinence, and paralysis. The onset of these manifestations is unique and considered biphasic, as symptoms generally occur concurrently with fevers or sedating medication during phase 1 (days 0–5) or phase 2 (after day 5). Manifestations are usually reversible and last about 2 to 4 days but can persist for a few weeks (Laubach, 2025; LLS, n.d.). In addition to inhibition of the programmed cell death complex, other inhibitory modulators, such as CTLA-4, have been worthwhile targets in immunotherapy. Inhibitory effects of CTLA-4 occur in the priming phase of the immune response by interfering with signals required for T-cell activation. The binding of CTLA-4 to CD80/86 inhibits T-lymphocyte activation, resulting in a negative feedback signal that decreases immune response. Ipilimumab (Yervoy) and tremelimumab (Imjudo) induce T-cell activation by disabling the CTLA-4 feedback inhibition and activating the immune system. Ipilimumab (Yervoy) is currently FDA-approved for treating colorectal cancer, esophageal carcinoma, non–small cell lung cancer, melanoma, advanced renal cell carcinoma, hepatocellular carcinoma, and malignant pleural mesothelioma. It has demonstrated a synergistic effect with increased T-cell priming via CTLA-4 inhibition in the lymph nodes and cytotoxic activity via PD-1 inhibition in the tumor environment. Tremelimumab (Imjudo) is approved for unresectable hepatocellular carcinoma and metastatic non–small cell lung cancer. Compared to drugs that target PD-1 or PD-L1, serious side effects are much more likely with these agents. Fatigue, skin rash, pruritus, and diarrhea are common. Adverse effects are related to the drug’s ability to induce nonspecific inflammation throughout the body, leading to immune-mediated complications in the lungs (pneumonitis), intestines (enterocolitis), liver (hepatitis), kidneys (nephritis), hormone-making glands (endocrinopathies including thyroiditis and hypophysitis), eyes (uveitis), pancreas (pancreatitis), and so forth. Any organ system can be affected, but most immune-related events are reversible with immunosuppressive steroid treatment. They should be graded according to Common Terminology Criteria for Adverse Events (CTCAE) Version 6 (2020) and managed per specific medication guidelines (Postow & Johnson, 2025; Shoushtari & Johnson, 2024; UpToDate Lexidrug, n.d.-a, n.d.-d). Other types of immunotherapies currently being explored include cancer vaccines and virus immunotherapy, in which viruses are deliberately used to infect cancer cells. This triggers an immune response against the infected cancer cells. There have been four preventive and two therapeutic cancer vaccines approved by the FDA (Cancer Research Institute [CRI], n.d.). Examples of preventive vaccines include: Examples of therapeutic cancer vaccines include Bacillus Calmette-Guerin (TICE BCG), used in patients with early-stage non-muscle invasive bladder cancer, sipuleucel-T (Provenge), used in patients with certain forms of prostate cancer, and talimogene laherparepvec (T-VEC, Imlygic), which is approved for unresectable melanoma. Cancer vaccine side effects vary based on the type of vaccine but may include anorexia, chills, fatigue, headaches, nausea, and myalgia (CRI, n.d.). Targeted Agents In 2003, the completion of the Human Genome Project marked a dramatic shift in the understanding of cancer and other diseases. Project researchers mapped the entire human genetic code and discovered that every human cell comprises 20,000–30,000 genes. As a result, the past few decades have witnessed exploration into novel approaches to treating cancer and drug discovery (National Human Genome Research Institute, 2025). Targeted agents are drugs formulated to attack specific parts of cancer cells to prevent tumor development or shrink existing tumors. Numerous proteins are located on the cellular membranes called growth factor receptors, which connect the external and internal cellular environments and are essential for cell growth and development. Alterations in genes lead to changes in these cellular proteins, stimulating the disruption of routine processes, inducing malfunction, and subsequently sanctioning cancer growth. Developing specialized drugs that block these growth factor receptors has been a massive part of cancer research in recent decades. Targeted therapies impede the specific proteins that promote cancer growth through unique and distinctive pathways. Each type of targeted therapy works a little differently. However, all generally interfere with the ability of the cancer cells to grow, divide, repair, and communicate with other cells, as depicted in Figure 3. Some targeted therapies focus on the external components and function of cancer cells. In contrast, others use small molecules that can enter the cell and disrupt the function of the cells, causing them to die. Others target receptors outside the