About this course:
This module provides an overview of ovarian cancer, including its risk factors, signs and symptoms, and treatment options, to help diagnose women as early as possible and provide optimal care and support throughout the disease trajectory. Empowering nurses with the right tools and information can significantly impact the clinical outcomes of women affected by this disease.
Course preview
Ovarian Cancer
This module provides an overview of ovarian cancer, including its risk factors, signs and symptoms, and treatment options, to help diagnose women as early as possible and provide optimal care and support throughout the disease trajectory. Empowering nurses with the right tools and information can significantly impact the clinical outcomes of women affected by this disease.
By the completion of this module, the nurse should be able to:
- outline the epidemiology of ovarian cancer in the US and common risk factors
- review the pathophysiology of ovarian cancer, identify the signs and symptoms of the disease, and discuss ovarian cancer subtypes
- review the diagnostic workup and staging criteria for ovarian cancer
- discuss treatment options, including surgery, chemotherapy, targeted therapy, immunotherapy, and hormonal therapy, and the most common side effects, monitoring, and components of patient education
Ovarian cancer is the second most common gynecologic malignancy in the US, leading to more deaths than any other female reproductive cancer. Ovarian cancer is a disease in which malignant (i.e., cancerous) cells originate within the ovaries, fallopian tubes, or peritoneum (primary peritoneal cancer [PPC]). These three cancers are grouped into one disease based on their anatomical proximity and similarities in risk factors and medical treatments. In this module, ovarian cancer is the term used to describe all three cancers unless otherwise specified. Nurses must be informed about the clinical features of ovarian cancer, as early diagnosis is essential for improving survival rates. Treatment options vary depending on the stage of the disease and patients’ overall health. Nurses support patients and their families throughout the disease trajectory, providing education about the disease, treatment, and side effects and offering emotional support (Centers for Disease Control and Prevention [CDC], 2023b; National Comprehensive Cancer Network [NCCN], 2023b).
Epidemiology
According to the American Cancer Society (ACS, 2023a), there will be an estimated 19,710 new cases and 13,270 deaths from ovarian cancer this year, a decline from 2020 estimates of 21,750 new cases and 13,940 deaths. While ovarian cancer is uncommon, accounting for only 1% of all new cancer diagnoses in the US, it ranks fifth among cancer-related deaths in women and is the most lethal gynecological malignancy. Based on data from the National Cancer Institute’s (NCI, 2023c) Surveillance, Epidemiology, and End Results Program (SEER), the median age at diagnosis is 63; it is most frequently diagnosed in women aged 55 to 64 (24.5%), followed by women aged 65 to 74 (24.1%). The average lifetime risk for developing ovarian cancer among the general US population is roughly 1 in 78, and the lifetime risk of dying is around 1 in 108 (ACS, 2023a). The median age at death is 70 years, with the highest percentage of deaths among women aged 65 to 74 (29.1%), followed by those aged 75 to 84 (24.9%). The 5-year overall survival rate for ovarian cancer is 50.8%, which drops to 31.5% for patients with distant metastases (i.e., cancer spread) at diagnosis (NCI, 2023c).
Incidence rates are highest for non-Hispanic American Indian/Alaska Native women (11.3 per 100,000), followed by non-Hispanic White (10.5 per 100,000), Hispanic (10.0 per 100,000), and non-Hispanic Asian/Pacific Islander (9.3 per 100,000). Based on the latest age-adjusted data from 2016-2020, Black women have the lowest incidence rate (8.9 per 100,000). Statistical analyses demonstrate declining new ovarian cancer diagnoses and deaths over the last decade. Between 2008 and 2017, the age-adjusted rates for new ovarian cancer diagnoses have decreased on average by 2.5% each year, and death rates have fallen by 2.3% per year between 2009 and 2018. Non-Hispanic White women have the highest mortality (6.7 per 100,000), followed by non-Hispanic American Indian/Alaska Native (6.4 per 100,000; NCI, 2023c).
