About this course:
This course explores cardiovascular disease (CVD) pathophysiology in patients with type 2 diabetes mellitus (T2DM). In addition, it reviews the national guidelines for optimum cardiovascular (CV) health in diabetes and describes the American Diabetes Association's (ADA) evidence-based prescribing recommendations.
Course preview
Cardiovascular Health and Prescribing for Patients with Type 2 Diabetes
This course explores cardiovascular disease (CVD) pathophysiology in patients with type 2 diabetes mellitus (T2DM). In addition, it reviews the national guidelines for optimum cardiovascular (CV) health in diabetes and describes the American Diabetes Association's (ADA) evidence-based prescribing recommendations.
Upon completion of this module, learners will be able to:
- describe the pathophysiology of cardiovascular disease in type 2 diabetes mellitus
- discuss national guideline recommendations for optimal cardiovascular health in diabetes mellitus
- review evidence-based recommendations for diabetic patients with cardiovascular disease, including an overview of recommended medications, dosing, and side effects
Cardiovascular disease (CVD) is the leading cause of death among men and women of most racial and ethnic groups in the US, with approximately 697,000 deaths each year or one death every 34 seconds (The Centers for Disease Control and Prevention [CDC], 2022a). CVD costs $229 billion annually due to healthcare services, medications, and lost productivity. More specifically, 20.1 million adults age 20 and older have coronary artery disease (CAD). In addition, 805,000 people in the US experience a myocardial infarction (MI) annually. According to the National Diabetes Statistics Report, 37.3 million (11.3%) people in the US have diabetes; of those, 28.7 million have been diagnosed, and 8.5 million (23%) remain undiagnosed (CDC, 2022b). In addition, approximately 96 million (38%) people 18 years and older have prediabetes. Patients with type 2 diabetes mellitus (T2DM) are disproportionately affected by CVD compared to those without T2DM, as CVD is the leading cause of morbidity and mortality in these patients. According to the American Heart Association (AHA, 2021), adults with T2DM are 2 to 4 times as likely to die from CVD than those without diabetes. Diabetes is considered one of the seven primary modifiable risk factors for CVD. Nearly 70% of patients with T2DM who are 65 years or older die from some form of CVD. Even with the therapeutic effects of antihypertensive and lipid-lowering medications, most patients with T2DM will die from CV events (Goyal & Jialal, 2022; Rodriguez et al., 2017). Einarson and colleagues (2018) conducted a systematic review of scientific evidence regarding the prevalence of CVD in T2DM across ten years (2007 to 2017). The researchers concluded that worldwide, CVD affects more than 30% percent of individuals diagnosed with T2DM, accounts for nearly 50% of all deaths in patients with T2DM, and CAD, stroke, and MI are cited as the chief offenders (Einarson et al., 2018). Advanced practice registered nurses (APRNs) must understand the basis for the underlying pathophysiological processes, strategies for and challenges associated with managing CVD risk in T2DM, and the evidence-based prescribing guidelines for lowering cardiovascular (CV) risk to provide optimal care to this high-risk patient population (Nestro, 2023).
Pathophysiology of Cardiovascular Disease in Type 2 Diabetes
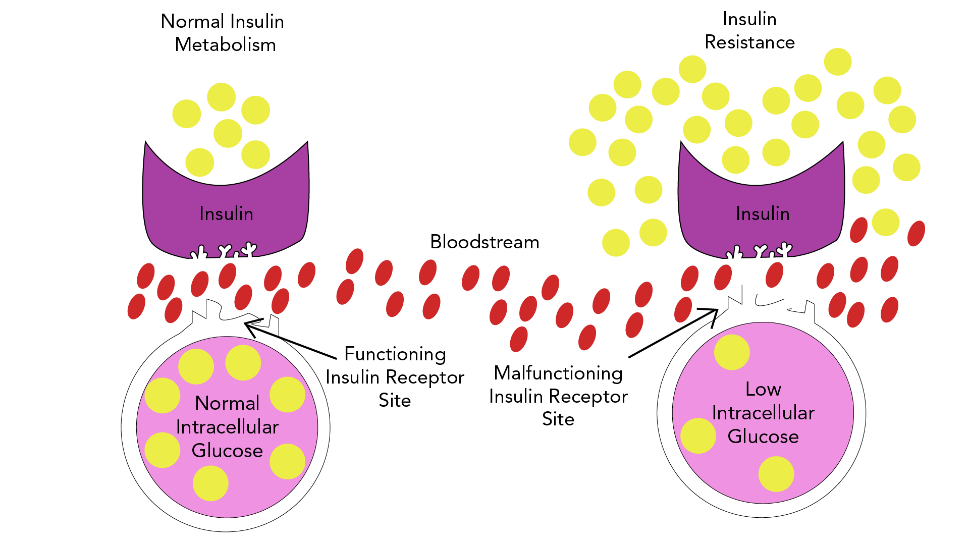
T2DM is a complex, chronic metabolic condition that impacts how the body metabolizes glucose. The condition is characterized by the body's inability to maintain balanced glucose due to an inadequate supply of insulin or insulin resistance mechanisms, where the body cannot effectively utilize the insulin it produces (Brutsaert, 2022; Hudspeth, 2018). This process is demonstrated in Figure 1.
Figure 1
Insulin Resistance Model
CVD, or heart disease, refers to a cluster of conditions affecting the heart and blood vessels, most commonly induced by atherosclerosis, which is the buildup of atheroma (or fatty plaque) within the arteries. As shown in Figure 2, as atherosclerosis progresses, plaque growth within the arteries accumulates, causing damage, narrowing, or blockage of the arteries, which can result in serious consequences (Brutsaert, 2022; Hudspeth, 2018).
Figure 2
Coronary Artery Disease
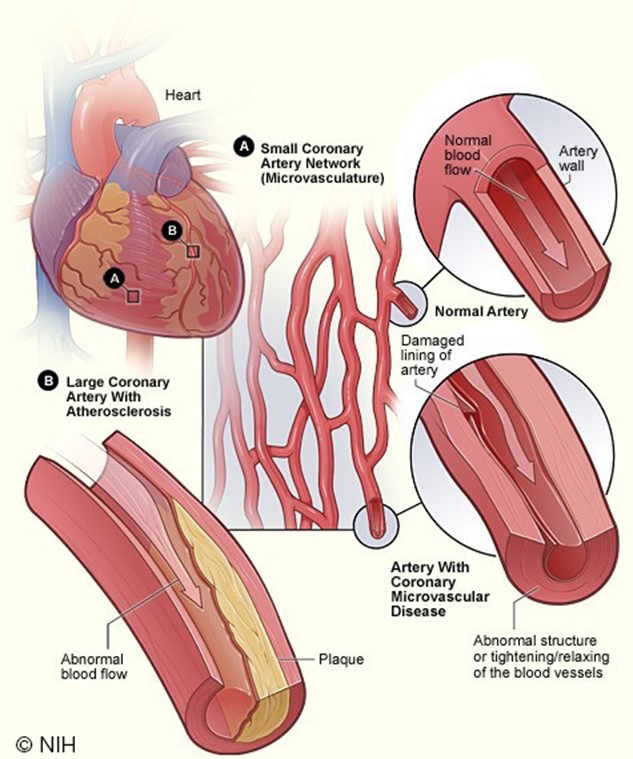
(National Heart, Lung and Blood Institute [NHLBI], 2022)
Atherosclerotic cardiovascular disease (ASCVD) is the largest contributor to T2DM-associated mortality. The risk of developing ASCVD increases in those with specified CV risk factors, as listed in Table 1. Some of these risk factors (i.e., hyperglycemia, insulin resistance) place patients with T2DM at increased risk for cardiac events (Hudspeth, 2018).
Table 1
Modifiable CV Risk Factors
Hypertension |
Obesity/overweight |
Hyperlipidemia (high blood cholesterol) or dyslipidemia (blood cholesterol levels are abnormally high or low, such as low levels of cardioprotective HDL cholesterol) |
Cigarette smoking (tobacco use) |
Sedentary lifestyle (physical inactivity; lack of exercise or routine) |
Hyperglycemia and insulin resistance |
Poor nutrition (high fat/high sodium diet) |
(CDC, 2023; Lopez et al., 2022; Nesto, 2023)
Atherosclerotic lesions are formed through complex interactions of various factors, and T2DM accelerates all of these interactions (Katakami, 2018). CV damage occurs over time and is the byproduct of metabolic changes within the large blood vessels (macrovascular) and small blood vessels (microvascular) of tissues and organs. These changes begin to occur during the prediabetes stage, which elucidates why more than half of patients already have evidence of CVD at the time of T2DM diagnosis (Ignatavicius et al., 2021). As defined in Table 2, the International Diabetes Federation (IDF) cites the three most common forms of CVD as direct consequences of macrovascular tissue damage: CAD, stroke, and peripheral arterial disease (PAD). The American Diabetes Association (ADA) Professional Practice Committee released the 2022a clinical practice recommendations regarding CVD and risk management, defining ASCVD as "coronary heart disease (CHD), cerebrovascular disease, or PAD presumed to be of atherosclerotic origin" (ADA Professional Practice Committee, 2022a, p. S144). Further, the ADA cites heart failure (HF) as another significant cause of morbidity and mortality from CVD, with a twofold higher risk in T2DM patients, as hypertension often serves as the precursor to this condition. Patients with T2DM are at increased risk for all of these conditions, the onset is typically at earlier ages than those without T2DM, and they affect women more commonly than men (ADA Professional Practice Committee, 2022a).