cell (Gale, 2024; Sengupta, 2017). In summary, targeted therapies can function through any of the following mechanisms: Figure 3 Targeted Agents Mechanism of Action Directing effects at tumor cell growth through specific targets makes these therapies less toxic to normal cells and tissues than traditional chemotherapy. However, the downside is that cancer cells can become resistant since targeted therapies block only specific growth pathways. Cancer cells can circumvent their effects by developing numerous growth patterns and routes. Therefore, targeted therapies are most successful when combined with other cancer treatments, such as chemotherapy or radiation. While combination therapy has been deemed more effective, it promotes increased toxicity and potential side effects. Furthermore, despite significant advancements in targeted agents, they are generally not considered a replacement for traditional therapies. Targeted therapies are typically grouped into monoclonal antibodies and small-molecule inhibitors (Olsen et al., 2019; Sengupta, 2017). Monoclonal Antibodies Therapies that target receptors are also known as monoclonal antibodies. Some monoclonal antibodies are combined with toxins, chemotherapy drugs, and radiation. Once these monoclonal antibodies attach to targets on the surface of cancer cells, the cells take up the cell-killing substances, causing them to die. Cells that do not have the target will not be harmed. Antibodies are part of the adaptive immune system. Typically, the body creates antibodies in response to an antigen (e.g., a protein in a germ) entering the body. The antibodies attach to the antigen to mark it for destruction by the body’s immune system. In a laboratory, scientists analyze specific antigens on the surface of cancer cells (target) to determine a protein to match the antigen. Then, using animal and human proteins, scientists create a unique antibody that will attach to the target antigen just as a key fits a lock. This technology allows treatment to target specific cells, causing less toxicity to healthy cells. Monoclonal antibody therapy can be done only for cancers with identified antigens and the respective antibodies. Monoclonal antibodies work on cancer cells as natural antibodies do by recognizing and binding to the target cells and then alerting other cells in the immune system to the presence of the cancer cells (Manis, 2025; Olsen et al., 2019). Monoclonal antibodies may be used alone or combined with chemotherapy and are administered intravenously. How monoclonal agents are named provides clues about how they work. The ending letters (i.e., stem) of the name indicate what family the drug is from and how the drug works to kill cancer cells. Monoclonal antibodies always end with the stem “-mab.” The “-mab” family is used when receptor targets are overexpressed outside cancer cells. Monoclonal antibodies have an additional layer of classification within their “sub-stem,” representing how the drug was comprised. Murine monoclonals, drugs ending in “-omab” (i.e., ibritumomab [Zevalin]), are composed of 100% mouse components and include conjugated antibodies, which are agents that are physically attached to antitumor agents such as radioisotopes, chemotherapy drugs, toxins, or other biologic agents. Murine monoclonals have the highest risk of HSR due to human response to the high mouse components. Chimeric monoclonals, drugs ending in “-ximab” (i.e., rituximab [Rituxan]), are made primarily of mouse protein with a (lesser) human protein component and, therefore, also have a higher risk of an HSR among patients (Manis, 2025; Olsen et al., 2019). Humanized mouse monoclonals, drugs ending in “-zumab” (i.e., bevacizumab [Avastin]) include a small percentage of mouse components or other proteins, and the rest is human in origin. These medications generally induce less-severe allergic reactions. Finally, human monoclonal antibodies ending in “-umab” (i.e., panitumumab [Vectibix]) are composed entirely of human proteins, and HSRs to these medications are rare. Some monoclonal antibodies have additional information in their naming classification that designates the main target of the drug. For example, the “li” in ipilimumab identifies the immune system as the target, the “tu” in rituximab indicates the target is the tumor, and the “ci” in bevacizumab designates the circulatory system. Table 3 displays an overview of several monoclonal antibodies categorized according to class, target, and mechanism of action. The targets listed in Table 3, column 4, are receptors the drug has been specially designed to block. Two common targets of monoclonal antibodies include epidermal growth factor receptor (EGFR) and vascular endothelial growth factor (VEGF; Manis, 2025; Olsen et al., 2019). Table 3 Categories of Monoclonal Antibodies Category and Composition Stem Examples Target Mechanism of Action