Risk and Protective Factors
Although ovarian cancer is rare, all women are at risk, and the risk increases with age. Hormonal, environmental, and genetic risk factors all serve roles in the development of the condition. Although most women are diagnosed without identifiable risk factors, several influences can increase or decrease a person’s risk of developing the disease. Table 1 describes the major risk and preventative factors (Yarbro et al., 2018).
Table 1
Ovarian Cancer Risk and Protective Factors
Risk Factors Anything that increases the chance of developing ovarian cancer | |
Factor | Description of Evidence |
Advancing age | Over 50% of all ovarian cancers are diagnosed in women aged 63 and older. |
Body mass index (BMI) | A BMI of 30 or higher is associated with an increased risk, and obesity is linked to an increased risk of death from ovarian cancer. |
Family history
Refer to the next section on the inherited risk of ovarian cancer | Women are at increased risk if they have a first-degree relative (i.e., mother, sister, or daughter) with ovarian cancer. The risk heightens as the number of relatives with ovarian cancer increases. The lifetime risk of ovarian cancer is 1.6% in women without a family history of the disease, 5% in women with an affected first-degree relative, and 7% in women with two or more affected first-degree relatives. A family history of other types of cancers, such as colorectal or breast cancer, increases ovarian cancer risk. |
Pregnancy
| Nulliparity (i.e., those who have never carried a pregnancy to term) is one of the strongest risk factors for ovarian cancer since these women tend to have more ovulatory cycles. Women who had their first full-term pregnancy after age 35 are also at increased risk. |
Hormone replacement therapy (HRT) | Taking HRT after menopause (i.e., postmenopausal estrogen alone or estrogen with progesterone) is associated with an increased risk for ovarian cancer, even if taken for less than five years. The risk rises alongside the duration of use, but after HRT is discontinued, the risk declines gradually. |
Medical history | A medical history of endometriosis (i.e., a condition in which tissue from the lining of the uterus grows on other pelvic organs such as the ovaries or fallopian tubes) or breast cancer is associated with an increased risk of ovarian cancer. |
In vitro fertilization (IVF) | While infertility treatments (particularly IVF) are linked to an increased risk for ovarian tumors known as “borderline” or “low malignant potential,” research has not demonstrated a causal relationship between IVF treatments...
...purchase below to continue the course |
Talcum (talc) powder | Although there has been heightened attention on the risk for ovarian cancer in women who have used talc powder (i.e., dusted on the perineum, which is the area between the vagina and the anus), studies have not found clear evidence supporting this relationship. |
Protective Factors Anything that decreases the chance of developing ovarian cancer | |
Factor | Description of Evidence |
Pregnancy | Women who have their first full-term pregnancy before age 26 have a lower risk for ovarian cancer, which declines with each subsequent pregnancy. |
Breastfeeding | Breastfeeding is associated with a reduced risk of invasive ovarian cancer across multiple studies. Babic and colleagues (2020) evaluated 9,973 women with ovarian cancer and 13,843 controls and found that breastfeeding was associated with a 24% overall reduced risk. Breastfeeding for 12 months or longer was associated with a 34% lower risk. |
Oral contraceptive pills (OCPs) | Women who have taken OCPs have a 30% to 50% lower risk for ovarian cancer than women who never took OCPs, and the risk declines with increased duration of use. The protective features of OCPs can continue for up to 30 years after they are discontinued. In contrast, some studies suggest that women taking estrogen alone (without progesterone) for ten or more years may have an increased risk of ovarian cancer. The risk reduction for ovarian cancer with OCP use also applies to women with inherited mutations in BRCA1 or BRCA2 genes. |