Table 2
Three Major Types of CVD and Associated Conditions
CAD | Stroke | ...purchase below to continue the course | |
Description | Affects the blood vessels supplying blood to the heart | Affects the blood vessels supplying blood to the brain | Affects the blood vessels supplying blood to the legs and feet |
Conditions |
|
|
|
(IDF, 2016)
The pathogenesis of CVD in T2DM is a complex process mediated by several underlying cellular, molecular, and genetic processes. Cell-signaling defects in metabolic and inflammatory pathways affect the endothelium, liver, skeletal muscle, and beta cells of the pancreas. These defects are likely to have genetic components but are also largely influenced by environmental factors such as obesity, sedentary lifestyles, tobacco use, and certain medications. Patients with T2DM often have traditional risk factors for CVD, such as obesity, hypertension, dyslipidemia, and sedentary lifestyles, which creates a clear pathway for ASCVD development and its consequences (Brutsaert, 2022). The following three theories explain the mechanism of vascular complications in patients with T2DM:
- chronic hyperglycemia leads to permanent basement membrane thickening, tissue damage, and organ destruction and is a chief cause of premature development of macrovascular complications
- glucose toxicity directly or indirectly impairs the functional cell integrity
- chronic ischemia in small blood vessels causes tissue hypoxia (Brutsaert, 2022; Ignatavicius et al., 2021)
Endothelial cells are essential to maintaining homeostasis within the vasculature. These cells are tasked with generating and releasing various biochemical substances within the body to control and maintain the function and integrity of the vessels (Brutsaert, 2022; Katakami, 2018). Through the release of dilator and constrictor substances, endothelial cells serve critical roles in sustaining the balance between a series of mechanisms:
- oxidation and antioxidation
- inflammation and anti-inflammation within the vascular walls
- proliferation and antiproliferation of vascular smooth muscle cells
- dilatation and contraction of vessels
- coagulation and fibrinolysis of blood (Katakami, 2018)
The principal complication in T2DM is endothelial dysfunction, an independent risk marker for atherosclerosis and CV events. Various mechanisms can disrupt normal functioning and provoke adverse metabolic events within the endothelial cell of patients with T2DM, such as hyperglycemia, excess free fatty acid release, insulin resistance, increased levels of low-density lipoprotein (LDL) cholesterol, oxidative stress, and tobacco use. Endothelial dysfunction promotes leukocyte and platelet adhesion, thrombosis, and inflammation. Activation of these systems impairs normal functioning, increases vasoconstriction, and promotes inflammation and thrombosis, thereby rendering arteries susceptible to atherosclerosis (Brutsaert, 2022; Katakami, 2018).
Chronic hyperglycemia and insulin resistance serve essential roles in the initiation of vascular complications of DM through several mechanisms, such as: (1) increased formation of advanced glycation end products (AGEs) and activation of the receptor for advanced glycation end products (RAGE) AGE-RAGE axis, (2) oxidative stress, and (3) inflammation (Fishman et al., 2018). Insulin serves an essential dual-action role in maintaining homeostasis of the vasculature, as it stimulates endothelial cell production of nitric oxide (NO), a vasodilator that exerts antiaggregatory effects on smooth muscle. Insulin also mediates the release of endothelin (ET-1), a potent vasoconstrictor. Under normal physiological conditions, insulin activity is mediated by a vasoprotective signaling pathway called phosphoinositide 3-kinase (PI3K)/Akt. However, when insulin resistance develops, insulin responds to a pathological alternative, the mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway. The MAPK/ERK pathway causes inflammation, vasoconstriction, and vascular smooth muscle cell proliferation, thereby contributing to the consequential CV byproducts of insulin resistance (Janus et al., 2016). Patients with insulin resistance have higher rates of hyperglycemia, which accelerates plaque formation and accumulation. Acute hyperglycemia does not reduce the bioavailability and endothelial-dependent vasodilation, whereas chronic hyperglycemia promotes atherogenesis and accelerates the progression of atherosclerosis (Katakami, 2018). The relationship between hyperglycemia, increased insulin secretion, and the resulting consequences are multifaceted. Several factors prompt the beta cells within the pancreas to secrete insulin, such as increased plasma glucose or amino acid levels, increased glucose-dependent insulinotropic polypeptide (GIP) secretion, decreased epinephrine secretion, decreased sympathetic activity, or increased parasympathetic activity. Once the beta cells secrete insulin, review Table 3 for the various effects of insulin on the body's tissues
Table 3
Insulin Actions
Tissue Type | Effect of Insulin on Tissue |
Most tissues |
|
Adipose tissue |
|
Muscle |
|
Liver tissue |
|
(Walsh & Sved, 2019)
As displayed in Table 3, T2DM and dyslipidemia commonly occur together. Lipid abnormalities affect up to 70% of patients with T2DM. Atherosclerosis is also prompted by local inflammation in the vascular wall induced by hyperlipidemia, specifically high levels of LDL. Patients with T2DM have a higher prevalence of lipid abnormalities in the peripheral venous circulation, increased atherosclerotic plaque accumulation, and smaller coronary artery lumen diameter than those without T2DM. Hyperlipidemia and atherosclerotic plaques result in the recruitment and migration of monocytes and other immune and inflammatory cells into the vascular subendothelial layer. Recruited monocytes differentiate into macrophages or dendritic cells. Activated macrophages express scavenger receptors to facilitate the engulfment of native and oxidized LDL cholesterol and, along with other inflammatory cells, increase the production of chemokines and cytokines. These mechanisms promote atherosclerotic lesion progression within the inflammatory cycle (ADA Professional Practice Committee, 2022a; Janus et al., 2016; Low Wang et al., 2016).
National Guidelines for Optimal Cardiovascular Health in Type 2 Diabetes
Research has demonstrated that preventing or slowing the progression of CVD in patients with T2DM is based on controlling individual CV risk factors. Studies have consistently demonstrated that lowering the glycated hemoglobin, or hemoglobin A1C (HbA1C) level, in patients with T2DM minimally reduces CV risks when performed in isolation. Simultaneously correcting and controlling multiple CV risk factors markedly reduces CVD mortality in patients with T2DM (Nestro, 2023). Over the last decade, research has demonstrated a decline in CVD-associated morbidity and mortality when aggressive risk factor modifications are concurrently implemented. The ADA Professional Practice Committee's Standards of Care in Diabetes (2022a), a national resource for the optimal management of diabetes, includes annual updates on the evidence-based prevention and management of diabetes and diabetes-related complications. Embedded within these standards are specific guidelines on CVD and risk management in patients with T2DM, strongly emphasizing the concurrent control of hyperglycemia, hypertension, and hyperlipidemia as the central targets (ADA Professional Practice Committee, 2022a).
Evidence-Based Prescribing Recommendations
Unless specified otherwise, the prescribing recommendations in this section are adapted from the ADA Professional Practice Committee (2022d), which utilizes the following ABCE evidence-grading system to demonstrate the level of evidence of each recommendation:
- A - clear evidence from well-conducted and generalizable randomized controlled trials that are adequately powered
- B – supportive evidence from well-conducted cohort studies
- C - supportive evidence from poorly controlled or uncontrolled studies
- E - expert consensus or clinical experience (ADA Professional Practice Committee, 2022d)
CVD Risk Assessment
To improve patient outcomes and effectively manage CV risk in the context of T2DM, the American College of Cardiology (ACC, n.d.-c) and ADA Professional Practice Committee (2022a) both cite the following as essential components:
- Expand the narrow focus of glucose control to include a systematic assessment of all CV risk factors (Table 1) at least annually and perform aggressive risk reduction, emphasizing closely monitoring and controlling the ABCs of CV risk (Table 4).
- Encourage and implement an individualized, patient-centered, and collaborative approach to reducing CV risk through shared-decision making and open communication between clinicians and patients.
- Employ the ACC/AHA ASCVD risk calculator to stratify better the ASCVD risk and help guide therapy. This risk calculator, the Risk Estimator Plus, is an online tool that estimates the 10-year risk of a first ASCVD event, accounting for a diagnosis of T2DM as a risk factor.