Murine: All mouse components -omab ibritumomab (Zevalin) CD20 Chimeric: Primarily made of mouse protein with a (lesser) human protein component -ximab rituximab (Rituxan) CD20 cetuximab (Erbitux) Anti-EGFR Humanized: Includes a small percentage of mouse or other protein, while the rest is human in composition -zumab bevacizumab (Avastin) Anti-VEGF trastuzumab (Herceptin) Anti-HER2 alemtuzumab (Campath) CD52 Human: Fully human (most will also be categorized as immunotherapy as described earlier) -umab panitumumab (Vectibix) (Manis, 2025; Olsen et al., 2019) Due to their high mouse composition, HSRs are common with murine and chimeric monoclonal antibodies. Conjugate monoclonal antibodies present the highest risk for bone marrow suppression and additional side effects since they are attached to a toxic or poisonous agent. Hypersensitivity symptoms can present like chemotherapy HSRs and may be mild with facial flushing, pruritus, and hives. In other cases, they may progress to severe anaphylaxis requiring epinephrine. Other potential adverse effects of monoclonal antibodies include dyspnea and wheezing, fever and chills, headaches, rashes, nausea and vomiting, tachycardia, and bleeding. Since many patients experience rigors, shaking, chills, and fevers during the infusion, all patients should be premedicated with acetaminophen (Tylenol) and an antihistamine such as diphenhydramine (Benadryl). The APRN nurse must remain vigilant and attentive to patients receiving infusions with monoclonal antibodies, as rigors require interruption of the infusion and possibly medication such as meperidine (Demerol) or corticosteroids. Many institutions have policies outlining the importance of slowly titrating the infusion rate of all monoclonal antibodies to reduce the risk of rigors, chills, and fevers. Slowing the infusion rate can also reduce the risk of prolonged rigors (Manis, 2025; Olsen et al., 2019). Antibody-Drug Conjugates Antibody-drug conjugates (ADCs) are an innovative product developed for cancer treatment. They comprise a monoclonal antibody linked to a small molecule drug, usually a cytotoxic agent. There are currently over ten ADCs approved by the FDA for cancer treatment, but over 90 ADCs in clinical development. Examples of ADCs currently approved for use in the United States include mirvetuximab soravtansie (Elahere), tisotumab vedotin (Tivdak), datopotamab deruxtecan (Datroway), gemtuzumab ozogamicin (Mylotarg), brentuximab vedotin (Adcetris), inotuzumab ozogamicin (Besponsa), polatuzumab vedotin (Polivy), trastuzumab emtansine (Kadcyla), trastuzumab deruxtecan (Enhertu), enfortumab vedotin (Padcev), sacituzumab govitecan (Trodelvy), belantamab mafodotin (Blenrep), and loncastuximab tesirine-lpyl (Zynlonta). ADCs can target cancer cells without harming healthy cells and strengthen the cytotoxic potential of monoclonal antibodies. Although currently used for cancer treatment, there is hope that their use can be expanded to other diseases, such as atherosclerosis, bacteremia, and inflammatory diseases (Criscitiello, 2021; Manis, 2025; Pettinato, 2021; Shastry et al., 2023). Angiogenesis Inhibitors Blood vessels carry oxygen and nutrients to the tissues necessary for growth and survival. Tumors also need blood vessels to grow and spread. Angiogenesis is a process that occurs in both healthy and cancerous tissue to form new blood vessels by sending signals. Angiogenesis is modulated primarily through VEGF receptors, which are signal proteins produced to stimulate angiogenesis in normal and cancerous cells. Anti-angiogenesis stops the formation of new blood vessels by blocking the VEGF receptors. Angiogenesis inhibitors, or VEGF-inhibitors, target the blood vessels supplying oxygen to the tumor cells, ultimately causing them to starve by cutting off their nutrient supply. VEGF inhibitors such as bevacizumab (Avastin) disrupt the tumor’s blood supply by interfering with the VEGF receptor. Without a blood supply, tumors stay small and eventually starve. Other angiogenesis inhibitors include interferon (Extavia) and thalidomide (Thalomid; Gale, 2024; Nettina & Nelson-Tuttle, 2024; Olsen et al., 2019). The side effects of VEGF inhibitors can be severe and include bleeding, headaches, hypertension, and protein spilling into the urine (due to increased pressure in the kidneys). Patients require strict monitoring to ensure treatment is administered only if the blood pressure is below 140/90. Many patients require concurrent treatment with antihypertensives to maintain this range. A urinalysis is recommended to assess for proteinuria before each dose, and the results are graded on a scale of 0 to 3+. Patients with protein of 2+ or greater on a urine dipstick test should undergo a further assessment with a 24-hour urine collection. Treatment should be withheld for proteinuria of 2 grams or above on the 24-hour collection. Treatment