Prophylactic (preventative) surgery | A hysterectomy (i.e., uterus removal) reduces ovarian cancer risk by about one-third (33%). Risk-reducing salpingo-oophorectomy (RRSO) involves removing both ovaries and fallopian tubes. Research has demonstrated that RRSO in high-risk women (i.e., usually those with BRCA1 or BRCA2 gene mutations) effectively lowers the risk of ovarian and fallopian tube cancers and death. RRSO can reduce the risk of BRCA-related ovarian cancer by 80% to 90%. In premenopausal women, RRSO can reduce the risk of estrogen-driven breast cancers by up to 50%. RRSO does not reduce the risk of PPC, although the risk is minimal. |
(ACS, 2023a, 2023b; CDC, 2023a; Facing Our Risk of Cancer Empowered [FORCE], 2023; Mayo Clinic, 2022; NCI, 2018, 2023b; Woo & Long, 2021)
Inherited Risk
BRCA mutations
Up to 25% of ovarian cancers are part of familial cancer syndromes resulting from inherited mutations in specific genes. Approximately 3% of women with two or more first-degree relatives with ovarian cancer will have a hereditary cancer syndrome (Woo & Long, 2021). Mutations in BRCA1 and BRCA2 (BRCA1/2) genes commonly lead to hereditary breast and ovarian cancer syndrome (HBOC), characterized by a heightened lifetime risk of developing breast and ovarian cancer. Under healthy conditions, BRCA1/2 genes function as tumor suppressors and promote the normal and healthy growth, development, and division of various cells in the body. When these genes are mutated, growth processes are unregulated, increasing the propensity toward cancer development. Mutations in BRCA1/2 genes are inherited via the autosomal dominant inheritance pattern; one copy of the mutated gene in each cell is sufficient to increase the risk of developing cancer. Although ovarian cancer is a sex-dependent disease, the altered gene can be inherited from either sex parent. Each child of a parent with a BRCA1 or BRCA2 mutation has a 50% chance of inheriting the same gene mutation (CDC, 2023a, 2023c).
Approximately 1 in 400 women in the US inherit a mutation in a BRCA gene. Mutations in BRCA1/2 are about ten times more common in women of Ashkenazi Jewish descent than in the general US population. Roughly 10% of ovarian cancers (i.e., nearly 2,000 women per year) are attributed to inherited mutations in the BRCA1/2 genes. Patients with BRCA1/2 mutations are more likely to be diagnosed with high-grade serous ovarian cancer than other histologic subtypes. Approximately 30 in 100 women with an inherited BRCA1 or BRCA2 mutation will be diagnosed with ovarian cancer by age 70. A mutation in the BRCA1 gene is more closely linked to ovarian cancer, posing a lifetime risk between 35% and 70%. Women with BRCA1 mutations tend to be diagnosed with ovarian cancer up to a decade younger than women with ovarian cancer not associated with a known genetic mutation. Women with a BRCA2 mutation have a 10% to 30% lifetime risk of developing ovarian cancer by age 70. By comparison, about 1.1% of women will be diagnosed with ovarian cancer sometime during their lifetime (CDC, 2023a, 2023c; NCI, 2023a, 2023c; Temkin et al., 2018).
Hereditary nonpolyposis colorectal cancer (HNPCC)
HNPCC, or Lynch syndrome, is known for its high-risk association with colorectal cancer; however, it also correlates with an increased risk of ovarian and endometrial (uterine) cancers. The lifetime risk of ovarian cancer in women with HNPCC is 10%. Although changes in the MLH1, MSH2, MSH6, or PMS2 genes are the most common culprits, many different genes can cause this syndrome. Under physiologic conditions, these genes are responsible for repairing potential errors during DNA replication (i.e., the process during which deoxyribonucleic acid [DNA] is copied in preparation for cell division); collectively, they are known as mismatch repair (MMR) genes. Since mutations in any of these genes impede the cells’ ability to repair DNA replication errors, abnormal cells continue to divide. Over time, accumulated DNA replication errors lead to uncontrollable cell growth and malignant tumors (US National Library of Medicine [NLM], 2021). MUTYH-associated polyposis (MAP) is a disorder caused by a mutation in the MUTYH gene, which prevents cells from fixing DNA errors. As these errors accumulate, more polyps develop, increasing the likelihood of cancer. Pathogenic mutations in the MUTYH gene are associated with a 6% to 14% lifetime risk for ovarian cancer with a median age at diagnosis of 51 years (Nielson et al., 2021). Additional genes linked to ovarian cancer with evolving clinical evidence include ATM, BRIP1, NBN, STK11, RAD51C, RAD51D, and PALB2 (Kurian et al., 2017).