- Routine screening for CAD is not recommended for asymptomatic patients since it does not improve as long as ASCVD risk factors are treated.
- CAD investigation should be considered for the following atypical cardiac symptoms (i.e., chest discomfort, unexplained dyspnea), associated vascular disease (i.e., transient ischemic attack [TIA], stroke, PAD, carotid bruits, claudication), or electrocardiogram [ECG] abnormalities.
Table 4
The ABCs of CVD Risk in Diabetics and Targets
A: A1C
|
B: Blood Pressure (BP)
|
C: Cholesterol
|
(ADA Professional Practice Committee, 2022a, 2022c)
In addition to the above, the following CV risks should also be assessed at least annually (ADA Professional Practice Committee, 2022a):
- Smoking: affects microcirculation and accelerates CV complications; smoking cessation is strongly advised.
- Family history of premature CAD: is a non-modifiable risk factor for T2DM and CVD.
- Chronic kidney disease (CKD): BP control should be maintained to reduce the risk of kidney disease; CKD is a common complication of uncontrolled hypertension and diabetes.
- Presence of albuminuria (protein in the urine): is a biomarker for CVD and coronary events.
Treatment/Management
The ACC cites six steps for optimizing CV risk reduction among patients with T2DM when developing each patient's individualized treatment plan (ACC, n.d.-a; Das et al., 2020):
- Educate patients with T2DM about CV risks (beyond poor glucose control) that might contribute to and accelerate CV damage (Table 1).
- Empower patients to take action by setting personal goals for lowering CV risk.
- Create an individualized plan to assess and manage CV risk on an ongoing basis and revisit this plan to reevaluate, update, and make necessary changes to promote compliance.
- Ensure adequate glycemic control is obtained by developing a realistic plan for lifestyle changes and adhering to prescribed glucose-lowering medications.
- Consider prescribing novel antihyperglycemic agents when appropriate (discussed in the next section).
- Assess adherence and identify hurdles, such as cost of treatment, side effects, personal preferences, or treatment complexity.
Lifestyle Management
Lifestyle management for T2DM and CVD risk reduction should begin at the initial contact with the patient and continue throughout all subsequent evaluations, including during the assessment for complications and management of comorbid conditions. Lifestyle strategies should be implemented preventatively, across the spectrum of care, and as part of the treatment plan in collaboration with any medications prescribed. The combination of lifestyle and pharmacologic interventions enhances treatment efficacy, aids in controlling CV risk factors, and more successfully reduces morbidity and mortality. Clinicians and patients should engage in shared decision-making to determine appropriate goals and targets across the spectrum of their diabetes care. Heart-healthy lifestyle interventions are advised for all patients with T2DM, focusing on weight reduction to reduce CV risk. The ADA Professional Practice Committee (2022a, 2022b, 2022e) recommends the following monitoring and lifestyle recommendations:
- Glucose monitoring is key to achieving glycemic targets for many patients with T2DM. Self-monitoring of blood glucose (SMBG) may help with self-management and medication adjustment, and diabetes self-management education (DSME) should be patient-centered and help guide clinical decisions; (A).
- Reduce excess body weight through caloric restriction, physical activity, and behavioral therapy (losing ≥5% of body weight can benefit glycemic control, lipids, and blood pressure). Additional weight loss often improves T2DM and CV risk; (B).
- Follow the DASH eating pattern to reduce sodium intake (less than 2,300 mg/day); (A).
- High-frequency counseling (i.e., ≥16 sessions in 6 months) should focus on dietary changes, behavioral strategies, and physical activity to achieve a 500 to 700 kcal/day energy deficit; (A).
- Increase dietary consumption of fruits and vegetables (eight to ten servings/day).
- Moderate alcohol intake (no more than two servings/day for men and no more than one serving/day for women); (A).
- Increase physical activity (150 minutes or more of moderate-to-vigorous intensive aerobic activity each week); (A).
Pharmacologic Therapy
While lifestyle interventions and modifications are essential, gaining control over T2DM and reducing CV risk usually requires adjunctive pharmacologic therapy. CV risk reduction in T2DM is primarily premised on four categories: (1) antiplatelet therapy, (2) antihypertensive medications, (3) lipid-lowering agents, and (4) antihyperglycemic drugs (ADA Professional Practice Committee, 2022a, 2022g).
Antiplatelet Therapy
Antiplatelet therapy may be used to prevent blood clots, thereby reducing the risk of stroke or MI. The most common and well-studied form of antiplatelet therapy is low-dose acetylsalicylic acid (ASA) or aspirin. For patients with documented acetylsalicylic acid (ASA) allergy, P2Y12 inhibitors, such as clopidogrel (Plavix), are recommended alternatives (ADA Professional Practice Committee, 2022a).
Aspirin. Acetylsalicylic acid (ASA) has effectively reduced CV morbidity and mortality in high-risk patients who have endured a prior MI or stroke and is strongly recommended for secondary prevention. The benefits are less clear in primary prevention, and its use is more controversial among patients with no previous CV events. The risks associated with acetylsalicylic acid (ASA) therapy or second-line antiplatelet treatments, such as P2Y12 inhibitors, must be considered and balanced against the benefits. Acetylsalicylic acid (ASA) is an over-the-counter (OTC) nonsteroidal anti-inflammatory drug (NSAID) that is widely used to treat several conditions, such as fever, pain, and inflammation. Acetylsalicylic acid (ASA) reduces CV risk by blocking the enzyme that makes prostaglandins (cyclooxygenase), thereby reducing concentrations of prostaglandins and lowering pain levels, inflammation, and body temperature. Since acetylsalicylic acid (ASA) is a potent inhibitor of prostaglandin synthesis and platelet aggregation, it inhibits platelets for the entire cell lifespan of 7 to 10 days. Therefore, it decelerates the blood's clotting action by reducing the clumping of platelets. Acetylsalicylic acid (ASA) inhibits the function of platelets in a manner different from other NSAIDs, such as ibuprofen (Motrin), as its antithrombotic effects last longer, making it the ideal agent for MI and stroke reduction. There is a risk for bleeding events in patients taking acetylsalicylic acid (ASA), particularly gastrointestinal (GI) bleeding. The drug is listed within the Beers Criteria and should be used cautiously (or avoided) in older adults due to its anticoagulation effects. The risk for GI bleeding is heightened in individuals aged 60 or older with a history of stomach ulcers, bleeding disorders, and those taking other types of anticoagulants (blood thinners). Further, those who consume three or more alcoholic beverages daily are at heightened risk for bleeding events. Aside from GI bleeding, the most common side effect is tinnitus (ringing in the ears). Enteric-coated formulations of acetylsalicylic acid (ASA) are considered safer regarding the risk of GI bleeding. They are designed to pass through the stomach and not disintegrate until it reaches the small intestine. Acetylsalicylic acid (ASA) may cause a severe allergic reaction, causing hives, facial swelling, shock, or asthma (wheezing). Children, adolescents, and young adults under 21 who have or are recovering from chickenpox or flu-like syndromes should not take aspirin due to the risk of a rare but serious illness known as Reye's syndrome (ADA Professional Practice Committee, 2022a; American Geriatrics Society Beers Criteria Update Expert Panel, 2019; Woods, 2023).
P2Y12 Inhibitors. P2Y12 inhibitors are a group of antiplatelet drugs that may be used instead of acetylsalicylic acid (ASA) for patients with an allergy or other contraindication. Clopidogrel (Plavix) is the most widely used and studied P2Y12 inhibitor for reducing CV risk. Still, other medications in this class may also be considered, such as ticlopidine (Ticlid), ticagrelor (Brilinta), and prasugrel (Effient). Clopidogrel (Plavix) binds to the P2Y12 receptor on platelets, preventing adenosine diphosphate (ADP) from activating platelets. The tolerability and side effects of clopidogrel (Plavix) are similar to that of acetylsalicylic acid (ASA), as it also poses a risk for bleeding events, particularly GI bleeding and ulcers. Ticlopidine (Ticlid) carries an added risk of neutropenia (a decline in the white blood cell count), which heightens the risk of acquiring an infection, and thrombotic thrombocytopenic purpura (TTP), an immune disorder that destroys platelets and occurs in about 1 out of every 250,000 people. Ticagrelor (Brilinta) may worsen kidney function and induce shortness of breath and therefore is not advised in patients with T2DM who have underlying renal dysfunction (ADA Professional Practice Committee, 2022a; Hennekens, 2021; Woods, 2023). The ADA endorses the following antiplatelet prescribing recommendations (ADA Professional Practice Committee, 2022a):
- Acetylsalicylic acid (ASA) is not recommended in patients under 50 years of age with T2DM who are at low risk of ASCVD (have no major ASCVD risk factors), as the benefit is low and the risk of bleeding is higher (A).