may resume when proteinuria is below 2 grams per 24 hours but should be discontinued permanently for patients who develop nephrotic syndrome (Nettina & Nelson-Tuttle, 2024; Olsen et al., 2019). Given the risk of bleeding and delayed wound healing, it is contraindicated for patients to receive a VEGF inhibitor within 6 weeks of any surgical procedure. In addition, there is a risk of fistula development, an abnormal connection between two hollow spaces within the body, such as blood vessels, intestines, or other organs. Some VEGF inhibitors also carry a boxed warning of bowel perforation, a hole in the lining of the intestines (Olsen et al., 2019). Patients must be counseled on the importance of monitoring for any of the following signs and symptoms, which can vary in presentation and progression: Small Molecule Inhibitors Small molecule drugs are pills or capsules taken orally that work on specific pathways. As with monoclonal antibodies, the generic naming of small molecule inhibitors is strategically devised to explain how these medications work. Small molecule inhibitors end with the stem “-ib.” Since small molecule inhibitors target processes inside the cell, they must be small enough in molecular weight to enter the cell and interfere with proteins inside and outside the cell (Gale, 2024; Olsen et al., 2019; Shuel, 2022). Some examples include: The FDA has approved several small molecule inhibitors for cancer therapy. The most common molecular targets include TKIs, proteasome inhibitors, apoptosis targets, matrix metalloproteinases (MMPs), and heat shock proteins (HSPs). Poly (ADP-ribose) polymerase (PARP) is a protein that repairs damaged DNA, making it critical in cell growth, regulation, and repair. PARP inhibitors block this, preventing cancer cells from repairing damaged DNA and causing cell death. PARP inhibitors (e.g., olaparib [Lynparza]) have revolutionized how BRCA mutation-positive cancers, such as ovarian and breast cancers, are treated. Over the last few years, PARP inhibitors have gained FDA approval in prostate, pancreatic, primary peritoneal, and fallopian tube cancers, with their indication labels continuing to expand (Shuel, 2022; Jackson Laboratory, 2024; NCI, n.d.). Table 4 displays some small molecule inhibitors used for cancer treatment, although this list is not comprehensive. Table 4 FDA-Approved Small Molecular Inhibitors for Cancer Therapy Molecular Target Drug Specific Target or Pathway Cancer Type Tyrosine kinase inhibitors (TKIs) erlotinib (Tarceva) gefitinib (Iressa) lapatinib (Tykerb) imatinib (Gleevac) everolimus (Afinitor) ruxolitinib (Jafaki) pazopanib (Votrient) regorafenib (Stivarga) vemurafenib (Zelboraf) crizotinib (Xalkori) Proteosomes carfilzomib (Kyprolis) bortezomib (Velcade) Cyclin-dependent kinase inhibitors (CDK 4/6) palbociclib (Ibrance) abemaciclib (Verzenio) ribociclib (Kisqali) ADP-ribose polymerase (PARP) Inhibitors niraparib (Zejula) olaparib (Lynparza) rucaparib (Rubraca) talazoparib (Talzenna) (Jackson Laboratory, 2024; Liu et al., 2022; Olsen et al., 2019; Shuel, 2022) The side effect profile of these agents is distinct from cytotoxic chemotherapy. Oncology nurses should be aware of the toxicities and management of adverse effects. While some of them can induce bone marrow suppression (i.e., palbociclib [Ibrance] and abemaciclib [Verzenio]), these drugs are generally not associated with as much risk for infection and febrile neutropenia as cytotoxic chemotherapies are. The most common toxicities are GI and dermatologic reactions, such as abdominal pain, anorexia, nausea, diarrhea, constipation, redness, swelling, dermatitis/skin rashes, pruritis, dry skin, xerostomia (dry mouth), skin discoloration, hair discoloration, and nail changes. In addition, they can induce toxic effects on the liver and kidneys, so routine laboratory monitoring and clinical assessment are essential (Shuel, 2022). Hormonal Therapy Some cancers require certain hormones to grow. Hormone therapies primarily follow two mechanisms of action: (a) prevent the body from producing the hormones that drive cancer growth and (b) prevent the hormones from reaching and acting on the cancer cells. The most common hormone-dependent cancers are breast and prostate, but other potentially hormone-driven cancers include endometrial (uterine), kidney, and ovarian cancers. The adverse effects of hormone treatment depend on the type of drug and can differ between male and female patients. Side effects generally include hot flashes, night sweats, loss of libido, weight gain, vaginal dryness/atrophic vaginitis, joint aches or pains, mood changes, weight gain, and bone thinning or weakening (osteopenia or osteoporosis). Male patients may also experience erectile dysfunction (inability to have or maintain an erection), a shrinking of the testicles, and gynecomastia (enlargement of breast tissue). Patients on hormonal therapy should be counseled on following a calcium-rich diet with at least 1,200 mg of dietary calcium daily. Patients who cannot get this recommended amount of calcium in their diet should consider calcium supplementation (DeVita et al., 2023; Olsen et al., 2019). Table 5 displays some hormonal therapies used for cancer treatment, although this is not a comprehensive list. Table 5 Hormonal Therapies for Cancer Treatment Category Drug Cancer Type(s) Mechanism of Action Selective estrogen receptor modulators (SERM) tamoxifen (Soltamox) Steroidal antiestrogens/selective estrogen receptor degrader (SERD) fulvestrant (Faslodex) elacestrant (Orserdu) Luteinizing hormone-releasing hormone (LHRH) analog goserelin (Zoladex) leuprolide (Lupron, Eligard) LHRH antagonists degarelix (Firmagon) Aromatase inhibitors (AI) letrozole (Femara) anastrozole (Armidex) exemestane (Aromasin) Anti-androgens bicalutamide (Casodex) Androgen blockers abiraterone (Zytiga) enzalutamide (Xtandi) (DeVita et al., 2023; Olsen et al., 2019) Oral Cancer Drugs An oral cancer drug is any medication taken by mouth (in liquid, tablet, or capsule formulation) to treat cancer. Advancements in cancer treatment have led to the development of many new oral agents that offer the convenience of taking the medication at home, with less time spent traveling to and from medical offices and clinics. However, it is a misconception that oral cancer drugs are less toxic, as they are equally as strong and effective as IV or injected drugs. There has been a surge in the development of novel oral treatments, many of which are not classified as cytotoxic chemotherapy based on their mechanism of action but still require the same precautions and patient education (Olsen et al., 2019). Unique Features of Oral Agents There are several special considerations with oral cancer treatments, as they pose unique safety challenges compared to traditional IV therapies. One of the most challenging issues with oral cancer treatment involves poor adherence, which substantially impacts the success of the treatment, side effects, toxicity, and patient safety. Potential safety issues include a lack of checks and balances in administration, a risk of patient nonadherence, a lack of monitoring techniques, and a shift to patient management of oral chemotherapy. Oral cancer medications are prescribed at defined intervals based on the drug’s mechanism of action, its half-life (the amount of time it takes for 50% of the drug to be excreted from the body), and its side effect profile. Patients must be educated to take the medication as prescribed to ensure a constant level of the drug remains in the body to kill the cancer cells. Even a slight increase or decrease in the dose level can negatively impact the drug’s efficacy or increase side effects. Patients must be counseled not to crush, chew, or split oral cancer pills, as this can affect how the medication works. Establishing a routine can help patients track their medication schedule. Some strategies may include pillboxes that are filled each week, setting pill reminders on smartphones or tablets, enrolling in electronic medication reminders through a pharmacy, or using a simple paper pill diary, marking when the pill was taken to avoid overdosing (Olsen et al., 2019; Weingart et al., 2018). Oral cancer drugs can be costly and may not be covered entirely by insurance and prescription plans. The financial burden is among the most common reasons for nonadherence with oral medications. Patients should be encouraged to speak to their HCP if they have difficulty affording their medication(s) before discontinuing therapy. Some manufacturers offer co-pay assistance programs or have grants to fund free drug/compassionate use offerings. Several states have passed laws requiring insurance companies to cover oral cancer medications similarly to the case with IV cancer treatments (ACS, 2025a). Safe Medication Handling Oral cancer drugs are potentially hazardous and require special handling precautions, especially for caregivers. As described within the 2024 ASCO and ONS chemotherapy administration safety standards, nurses should educate patients and caregivers on drug safety before oral cancer medications are prescribed (Olsen et al., 2019; Siegel et al., 2024). These recommendations are summarized according to the administration standards with the points listed in what follows (Siegel et al., 2024). Critical Consideration. Patients must be properly educated about the oral treatment plan, administration schedule, storage, potential side effects, drug and food interactions, and when and how to contact their provider (Siegel et al., 2024). Chemoprevention Chemoprevention is the use of natural or synthetic substances to reduce the risk of developing cancer or recurrence. Only a few agents have been shown to decrease cancer risk in high-risk individuals. These