- Acetylsalicylic acid (ASA) 75 to 162 mg/day may be considered a primary prevention strategy in patients with T2DM who are at increased CV risk, following a comprehensive discussion with the patient on the potential benefits compared with the increased risk of bleeding (A).
- Use acetylsalicylic acid (ASA) 75 to 162 mg/day as a secondary prevention strategy in patients with T2DM who have a history of ASCVD (A).
- In patients with ASCVD and acetylsalicylic acid (ASA) allergy, clopidogrel (Plavix), 75 mg/day, should be used (B).
- For a year following an acute cardiac event, dual antiplatelet therapy with low-dose aspirin and a P2Y12 inhibitor is reasonable (A). It may have benefits extending beyond this one year (B).
- Long-term dual antiplatelet treatment should be considered for patients with prior coronary intervention, high ischemic risk, and low bleeding risk to prevent cardiovascular events (A).
- For patients with stable coronary and/or PAD with a low risk for bleeding, a combination of acetylsalicylic acid (ASA) and rivaroxaban (Xarelto) should be considered to prevent limb and cardiovascular events (A).
Antihypertensive Medications
Treatment of hypertension reduces CV events and microvascular complications in patients with T2DM. Several studies have demonstrated that antihypertensive therapy reduces ASCVD events, HF, and microvascular complications in patients with T2DM. Treatment should focus on controlling BP to achieve individualized targets utilizing drug classes demonstrated to reduce CV events, specifically in patients with T2DM. These agents include angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), thiazide-like diuretics, or dihydropyridine calcium channel blockers (CCBs), as outlined in Table 5. ACEIs and ARBs are considered first-line antihypertensive agents in patients with T2DM, as they lower blood pressure and reduce the risk of kidney dysfunction from microvascular complications. The ADA cautions that BP targets more aggressive than < 140/90 mmHg are not likely to improve CV outcomes among most patients with T2DM and are more likely to contribute to adverse effects and costs. Therefore, a balance between the potential benefits and risks must be considered before prescribing more intensive antihypertensive therapy (ADA Professional Practice Committee, 2022a).
Table 5
Antihypertensive Medication Therapy in Type 2 Diabetes
Drug Class and Medications |
Mechanism of Action |
Side Effects, Warnings, and Precautions |
ACEI
|
|
|
ARB
|
|
|
Thiazide-like Diuretic
|
|
|
CCBs
|
|
|
*Preferred agents in patients with T2DM
(ADA Professional Practice Committee, 2022a; Bloch & Basile, 2022; Heidenreich et al., 2022; Scheen, 2018)
The ADA Professional Practice Committee (2022a) endorses the following antihypertensive prescribing recommendations, which are intended to be used in conjunction with continued lifestyle interventions:
- In patients with known ASCVD with T2DM, consider prescribing an ACEI or an ARB to reduce the risk of CV events (A).
- In patients with BP of at least 140/90 mmHg (confirmed in an office), prompt initiation and timely titration of one pharmacologic agent is advised to achieve blood pressure goals (A).
- In patients with BP above 160/100 mmHg (confirmed in an office), prompt initiation and timely titration of two drugs or a single-pill combination of drugs demonstrated to reduce CV events in patients with diabetes is advised to achieve blood pressure goals (A).
- Multi-drug therapy is usually necessary to achieve BP targets, but combinations of ACEI and ARBs or combinations of ACEI or ARBs with direct renin inhibitors should not be used (A).
- An ACEI or ARB, at the maximum tolerated dose indicated for BP treatment, is the recommended first-line treatment for hypertension in patients with T2DM and urinary albumin-to-creatinine ratio [UACR] ≥300 mg/g creatinine (A), or 30 to 299 mg/g creatinine (B), and if one class is not tolerated, the other should be substituted (B).
- Patients treated with ACEI, ARB, or diuretic therapy should have serum creatinine, estimated glomerular filtration rate (eGFR), and serum potassium levels monitored at least annually (B).
The initial treatment for patients with T2DM depends upon the severity of hypertension and clinical risk factors. The following basic prescribing guidelines apply when initiating antihypertensive therapy in patients with T2DM (ADA Professional Practice Committee, 2022a; Scheen, 2018):
- For initial BP between 140/99 mmHg and 159/99 mmHg, begin single-drug therapy with an ACEI, ARB, thiazide diuretic, or CCB.
- For initial BP above ≥160/100 mmHg, begin dual-drug therapy with two antihypertensive medications, including an ACEI or ARB and a CCB or thiazide diuretic.
- Initial treatment should include an ACEI or ARB in patients with established CAD and those with albuminuria to reduce the risk of progressive kidney disease.
- ACEIs or ARBs do not provide superior cardioprotection compared to thiazide-like diuretics or CCBs in patients without albuminuria.
- Certain types of thiazide diuretics can contribute to hyperglycemia and metabolic dysfunction in patients with T2DM. The preferred thiazide agents include chlorthalidone (Thalitone) and indapamide (Lozol), as they carry a reduced risk for these metabolic effects than thiazide diuretics such as hydrochlorothiazide (HCTZ).
- Beta-blockers are indicated for patients with a prior MI, HF, or active angina. However, they have not been shown to reduce mortality in patients without these conditions.
For patients with resistant hypertension (defined as a BP ≥140/90 mmHg despite a therapeutic strategy that includes lifestyle modifications and three classes of antihypertensive medications, including a thiazide-like diuretic), adding a mineralocorticoid receptor antagonist (MRA) should be considered as an additive to existing treatment with an ACEI or ARB, thiazide-like diuretic, and CCB. However, before diagnosing resistant hypertension, clinicians should confirm the absence of any barriers that may impair adherence to the existing antihypertensive medication regimen and the potential for secondary hypertension. The ADA Professional Practice Committee (2022a) makes the following recommendation for patients with confirmed resistant hypertension:
- MRAs should be reserved for managing resistant hypertension. These agents should be added to the patient's existing treatment with an ACEI or ARB, thiazide-like diuretic, and CCB (B).
Numerous studies have demonstrated that primary aldosteronism and hyperaldosteronism are common in patients with resistant hypertension. MRAs, also referred to as aldosterone antagonists, have well-established benefits regarding their efficacy in managing resistant hypertension. MRAs effectively act on the renin-angiotensin-aldosterone pathway, providing CV and renal protection. Spironolactone (Aldactone) and eplerenone (Inspra) are the most extensively studied MRAs in managing resistant hypertension. MRAs are classified as potassium-sparing diuretics, as they prevent the body from absorbing too much salt and avert potassium excretion. These drugs bind to the androgen receptors to prevent their interaction with testosterone. Therefore, typical side effects include gynecomastia, breast pain, erectile dysfunction, and menstrual irregularities, occurring in up to 9% of patients and reversible upon drug discontinuation (Yugar-Toledo et al., 2017).
These side effects are most common with spironolactone (Aldactone), the prototype of MRAs, which has been the subject of numerous studies over time with demonstrated efficacy. Spironolactone (Aldactone) is much more potent than eplerenone (Inspra), a second-generation selective MRA. Eplerenone (Inspra) carries a higher affinity for the mineralocorticoid receptor and a lower affinity for androgen receptors than spironolactone (Aldactone). While it poses a lower risk of adverse effects, it is more expensive. MRAs can also reduce albuminuria and have additional CV risk reduction benefits. The most common adverse effect among all MRAs is hyperkalemia, so monitoring serum creatinine and potassium levels is strongly advised. Additional side effects include drowsiness, lightheadedness, blurred vision, nausea, vomiting, diarrhea, headache, increased thirst, and orthostatic hypotension. Newer data have suggested that MRAs may provide preferential benefits in treating obesity-related hypertension, particularly in individuals with high dietary sodium intake (ADA Professional Practice Committee, 2022a; Dudenbostel & Calhoun, 2017; Leopold & Ingelfinger, 2023; Yugar-Toledo et al., 2017).
Statins or Other Lipid-Lowering Therapy
The most prevalent pattern of dyslipidemia in patients with T2DM includes low levels of HDL cholesterol combined with elevated LDL and triglyceride levels. Significant evidence supports the critical importance of reducing LDL levels as one of the most effective ways to reduce ASCVD. Statins are the preferred first-line pharmacologic therapy for most patients with T2DM due to their well-established benefits in lowering LDL cholesterol and cardioprotective factors. They work by inhibiting hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase, which is the enzyme in the cholesterol biosynthesis pathway. While statins are highly effective at lowering LDL, they can be associated with toxicity. The most common adverse effect and reason for discontinuation of statin therapy are statin-associated muscle symptoms (SAMSs); up to 72% of all statin adverse events are muscle related. SAMSs can present as myalgia, myopathy, myositis with elevated creatinine kinase, or rhabdomyolysis in its most severe form. Other side effects include joint and abdominal pain, neurological and neurocognitive effects, hepatotoxicity, and renal toxicity (ADA Professional Practice Committee, 2022a; Ward et al., 2019).