hormonal therapies used in breast cancer include tamoxifen (Nolvadex) and raloxifene (Evista). Tamoxifen (Nolvadex) was the first chemopreventive agent approved by the FDA for the risk reduction of breast cancer. Tamoxifen (Nolvadex) is a selective estrogen receptor modulator (SERM) widely used to treat premenopausal patients who have already been diagnosed with breast cancer to prevent a recurrence. More recent clinical trials have shown that tamoxifen (Nolvadex) may reduce the risk of breast cancer by nearly 50% in patients who are at high risk but have not yet been diagnosed. Therefore, tamoxifen (Nolvadex) can be used as primary chemoprevention to reduce the risk of cancer recurrence. Tamoxifen (Nolvadex) does carry a risk of the rare yet severe adverse effects of uterine sarcoma and venous thromboembolism/deep venous thrombosis (VTE/DVT). These risks must be considered and patients counseled on them; unilateral extremity swelling or postmenopausal bleeding must also be monitored. Raloxifene (Evista) was initially approved to treat osteoporosis but was found to have a similar impact as chemoprevention for breast cancer without the risk of uterine sarcoma or VTE/DVT (NCI, 2025a). Third-generation aromatase inhibitors such as exemestane (Aromasin) and anastrozole (Arimidex) reduce the risk of hormone receptor (HR)–positive breast cancer in postmenopausal patients. Adverse effects include joint aches and bone thinning (osteopenia or osteoporosis; NCI, 2025a). Fluorouracil (Efudex) and imiquimod (Aldara) cream are approved to prevent nonmelanoma skin cancer. When applied to actinic keratosis, which are premalignant skin lesions, these agents limit the progression of the lesions to skin cancer. These medications are often prescribed to patients after genetic counseling and evaluation of hereditary risk patterns for cancer based on family history and other risk factors (NCI, 2025c). Other domains of chemoprevention include: Clinical Trials Clinical trials include studies of new drugs, devices, or other forms of cancer treatment. Clinical trials are an essential component of cancer care and research, as they comprise a large sector of the field of oncology. Researchers use clinical trials to test the efficacy of new drugs or a new combination of already approved drugs. To acquire FDA approval, all new cancer treatments must undergo a rigorous clinical trial investigation to ensure safety and efficacy. Without clinical trials, there would be no new drug development and advancement in cancer care, such as the novel, cutting-edge field of immunotherapy (ACS, 2025d). References American Cancer Society. (2023). Viruses that can lead to cancer. https://www.cancer.org/healthy/cancer-causes/infectious-agents/infections-that-can-lead-to-cancer/viruses.html American Cancer Society. (2025a). Getting oral chemotherapy. https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/chemotherapy/oral-chemotherapy.html American Cancer Society. (2025b). Immune checkpoint inhibitors and their side effects. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy/immune-checkpoint-inhibitors.html American Cancer Society. (2025c). Immunotherapy. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy.html American Cancer Society. (2025d). Phases of clinical trials. https://www.cancer.org/treatment/treatments-and-side-effects/clinical-trials/what-you-need-to-know/phases-of-clinical-trials.html American Cancer Society. (2026a). Cancer facts & figures 2026. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/2026-cancer-facts-figures.html American Cancer Society. (2026b). Cold caps and scalp cooling. https://www.cancer.org/cancer/managing-cancer/side-effects/hair-skin-nails/hair-loss/cold-caps.html American Society of Clinical Oncology. (n.d.). QOPI certification program. Retrieved February 5, 2026, from https://www.asco.org/practice-patients/quality-improvement/quality-programs/qopi-certification-program Amjad, M. T., Chidharla, A., & Kasi, A. (2023). Cancer chemotherapy. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK564367 Brant, J. M., Cope, D., & Saria, M. (2024). Core curriculum for oncology nursing (7th ed.). Elsevier. Cancer Research Institute. (n.d.). Immunotherapy: Cancer vaccines. Retrieved February 6, 2026, from https://www.cancerresearch.org/treatment-types/cancer-vaccines#treatments Celano, P., Fausel, C. A., Kennedy, E. B., Miller, T. M., Oliver, T. K., Page, R., Ward, J. C., & Zon, R. T. (2019). Safe handling of hazardous drugs: ASCO standards. Journal of Clinical Oncology, 37(7), 598–609. https://doi.org/10.1200/JCO.18.01616 Centers for Disease Control and Prevention. (2024). Clinical overview of HPV. https://www.cdc.gov/hpv/hcp/clinical-overview/index.html Colditz, G. A. (2025). Overview of cancer prevention. UpToDate. Retrieved February 10, 2026, from https://www.uptodate.com/contents/overview-of-cancer-prevention