There are two dosing regimens recommended by the ADA, which are listed in Table 6. High-intensity statin therapy is cited for achieving greater than 50% reduction in LDL cholesterol; moderate-intensity statin regimens typically achieve 30% to 49% reductions in LDL cholesterol. Low-dose statin therapy is generally not recommended in patients with T2DM, but at times may be the maximal dose of statin that a patient can tolerate. For patients who do not tolerate the intended intensity of statin, the maximally tolerated statin dose should be used (ADA Professional Practice Committee, 2022a).
Table 6
High-Intensity vs. Moderate-Intensity Statin Treatment
High Intensity | Moderate Intensity |
Atorvastatin (Lipitor) 40 to 80 mg/day | Atorvastatin (Lipitor) 10 to 20 mg/day |
Rosuvastatin (Crestor) 20 to 40 mg/day | Rosuvastatin (Crestor) 5 to 10 mg/day |
Simvastatin (Zocor) 20 to 40 mg/day | |
Pravastatin (Pravachol) 40 to 80 mg/day | |
Lovastatin (Altoprev) 40 mg/day | |
Fluvastatin XL (Lescol XL) 80 mg/day | |
Pitavastatin (Livalo) 1 to 4 mg/day |
(ADA Professional Practice Committee, 2022a)
While other lipid-lowering agents are available, the evidence for using drugs that target these lipid fractions is not nearly as rigorous or extensive as the literature surrounding statin therapy, particularly regarding efficacy in patients with T2DM. In patients that are not achieving individualized lipid profile targets on statin therapy or those with intolerance to an increased dosage of statin, the ADA Professional Practice Committee recommends combination therapy with ezetimibe (Zetia) or a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor such as evolocumab (Repatha) or alirocumab (Praluent; ADA Professional Practice Committee, 2022a).
Ezetimibe (Zetia). Ezetimibe (Zetia) belongs to a class of drugs called cholesterol absorption inhibitors. They work by inhibiting the absorption of dietary cholesterol in the intestines and lowering LDL levels in the blood. In a randomized controlled trial, ezetimibe (Zetia) 10 mg was added to moderate-intensity simvastatin (Zocor) 40 mg and compared with simvastatin (Zocor) 40 mg alone. Among patients with T2DM, this combination demonstrated a significant reduction in major CV events with an absolute risk reduction of 5% and a relative risk reduction of 14% over single-agent simvastatin (Zocor; Giugliano et al., 2018). Ezetimibe (Zetia) may be taken at the same time as statins. A commonly cited benefit is that it can help reduce the statin dose and, consequently, the associated risk for muscle injury and SAMSs. It is tolerable across clinical trials; the most common side effects reported include drowsiness, diarrhea, sinus congestion, and joint pain. Ezetimibe (Zetia) should be avoided in patients with moderate to severe liver dysfunction due to the rare risk of liver failure. Other rare adverse effects include allergic reactions, rhabdomyolysis, pancreatitis, and a severe skin rash characterized by red, blistering, and peeling skin (Rosenson, 2023).
PCSK9 inhibitors. PCSK9 inhibitors are approved for patients with inadequately treated levels of LDL while on other agents. PCSK9 is an enzyme that is predominantly produced in the liver. PCSK9 binds to the LDL receptor on the surface of hepatocytes (liver cells), destroying LDL receptors and increasing plasma LDL levels. PCSK9 inhibitors, such as evolocumab (Repatha) and alirocumab (Praluent), are humanized monoclonal antibodies that bind to free plasma PCSK9, promoting the destruction of this enzyme. This leaves less free PCSK9 to attach to the LDL receptors, thereby lowering LDL levels. These medications can lower LDL levels by as much as 60% in patients concurrently on statin therapy and reduce the rates of stroke and MI. They are approved for patients with ASCVD or familial hypercholesterolemia who receive maximally tolerated doses of statin therapy but require additional lowering of LDL levels. These medications can only be administered by subcutaneous injection and are unavailable in oral preparations. The most commonly reported side effects are injection site reactions, usually mild and limited to erythema, pain, and bruising. There are no reports of these medications inducing muscle breakdown or liver impairment; however, hypersensitivity reactions have been reported, including rash, pruritus, and urticaria. There is concern that PCSK9 inhibitors can potentially influence hepatitis C infectivity, but this is not yet confirmed, and more studies are needed to establish this as a definitive risk factor (ADA Professional Practice Committee, 2022a; Stroes et al., 2023). Medication dosing is as follows (Stroes et al., 2023):
- evolocumab (Repatha) 140 mg subcutaneous injection every 2 weeks, or 420 mg once monthly
- alirocumab (Praluent) 150 mg subcutaneous injection every 2 weeks initially; assess LDL levels within 4 to 12 weeks
- alternatively, patients may be given a 300 mg subcutaneous injection every 4 weeks
- 75 mg every 2 weeks is available but with a reduced LDL-lowering effect
The ADA Professional Practice Committee (2022a) endorses the following recommendations for patients with hyperlipidemia and T2DM.
- Intensify lifestyle therapy and optimize glycemic control for patients with elevated triglyceride levels (≥150 mg/dL) and/or low HDL cholesterol (<40 mg/dL for men or <50 mg/dL for women); (C).
- For adults not taking lipid-lowering medications, obtain a lipid profile at the time of T2DM diagnosis, at an initial medical evaluation, and every five years after that if under 40 or more frequently as needed (E).
- Obtain a lipid profile at the initiation of statin therapy or other lipid-lowering therapy, 4 to 12 weeks after initiation or a dose change, and annually after that to monitor the response to therapy and inform medication adherence (E).
- For patients aged 40 to 75 with T2DM without ASCVD, moderate-intensity statin therapy is recommended with lifestyle therapy (A).
- For patients aged 20 to 39 with T2DM with ASCVD risk factors, consider initiating statin therapy with lifestyle therapy (C).
- High-intensity statin therapy is recommended for patients aged 50 to 70 with T2DM, especially with multiple ASCVD risk factors (B).
- For patients with T2DM and a 10-year ASVCD risk of 20% or higher, adding ezetimibe (Zetia) to maximally tolerated statin therapy to reduce LDL levels by 50% or more is recommended (C).
- For patients of all ages with T2DM and established ASCVD disease, high-intensity statin therapy should be added to lifestyle therapy (A).
- For patients with ASCVD considered very high risk, if LDL is ≥70 mg/dL on maximum tolerated statin dose, consider adding additional LDL-lowering therapy such as ezetimibe (Zetia) or PCSK9 inhibitor (A).
- The maximum tolerated statin dose should be used for patients who do not tolerate the intended statin intensity (E).
- For patients over 75 years of age who are already taking statin therapy, it is reasonable to continue statin treatment (B).
- It may be reasonable for patients over 75 years of age to initiate statin therapy after discussing potential benefits and risks (C).
- Statin therapy is contraindicated in pregnancy (B).
- Statin plus fibrate combination therapy has not been shown to improve ASCVD outcomes and is generally not recommended (A).
- Statin plus niacin combination therapy has not been shown to provide additional CV benefit above statin therapy alone, may increase the risk of stroke with additional side effects, and is generally not recommended (A).
Novel Antihyperglycemic Medications and Cardiovascular Risk Reduction
Glucose control is the cornerstone of T2DM management regarding reducing target organ damage and limiting complications. It is well-established that metformin (Glucophage) is the first-line antihyperglycemic treatment for T2DM and also helps lower the risk of CVD as a byproduct. The ADA Professional Practice Committee states that metformin (Glucophage) should be continued for glucose-lowering as long as the eGFR remains above 30 mL/min/1.73 m2; however, it should be avoided in unstable or hospitalized patients with HF. In 2008, the US Food & Drug Administration (FDA) issued guidance for CV outcome trials to be performed for all new medications for patients with T2DM due to concerns for increased CV risk. Over the last decade, several large randomized controlled trials have reported statistically significant reductions in CV events using the novel antihyperglycemic medications listed below. Both classes reduced the risk of major adverse CV events to a comparable degree in patients with T2DM and established ASCVD across large meta-analyses (ADA Professional Practice Committee, 2022a).
Sodium-glucose cotransporter-2 (SGLT2) inhibitors. SGLT2 inhibitors are FDA-approved for use with other diabetic medications to lower blood sugar in adults with T2DM and include empagliflozin (Jardiance), canagliflozin (Invokana) and dapagliflozin (Farxiga). These agents work by causing the kidneys to excrete excess sugar through the urine and, over time, have an impact on reducing A1C levels. SGLT2 inhibitors have demonstrated a reduction in the progression of kidney disease across numerous clinical trials but have limited efficacy in patients with eGFR under 45 mL/ min/1.73 m2. They carry secondary benefits of weight loss, reductions in systolic BP, and circulating fluid levels (edema), all of which reduce the stress on the CV and renal systems. Since these agents cause increased diuresis, the most common side effects include dehydration, hypotension, syncope, and falls. They carry the rare but serious side effect of necrotizing fasciitis of the perineum, which is a severe infection of the genitals and surrounding area. These agents are also associated with an increased risk of yeast infection and urinary tract infection in females. There are ongoing investigations into case reports of SGLT2 inhibitor-associated diabetic ketoacidosis (DKA; Blonde et al., 2022). There are a few variances in efficacy and side effect profiles between the three agents, as follows:
- Empagliflozin (Jardiance) is the most well-established regarding CV risk reduction across clinical trials; it significantly reduces the risk of death from MI and stroke in adults with T2DM and CVD. The EMPA-REG OUTCOME trial was associated with significantly lower rates of all-cause and CV death and a lower risk of hospitalization for HF (Zinman et al., 2015). Therefore, the FDA added an indication for empagliflozin (Jardiance) to reduce the risk of CV mortality in adults with T2DM and CVD. Empagliflozin (Jardiance) is dosed as a 100 to 300 mg tablet taken orally once daily before breakfast (Blonde et al., 2022).
- Canagliflozin (Invokana) carries an increased risk of leg and foot amputation. The Canagliflozin Cardiovascular Assessment Study (CANVAS) trial demonstrated that over a year, the risk of amputation for patients in the trial was equivalent to 5.9 out of every 1,000 patients versus 2.8 out of every 1,000 patients treated with a placebo. The CANVAS study did find that canagliflozin (Invokana) significantly reduced CV mortality, MI, and stroke compared to the placebo (Neal et al., 2017). For this reason, it should not be prescribed to patients with PAD or a history of prior amputation, peripheral vascular disease, neuropathy, and diabetic foot ulcers. In addition, canagliflozin (Invokana) is associated with an increased risk of bone fractures associated with decreased bone mineral density (FDA, 2018). The Canagliflozin and Renal Events in Diabetes with Established Neuropathy Clinical Evaluation (CREDENCE) trial found that canagliflozin (Invokana) reduced end-stage renal disease (ESRD) by 32% and lowered the risk of CV mortality, MI, and stroke. In addition, canagliflozin (Invokana) had no significant increase in lower-limb amputations (Perkovic et al., 2019). Canagliflozin (Invokana) is dosed as a 5 to 10 mg tablet taken orally once daily before breakfast (Blonde et al., 2022).
- Dapagliflozin (Farxiga) is dosed as a 10 to 25 mg tablet taken orally once daily before breakfast (Blonde et al., 2022). In the Dapagliflozin Effect on Cardiovascular Events - Thrombosis in Myocardial Infarction (DECLARE-TIMI) trial, Dapagliflozin (Farxiga) reduced CV death and HF hospitalizations; however, it did not significantly lower the combined risk of CV death and nonfatal MI or stroke (Wiviott et al., 2019). The Dapagliflozin and Prevention of Adverse Outcomes in Chronic Kidney Disease (DAPA-CKD) trial found a lowered risk of ESRD, CV mortality, and hospitalization for HR in the dapagliflozin (Farxiga) group compared to the placebo (Heerspink et al., 2020).
Glucagon-like peptide 1 receptor agonists (GLP-1RAs). Clinical trial data have demonstrated CV risk reduction in patients with T2DM taking liraglutide (Victoza), semaglutide (Ozempic), and dulaglutide (Trulicity). These agents work by interfering with the functioning of GLP-1, an incretin peptide hormone released from the ileum and colon after intake. GLP-1 receptor activation stimulates insulin release, inhibits glucagon secretion, slows gastrointestinal transit, and suppresses appetite (Brown & Everett, 2019). They are typically reserved for those requiring two or more antidiabetic agents to reach and maintain their target A1C level. GLP-1RAs have significant A1C-lowering properties, with an average A1C reduction of 1.5%. GLP-1RAs also pose secondary benefits, including weight loss and lowering lipids and blood pressure, which can be cardioprotective. GLP-1RAs approved for CV risk reduction in patients with T2DM were only available as injectable medications. Therefore, mild injection site reactions are routinely reported, manifested as discomfort, inflammation, redness, or bruising at the injection site. In 2019, the FDA approved semaglutide (Rybelsus), the first oral GLP-1RA. Semaglutide (Rybelsus) is not recommended as a first choice GLP-1RA for T2DM because there is a black box warning about the potential risk for thyroid c-cell tumors. Patients with a family history of medullary thyroid carcinoma (MTC), a diagnosis of MTC, or multiple endocrine neoplasia syndrome type 2 (MEN2) should not take semaglutide (Rybelsus). GLP-1RAs can delay gastric emptying, causing fullness and nausea or vomiting. They should, therefore, be used with caution in patients with gastroparesis, those with a history of gastric bypass, or severe gastroesophageal reflux disease (GERD). Patients should be educated on the importance of staying well-hydrated and consuming smaller meals to avoid fullness and vomiting (Blonde et al., 2022; Munoz, 2018). There are a few variances in efficacy and side effect profiles between the three agents, as follows (Blonde et al., 2022; Munoz, 2018):
- Dulaglutide (Trulicity) is dispensed as a single-dose pen and prefilled syringe that needs to be refrigerated and protected from sunlight. Dulaglutide (Trulicity) dosing guidelines recommend starting with a 0.75 mg SC injection once every week, followed by dosing titration of up to 1.5 mg weekly if there is an inadequate glycemic response. Nausea is a commonly reported side effect in up to 12.4% of patients taking the 0.75 mg dose, which increases to 21.1% in patients taking the 1.5 mg dose. The average weight loss on dulaglutide (Trulicity) is 2.5 kg. The drug also carries a low risk of sinus tachycardia in up to 6% of patients.
- Liraglutide (Victoza) and semaglutide (Ozempic) are similar agents. They are dispensed as multi-dose pens and must be refrigerated before first use but can be stored at room temperature afterward. Their dosing regimens are also distinct, as Liraglutide (Victoza) is administered as a 0.6 mg subcutaneous (SC) injection daily for seven days, followed by 1.2 mg SC daily. If there is an inadequate glycemic response, dosing guidelines recommend increasing the dose to 1.8 mg SC daily. Semaglutide (Ozempic) is administered at a dose of 0.25 mg SC daily for four weeks, followed by an increase to 0.5 mg once weekly. The maximum dose of semaglutide (Ozempic) is 1.0 mg once weekly. Nausea occurs in up to 20% of patients taking these medications, and semaglutide (Ozempic) is associated with more significant weight loss than liraglutide (Victoza; 4.5 kg versus 2.5 kg). Liraglutide (Victoza) is FDA-approved to reduce the risk of CV mortality, nonfatal MI, and nonfatal stroke in adults with T2D and CVD.
Dual-targeted treatment. In May 2022, the FDA approved tirzepatide (Mounjaro) injection for use in patients with T2DM. Tirzepatide (Mounjaro) is the first approved dual-targeted therapy that activates GLP-1 and GIP receptors that help control glucose levels. It is administered by injection once weekly. Clinical trials have evaluated three different dosing options (5 mg, 10 mg, and 15 mg) as either stand-alone therapy or as an add-on to other T2DM medications. When patients received the 15 mg dose, tirzepatide (Mounjaro) effectively reduced A1C levels by 1.6% compared to the placebo in stand-alone trials and 0.5% compared to other diabetic medications. Tirzepatide (Mounjaro) also reduced weight by an average of 12 pounds compared to the placebo. Side effects can include nausea, vomiting, diarrhea, constipation, upper abdominal comfort, and decreased appetite. Tirzepatide (Mounjaro) did cause thyroid c-cell tumors in rats, but whether it causes these tumors in humans is unknown. Patients with a family history of MTC, a diagnosis of MTC, or MEN2 should not take tirzepatide (Mounjaro; FDA, 2022).
The ACC (n.d.-c) has established guidelines for when to consider adding an SGLT2-inhibitor or GLP-IRA to a patient's glucose-lowering regimen. These medications are intended to be used with the ADA Professional Practice Committee's (2022a) evidence-based CV risk reduction prescribing guidelines.