Criscitiello, C., Morganti, S., & Curigliano, G. (2021). Antibody-drug conjugates in solid tumors: a look into novel targets. Journal of Hematology & Oncology, 14, 20. https://doi.org/10.1186/s13045-021-01035-z DeVita, V. T., Lawrence, T. S., & Rosenberg, S. A. (Eds.). (2023). DeVita, Hellman, and Rosenberg’s cancer: Principles and practice of oncology (12th ed.). Wolters Kluwer. Gale, R. P. (2024). Systemic cancer therapy. Merck Manual Professional Version. https://www.merckmanuals.com/professional/hematology-and-oncology/principles-of-cancer-therapy/systemic-cancer-therapy Healy, M. (2024). Chemotherapy: The basics. OncoLink. https://www.oncolink.org/cancer-treatment/cancer-medications/overview/chemotherapy-the-basics Hesketh, P. J., Kris, M. G., Basch, E., Bohlke, K., Barbour, S. Y., Clark-Snow, R. A., Danso, M. A., Dennis, K., Dupuis , L. L., Dusetzina, S. B., Eng, C., Feyer, P. C., Jordan, K., Noonan, K., Sparacio, D., & Lyman, G. H. (2020). Antiemetics: ASCO guideline update. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology, 38(24), 2782–2797. https://doi.org/10.1200/JCO.20.01296 Hinkle, J. L., Cheever, K. H., & Overbaugh, K. (2021). Brunner & Suddarth’s textbook of medical-surgical nursing (15th ed.). Wolters Kluwer. Konieczkowski, D., Johannessen, C. M., & Garraway, L. A. (2018). A convergence-based framework for cancer drug resistance. Cancer Cell, 33(5), 801–815. https://doi.org/10.1016/j.ccell.2018.03.025 The Jackson Laboratory. (2024). PARP inhibitors: Overview and indications. Retrieved February 6, 2026, from https://www.jax.org/education-and-learning/clinical-and-continuing-education/clinical-topics/tumor-testing/parpi-detail Laubach, J. P. (2025). Multiple myeloma: Treatment of second or later relapse. UpToDate. Retrieved February 10, 2026, from https://www.uptodate.com/contents/multiple-myeloma-treatment-of-second-or-later-relapse Lei, Z. N., Tian, Q., Teng, Q. X., Wurpel, J. N. D., Zeng, L., Pan, Y., & Chen, Z. S. (2023). Understanding and targeting resistance mechanisms in cancer. MedComm, 4(3), e265. https://doi.org/10.1002/mco2.265 Leukemia & Lymphoma Society. (n.d.). Chimeric antigen receptor (CAR) T-cell therapy. Retrieved February 6, 2026, from https://www.lls.org/treatment/types-treatment/immunotherapy/chimeric-antigen-receptor-car-t-cell-therapy Liede, A., Cai, M., Crouter, T. F., Niepel, D., Callaghan, F., & Evans, D. G. (2018). Risk-reducing mastectomy rates in the US: A closer examination of the Angelina Jolie effect. Breast Cancer Research and Treatment, 171(2), 435–442. https://doi.org/10.1007/s10549-018-4824-9 Liu, G. H., Chen, T., Zhang, X., Ma, X. L., & Shi, H. S. (2022). Small molecule inhibitors targeting the cancers. MedComm, 3(4), e181. https://doi.org/10.1002/mco2.181 Lustberg, M. B. (2026). Prevention and treatment of chemotherapy-induced peripheral neuropathy. UpToDate. Retrieved February 6, 2026, from https://www.uptodate.com/contents/prevention-and-treatment-of-chemotherapy-induced-peripheral-neuropathy Manis, J. P. (2025). Overview of therapeutic monoclonal antibodies. UpToDate. Retrieved February 10, 2026, from https://www.uptodate.com/contents/overview-of-therapeutic-monoclonal-antibodies Miliotou, A. N., & Papadopoulou, L. C. (2018). CAR T-cell therapy: A new era in cancer immunotherapy. Current Pharmceutical Biotechnology, 19(1), 5–18. https://doi.org/10.2174/1389201019666180418095526 National Cancer Institute. (n.d.). PARP inhibitor. Retrieved February 6, 2026, from https://www.cancer.gov/publications/dictionaries/cancer-terms/def/parp-inhibitor National Cancer Institute. (2022). Immune checkpoint inhibitors. https://www.cancer.gov/about-cancer/treatment/types/immunotherapy/checkpoint-inhibitors National Cancer Institute. (2025a). Breast cancer prevention (PDQ)—Health professional version. https://www.cancer.gov/types/breast/hp/breast-prevention-pdq National Cancer Institute. (2025b). Cancer prevention overview (PDQ)—Health professional version. https://www.cancer.gov/about-cancer/causes-prevention/hp-prevention-overview-pdq National Cancer Institute. (2025c). Skin cancer prevention (PDQ)—Health professional version. https://www.cancer.gov/types/skin/hp/skin-prevention-pdq National Comprehensive Cancer Network. (n.d.-a). About. Retrieved February 6, 2026, from https://www.nccn.org/home/about National Comprehensive Cancer Network. (n.d.