- at the time of T2DM diagnosis in patients with clinical ASCVD
- at the time of ASCVD diagnosis in patients with T2DM
- in patients with T2DM who are not meeting glycemic targets and have clinical ASCVD
- at the time of hospital discharge following admission for an ASCVD-related or T2DM-related clinical event
- consider initiating these medications for primary prevention in patients with T2DM who have additional risk factors for CVD, such as:
- age over 65 years
- poorly controlled hypertension (BP above 140/90 mmHg)
- hyperlipidemia (LDL above 100 mg/dl or non-HDL above 130 mg/dl)
- ongoing tobacco use
- CKD stage III or higher (ACC, n.d.-c)
The ADA Professional Practice Committee (2022a, 2022g) endorses the following antihyperglycemic prescribing recommendations to help guide clinical decision-making when choosing between an SGLT2-inhibitor or a GLP-1RA:
- First-line therapy depends on comorbidities, patient-centered factors, and management needs but generally includes metformin (Glucophage) and lifestyle management (A).
- SGLT2 inhibitors or GLP-1RA, with or without metformin (Glucophage), can be considered based on glycemic needs as an initial therapy for patients with T2DM with or at high risk for ASCVD, HF, or CKD (A).
- Metformin (Glucophage) should be continued upon initiating insulin therapy for ongoing glycemic and metabolic effects (A).
- In patients with T2DM, a GLP-1RA is preferred over insulin (A).
- If insulin is used, combination therapy with a GLP-1RA is recommended for greater efficacy (A).
- In patients with T2DM who have established ASCVD, established kidney disease, or multiple risk factors, either group will effectively reduce the risk of major CV events (A); however, SGLT2 inhibitors may be slightly better in those with diabetic kidney disease and HF (A).
- In patients with T2DM and established ASCVD or multiple risk factors for ASCVD, combined therapy with an SGLT-2-inhibitor and a GLP-1RA may be considered for an additive reduction in the risk of CV and kidney events (A).
- In patients with T2DM and established HF, an SGLT2 inhibitor may be considered to reduce the risk of HF hospitalization, worsening HF, and CV mortality (A).
- Medication regimens should be reevaluated at regular intervals (i.e., 3 to 6 months) and adjusted to incorporate factors that impact treatment choice (E).
Older Adults
Special consideration is required when prescribing and monitoring therapies in the older adult population to reduce adverse effects and increase adherence to therapy. The ADA Professional Practice Committee (2022a, 2022f) makes the following prescribing recommendations for older adults with T2DM:
- Consider the assessment of medical, psychological, functional, and social domains to determine the target and therapeutic approaches for T2DM management (B).
- Screen for polypharmacy, cognitive impairment, urinary incontinence, persistent pain, frailty, depression, and falls that can affect T2DM self-management (B).
- Older adults have a greater risk of hypoglycemia, and episodes of hypoglycemia should be ascertained at routine visits (B).
- In those at increased risk for hypoglycemia, medication classes with a low risk of hypoglycemia are preferred (B).
- Overtreatment of T2DM is common, contributes to hypoglycemia, and should be avoided (B).
- Deintensification (simplification) of complex treatment regimens can reduce the risk of hypoglycemia and polypharmacy, and promote compliance, by lowering the dose or discontinuing some medications (B).
- The cost of treatments must be considered to reduce the risk of cost-related nonadherence (B).
References
American College of Cardiology. (n.d.-a). 6 steps for optimizing cardiovascular risk reduction among patients with diabetes. Retrieved April 18, 2023, from https://www.cardiosmart.org/docs/default-source/assets/discussion-guides/diabetes-clinician-handout-6-steps.pdf
American College of Cardiology. (n.d.-b). ASCVD risk estimator plus. Retrieved April 18, 2023, from https://tools.acc.org/ascvd-risk-estimator-plus/#!/calculate/estimate
American College of Cardiology. (n.d.-c). Type 2 diabetes and cardiovascular risk toolkit. Retrieved April 18, 2023, from https://www.cardiosmart.org/docs/default-source/assets/discussion-guides/diabetes-discussion-guide.pdf
American Diabetes Association Professional Practice Committee. (2022a). Cardiovascular disease and risk management: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S144-S174. https://doi.org/10.2337/dc22-S010
American Diabetes Association Professional Practice Committee. (2022b). Facilitating behavior change and well-being to improve health outcomes: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S60-S82. https://doi.org/10.2337/dc22-S005
American Diabetes Association Professional Practice Committee. (2022c). Glycemic targets: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S83-S96. https://doi.org/10.2337/dc22-S006
American Diabetes Association Professional Practice Committee. (2022d). Introduction: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S1-S2. https://doi.org/10.2337/dc22-Sint
American Diabetes Association Professional Practice Committee. (2022e). Obesity and weight management for the prevention and treatment of type 2 diabetes: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S113-S124. https://doi.org/10.2337/dc22-S008
American Diabetes Association Professional Practice Committee. (2022f). Older adults: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S195-S207. https://doi.org/10.2337/dc22-S013
American Diabetes Association Professional Practice Committee. (2022g). Pharmacologic approaches to glycemic treatment: Standards of medical care in diabetes-2022. Diabetes Care, 45(Suppl. 1), S125-S143. https://doi.org/10.2337/dc22-S009
American Geriatrics Society Beers Criteria Update Expert Panel. (2019). American Geriatrics Society 2019 Updated AGS Beers Criteria for potentially inappropriate medication use in older adults. Journal of the American Geriatrics Society, 67(4), 674-694. https://doi.org/10.1111/jgs.15767
American Heart Association. (2021). Cardiovascular disease & diabetes. https://www.heart.org/en/health-topics/diabetes/diabetes-complications-and-risks/cardiovascular-disease--diabetes
Bloch, M. J., & Basile, J. (2022). Major side effects and safety of calcium channel blockers. UpToDate. Retrieved April 19, 2023, from https://www.uptodate.com/contents/major-side-effects-and-safety-of-calcium-channel-blockers
Blonde, L., Umpierrez, G. E., Reddy, S. S., McGill, J. B., Berga, S. L., Bush, M., Chandrasekaran, S., DeFronzo, R. A., Einhorn, D., Galindo, R. J., Gardner, T. W., Garg, R., Garvey, W. T., Hirsch, I. B., Hurley, D. L., Izuora, K., Kosiborod, M., Olson, D., Patel, S., . . . Weber, S. L. (2022). American Association of Clinical Endocrinology clinical practice guideline: Developing a diabetes mellitus comprehensive care plan - 2022 update. Endocrine Practice, 28(10), 923-1049. https://doi.org/10.1016/j.eprac.2022.08.002
Brown, J. M., & Everett, B. M. (2019). Cardioprotective diabetes drugs: What cardiologists need to know. Cardiovascular Endocrinology & Metabolism, 8(4), 96-105. https://doi.org/10.1097/XCE.0000000000000181
Brutsaert, E. F. (2022). Diabetes mellitus (DM). Merck Manual Professional Version. https://www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/diabetes-mellitus-and-disorders-of-carbohydrate-metabolism/diabetes-mellitus-dm
Centers for Disease Control and Prevention. (2022a). Heart disease facts. https://www.cdc.gov/heartdisease/facts.htm
Centers for Disease Control and Prevention. (2022b). National diabetes statistics report. https://www.cdc.gov/diabetes/data/statistics-report/index.html
Centers for Disease Control and Prevention. (2023). Know your risk for heart disease. https://www.cdc.gov/heartdisease/risk_factors.htm
Das, S. R., Everett, B. M., Birtcher, K. K., Brown, J. M., Januzzi, J. L., Kalyani, R. R., Kosiborod, M., Magwire, M., Morris, P. B., Neumiller, J. J., & Sperling, L. S. (2020). 2020 expert consensus decision pathway on novel therapies for cardiovascular risk reduction in patients with type 2 diabetes: A report of the American College of Cardiology solution set oversight committee. Journal of the American College of Cardiology, 76(9), 1117-1145. https://doi.org/10.1016/j.jacc.2020.05.037
Dudenbostel, T., & Calhoun, D. A. (2017). Use of aldosterone antagonists for treatment of uncontrolled resistant hypertension. American Journal of Hypertension, 30(2), 103-109. https://doi.org/10.1093/ajh/hpw105
Einarson, T. R., Acs, A., Ludwig, C., & Panton, U. H. (2018). Prevalence of cardiovascular disease in type 2 diabetes: A systematic literature review of scientific evidence from across the world in 2007-2017. Cardiovascular Diabetology, 17(83), 1-19. https://doi.org/10.1186/s12933-018-0728-6
Fishman, S. L., Sonmez, H., Basman, C., Singh, V., & Poretsky, L. (2018). The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: A review. Molecular Medicine, 24(59), 1-12. https://doi.org/10.1186/s10020-018-0060-3
Giugliano, R. P., Cannon, C. P., Blazing, M. A., Nicolau, J. C., Corbalan, R., Spinar, J., Park, J. G., White, J. A., Bohula, E. A., & Braunwald, E. (2018). Benefit of adding ezetimibe to statin therapy on cardiovascular outcomes and safety in patients with versus without diabetes mellitus: Results from IMPROVE-IT (improved reduction of outcomes: Vytorin efficacy international trial). Circulation, 137(15), 1571-1582. https://doi.org/10.1161/CIRCULATIONAHA.117.030950
Goyal, R., & Jialal, I. (2022). Diabetes mellitus type 2. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK513253
Heerspink, H. J. L., Stefansson, B. V., Correa-Rotter, R., Chertow, G. M., Greene, T., Hou, F., Mann, J. F. E., McMurray, J. J. V., Lindberg, M., Rossing, P., Sjostrom, C. D., Toto, R. D., Langkilde, A., & Wheeler, D. C. (2020). Dapagliflozin in patients with chronic kidney disease. New England Journal of Medicine, 383, 1436-1446. https://doi.org/10.1056/NEJMoa2024816
Heidenreich, P. A., Bozkurt, B., Aguilar, D., Allen, L. A., Byun, J. J., Colvin, M. M., Deswal, A., Drazner, M. H., Dunlay, S. M., Evers, L. R., Fang, J. C., Fedson, S. E., Fonarow, G. C., Hayek, S. S., Hernandez, A. F., Khazanie, P., Kittleson, M. M., Lee, C. S., Link, M. S., . . . Yancy, C. W. (2022). 2022 AHA/ACC/HFSA guideline for the management of heart failure: A report of the American College of Cardiology/American Heart Association Joint Committee on clinical practice guidelines. Circulation, 145(18), e895-e1032. https://doi.org/10.1161/CIR.0000000000001063
Hennekens, C. H. (2021). Aspirin for the secondary prevention of atherosclerotic cardiovascular disease. UpToDate. Retrieved April 19, 2023, from https://www.uptodate.com/contents/aspirin-for-the-secondary-prevention-of-atherosclerotic-cardiovascular-disease
Hudspeth, B. (2018). The burden of cardiovascular disease in patients with diabetes. American Journal of Managed Care, 24(13), S268-S272. https://www.ajmc.com/view/the-burden-of-cardiovascular-disease-in-patients-with-diabetes
Ignatavicius, D. D., Workman, M. L., Rebar, C. R., & Heimgartner, N. M. (2021). Medical-surgical nursing: Concepts for interprofessional collaborative care (10th ed.). Elsevier
International Diabetes Federation. (2016). Diabetes and cardiovascular disease – executive summary. https://www.idf.org/our-activities/care-prevention/cardiovascular-disease/cvd-report/9-diabetes-and-cardiovascular-disease-executive-summary.html
Janus, A., Szahidewicz-Krupska, E., Mazur, E., & Doroszko, A. (2016). Insulin resistance and endothelial dysfunction constitute a common therapeutic target in cardiometabolic disorders. Mediators of Inflammation, 1-10. https://doi.org/10.1155/2016/3634948
Katakami, N. (2018). Mechanism of development of atherosclerosis and cardiovascular disease in diabetes mellitus. Journal of Atherosclerosis and Thrombosis, 25(1), 27-39. https://doi.org/10.5551/jat.RV17014
Leopold, J. A., & Ingelfinger, J. R. (2023). Aldosterone and treatment-resistant hypertension. New England Journal of Medicine, 388, 464-467. https://doi.org/10.1056/NEJMe2213559
Lopez, E. O., Ballard, B. D., & Jan, A. (2022). Cardiovascular disease. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK535419
Low Wang, C. C., Hess, C. N., Hiatt, W. R., & Goldfine, A. B. (2016). Clinical update: Cardiovascular disease in diabetes mellitus. Circulation, 133(24), 2459-2502. https://doi.org/10.1161/CIRCULATIONAHA.116.022194
Munoz, K. M. (2018). GLP-1 receptor agonists for type 2 diabetes currently available in the US. http://www.diabetesincontrol.com/wp-content/uploads/2014/09/GLP-1-Chart-Nov-1-2018.pdf
The National Heart, Lung, and Blood Institute. (2013). Coronary artery disease [Image]. https://www.nhlbi.nih.gov/health/coronary-heart-disease/causes
Neal, B., Perkovic, V., Mahaffey, K. W., de Zeeuw, D., Fulcher, G., Erondu, N., Shaw, W., Law, G., Desai, M., & Matthews, D. R. (2017). Canagliflozin and cardiovascular and renal events in type 2 diabetes. New England Journal of Medicine, 377, 644-657. https://doi.org/10.1056/NEJMoa1611925
Nestro, R. W. (2023). Prevalence of and risk factors for coronary heart disease in patients with diabetes mellitus. UpToDate. Retrieved April 19, 2023, from https://www.uptodate.com/contents/prevalence-of-and-risk-factors-for-coronary-heart-disease-in-patients-with-diabetes-mellitus
Perkovic, V., Jardine, M. J., Neal, B., Bompoint, S., Heerspink, H. J. L., Charytan, D. M., Edwards, R., Agarwal, R., Bakris, G., Bull, S., Cannon, C. P., Capuano, G., Chu, P., deZeeuw, D., Greene, T., Levin, A., Pollock, C., Wheeler, D. C., Yavin, Y., . . . Mahaffey, K. W. (2019). Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. New England Journal of Medicine, 380, 2295-2306. https://doi.org/10.1056/NEJMoa1811744
Rodriguez, V., Weiss, M., Weintraub, H., Goldberg, I., & Schwartzbard, A. (2017). Cardiovascular disease leads to a new algorithm for diabetes treatment. Journal of Clinical Lipidology, 11(5), 1126-1133. https://doi.org/10.1016/j.jacl.2017.07.004
Rosenson, R. S. (2023). Low-density lipoprotein cholesterol lowering with drugs other than statins and PSCK9 inhibitors. UpToDate. Retrieved April 19, 2023, from https://www.uptodate.com/contents/low-density-lipoprotein-cholesterol-lowering-with-drugs-other-than-statins-and-pcsk9-inhibitors
Scheen, A. J. (2018). Type 2 diabetes and thiazide diuretics. Current Diabetes Reports, 18(2), 6. https://doi.org/10.1007/s11892-018-0976-6
Stroes, E. S. G., Stiekema, L. C. A., & Rosenson, R. S. (2023). PCSK9 inhibitors: Pharmacology, adverse effects, and use. UpToDate. Retrieved April 19, 2023, from https://www.uptodate.com/contents/pcsk9-inhibitors-pharmacology-adverse-effects-and-use
US Food & Drug Administration. (2018). Sodium-glucose cotransporter-2 (SGLT2) inhibitors. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/sodium-glucose-cotransporter-2-sglt2-inhibitors
US Food & Drug Administration. (2019). FDA approves first oral GLP-1 treatment for type 2 diabetes. https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-glp-1-treatment-type-2-diabetes
US Food & Drug Administration. (2022). FDA approves novel, dual-targeted treatment for type 2 diabetes. https://www.fda.gov/news-events/press-announcements/fda-approves-novel-dual-targeted-treatment-type-2-diabetes
Walsh, D., & Sved, A. (2019). Insulin actions and stimuli for secretion [Image]. Wikimedia. https://commons.wikimedia.org/wiki/File:Insulin_-_actions_and_stimuli_for_secretion.png
Ward, N. C., Watts, G. F., & Eckel, R. H. (2019). Statin toxicity. Circulation Research, 124, 328-350. https://doi.org/10.1161/CIRCRESAHA.118.312782
Wiviott, S. D., Raz, I., Bonaca, M. P., Mosenzon, O., Kato, E. T., Cahn, A., Silverman, M. G., Zelniker, T. A., Kuder, J. F., Murphy, S. A., Bhatt, D. L., Leiter, L. A., McGuire, D. K., Wilding, J. P. H., Ruff, C. T., Guase-Nilsson, I. A. M., Fredriksson, M., Johansson, P. A. M, Langkilde, A. M., Sabatine, M. S. (2019). Dapagliflozin and cardiovascular outcomes in type 2 diabetes. New England Journal of Medicine, 380(4), 347-355. https://doi.org/10.1056/NEJMoa1812389
Woods, A. D. (2023). Nursing 2023 drug handbook (43rd ed.). Wolters Kluwer.
Yugar-Toledo, J. C., Modolo, R., de Faria, A. P., & Moreno, H. (2017). Managing resistant hypertension: Focus on mineralocorticoid-receptor antagonists. Vascular Health and Risk Management, 13, 403-411. https://doi.org/10.2147/VHRM.S138599
Zinman, B., Wanner, C., Lachin, J. M., Fitchett, D., Bluhmki, E., Hantel, S., Mattheus, M., Devins, T., Johansen, O. D., Woerle, H. J., Broedl, U. C., & Inzucchi, S. E. (2015). Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. New England Journal of Medicine, 373, 2117-2128. https://doi.org/10.1056/NEJMoa1504720