-b). NCCN guidelines: Treatment by cancer type. Retrieved February 6, 2026, from https://www.nccn.org/guidelines/category_1 National Human Genome Research Institute. (2025). The Human Genome Project. National Institutes of Health. https://www.genome.gov/human-genome-project Nettina, S. M., and Nelson-Tuttle, C. (2024). Lippincott manual of nursing practice (12th ed.). Wolters Kluwer. Olsen, M., LeFebvre K., & Brassil, K. (2019). Chemotherapy and immunotherapy guidelines and recommendations for practice (1st ed.). Oncology Nursing Society. Oncology Nursing Society. (n.d.). ONS/ONCC chemotherapy immunotherapy certificate course. Retrieved February 2026, from https://www.ons.org/courses/onsoncc-chemotherapy-immunotherapy-certificate-renewaltm Oncology Nursing Society. (2018). Toolkit for safe handling of hazardous drugs for nurses in oncology. https://www.ons.org/taxonomy/term/70551/06-2018/ons-develops-hazardous-drugs-safe-handling-toolkit-oncology-nurses Oncology Nursing Society. (2025). Chemotherapy-induced nausea and vomiting—Adult. Retrieved February 2026, from https://www.ons.org/pep/chemotherapy-induced-nausea-and-vomiting-adult Pettinato, M. C. (2021). Introduction to antibody-drug conjugates. Antibodies, 10(4), 42. https://doi.org/10.3390/antib10040042 Postow, M., & Johnson, D. B. (2025). Overview of toxicities associated with immune checkpoint inhibitors. UpToDate. Retrieved February 4, 2026, from https://www.uptodate.com/contents/overview-of-toxicities-associated-with-immune-checkpoint-inhibitors Rogers, J. L., & Brashers, V. L. (Eds.). (2023). McCance and Huether's pathophysiology: The biologic basis for disease in adults and children (9th ed.). Elsevier. Rugo, H. S., & van den Hurk, C. (2024). Alopecia related to systemic cancer therapy. UpToDate. Retrieved February 6, 2026, from https://www.uptodate.com/contents/alopecia-related-to-systemic-cancer-therapy Sasikumar, P. G., & Ramachandra, M. (2018). Small-molecule immune checkpoint inhibitors targeting PD-1/PD-L1 and other emerging checkpoint pathways. BioDrugs, 35(5), 481–497. https://doi.org/10.1007/s40259-018-0303-4 Sengupta, S. (2017). Cancer nanomedicine: Lessons for immune-oncology. Trends Cancer, 3(8), 551–560. https://doi.org/10.1016/j.trecan.2017.06.006 Shastry, M., Gupta, A., Chandarlapaty, S., Young, M., Powles, T., & Hamilton, E. (2023). Rise of antibody-drug conjugates: The present and future. American Society of Clinical Oncology Educational Book, 43, e390094. https://doi.org/10.1200/EDBK_390094 Shoushtari, A. N., & Johnson, D. B. (2024). Principles of cancer immunotherapy. UpToDate. Retrieved February 4, 2026, from https://www.uptodate.com/contents/principles-of-cancer-immunotherapy Shuel, S. L. (2022). Targeted cancer therapies: Clinical pearls for primary care. Canadian Family Physician, 68(7), 515–518. https://doi.org/10.46747/cfp.6807515 Siegel, R. D., LeFebvre, K. B., Temin, S., Evers, A., Barbarotta, L., Bowman, R. M., Chan, A., Dougherty, D. W., Ganio, M., Hunter, B., Klein, M., Miller, T. P., Mulvey, T. M., Ouzts, A., Polovich, M., Salazar-Abshire, M., Stenstrup, E. Z., Sydenstricker, C. M., Tsai, S., & Olsen, M. M. (2024). Antineoplastic therapy administration safety standards for adult and pediatric oncology: ASCO-ONS standards. JCO Oncology Practice, 20(10), 1314–1330. https://doi.org/10.1200/OP.24.00216 UpToDate Lexidrug. (n.d.-a). Ipilimumab: Drug information. UpToDate. Retrieved February 4, 2026, from https://www.uptodate.com/contents/ipilimumab-drug-information UpToDate Lexidrug. (n.d.-b). Obecabtagene autoleucel: Drug information. UpToDate. Retrieved February 28, 2026, from https://www.uptodate.com/contents/obecabtagene-autoleucel-drug-information UpToDate Lexidrug. (n.d.-c). Pembrolizumab: Drug information. UpToDate. Retrieved February 4, 2026, from https://www.uptodate.com/contents/pembrolizumab-drug-information UpToDate Lexidrug. (n.d.-d). Tremelimumab: Drug information. UpToDate. Retrieved February 4, 2026, from https://www.uptodate.com/contents/tremelimumab-drug-information US Preventive Services Task Force. (2021). Lung cancer: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening Viera, A. J. (2025). Overview of preventive care in adults. UpToDate. Retrieved February 10, 2026, from https://www.uptodate.com/contents/overview-of-preventive-care-in-adults Wagle, N. S., Nogueira, L., Devasia, T. P., Mariotto, A. B., Yabroff, K. R., Islami, F., Jemal, A., Alteri, R., Ganz, P. A., & Siegel, R. L. (2025). Cancer treatment and survivorship statistics, 2025. CA: A Cancer Journal for Clinicians, 75(4), 308–340. https://doi.org/10.3322/caac.70011 Weingart, S. N., Zhang, L., Sweeney, M., & Hassett, M. (2018). Chemotherapy medication errors. Lancet Oncology, 19(4), E191–E199. https://doi.org/10.1016/S1470-2045(18)30094-9 Xu, J. Q., Murphy, S. L., Kochanek, K. D., & Arias, E. (2026). Mortality in the United States, 2024. NCHS Data Brief, 548, 1−14. https://doi.org/10.15620/cdc/174641 Yarbro, C. H., Wujcik, D., & Gobel, B. H. (2018). Cancer nursing: Principles and practice (8th ed.). Jones & Bartlett Learning. Powered by Froala Editor
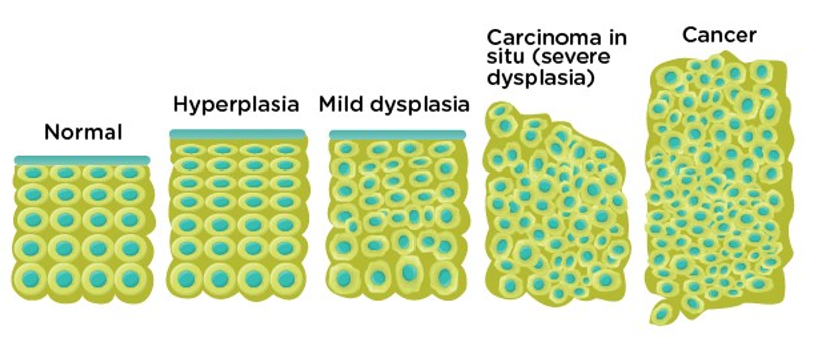
CTLA-4 Inhibitors
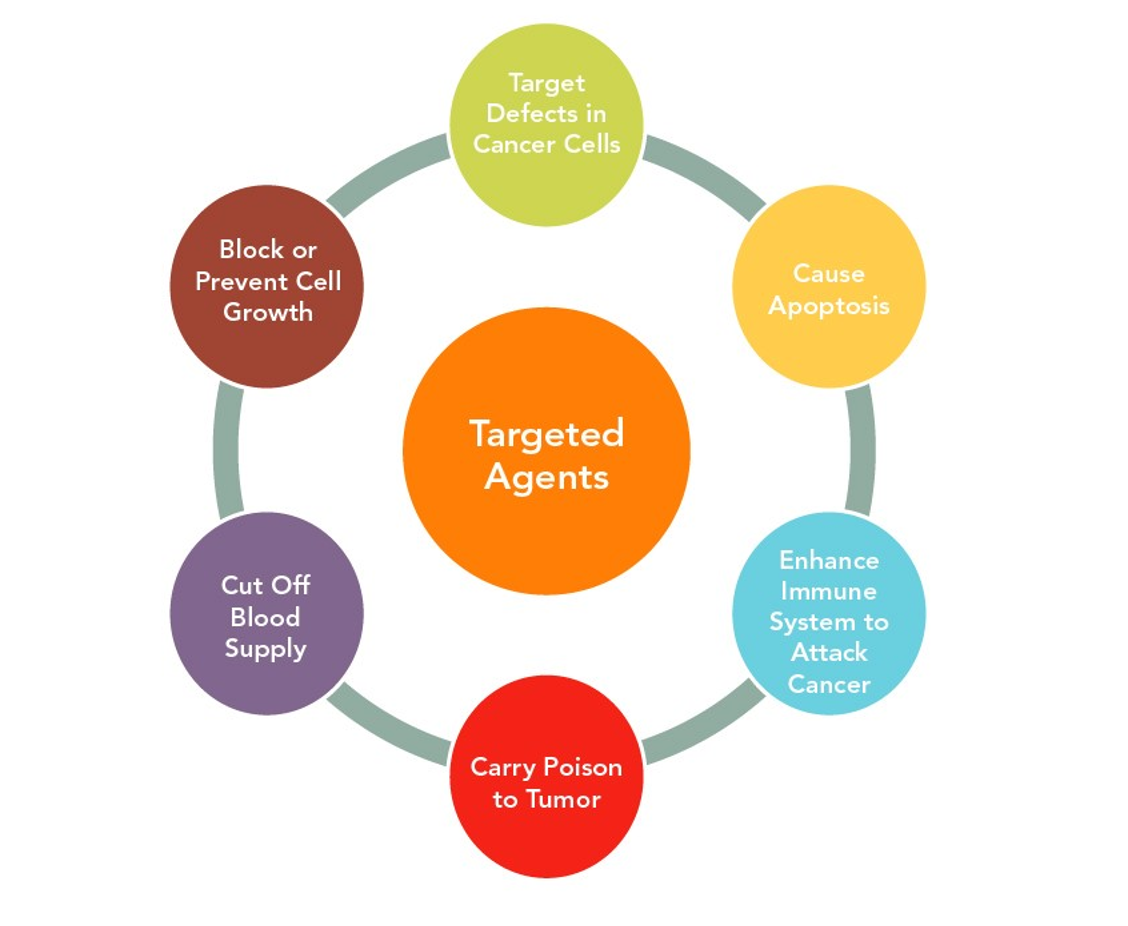
EGFR
Single Course Cost: $22.00
Add to CartSave while Futhering your Education
Oncology Medication Administration Nursing CE course for RNs and LPNs is offered in the packages below.- 71 ANCC Hours
- 24 Courses
- 522 ANCC Hours
- 189 Courses