Tumor Protein p53 (TP53)
Somatic (or acquired) mutations occur during a person’s lifetime due to environmental exposures, such as sun ultraviolet (UV) radiation, free radicals, carcinogen exposure, ionizing background radiation, and chemical exposure. Somatic mutations only occur in specific cells in the body. Unlike inherited mutations, these changes do not appear in families and are not passed between generations. TP53 gene mutations are common in ovarian cancer, occurring in at least half of diagnoses. Under healthy conditions, TP53 functions as a tumor suppressor gene, regulating cellular growth and division by preventing cells from growing and dividing too quickly or uncontrollably. TP53 prevents cells with mutated or damaged DNA from dividing, thereby preventing tumor formation. Mutations in TP53 impair its ability to control cell proliferation, as it cannot trigger apoptosis (i.e., programmed cell death) in cells with mutated or damaged DNA. Consequently, DNA damage accumulates, cells divide uncontrollably, and tumors form (NLM, 2020; Oien & Chien, 2016).
Pathophysiology
The ovaries and fallopian tubes are primary components of the female reproductive system. The ovaries, or female gonads, are a pair of organs located in the pelvis in a region called the ovarian fossa. The ovaries appear on either side of the uterus, which is the hollow, pear-shaped organ that carries a fetus (i.e., the womb). The ovaries are attached to the uterus by the ovarian ligament, which is embedded within the broad ligament. Figure 1 demonstrates the anatomical structures and positioning of the female reproductive system (McCance & Heuther, 2019).
The ovaries serve two primary functions: secretion of the female sex hormones and development and release of the female ova (i.e., egg). Oocytes are immature egg cells that develop and mature within the ovarian follicles. The ovaries comprise three major cell types: epithelial, germ, and stromal. The tunica albuginea is a thin layer of dense connective tissue covering the ovary’s outermost aspect. The cortex appears directly beneath the tunica albuginea and consists of a framework of tissues called the ovarian stroma. The cortex forms the bulk of the ovary and nurtures the follicles at various stages of maturation. The medulla is the middle layer of the ovary; it comprises loose connective tissues, elastic fibers, and neurovascular structures, including the lymph and blood vessels. The hilum is the ovary’s innermost layer and provides the entry point for the blood vessels and nerves (Lumen Learning, n.d.; McCance & Heuther, 2019).
Figure 1
Female Reproductive System

(iStock photo ID: 538949875)
The hypothalamic-pituitary-gonadal (HPG) axis is a tightly regulated feedback system that controls female sex hormones and reproduction. The HPG forms a complex interplay between the organs of the female endocrine system, including the hypothalamus, pituitary gland, and ovaries. During puberty, the hypothalamus releases increasing levels of gonadotropin-releasing hormone (GnRH). In response, the anterior pituitary produces follicle-stimulating hormone (FSH) and luteinizing hormone (LH), prompting the ovaries to secrete increasing amounts of sex hormones, primarily estrogen and progesterone, along with a smaller quantity of testosterone (Lumen Learning, n.d.; McCance & Heuther, 2019).
Ovarian follicles are tiny fluid-filled sacs composed of granulosa cells, theca cells, and oocytes, varying according to each oocyte’s maturation stage. Folliculogenesis (i.e., the maturation of ovarian follicles) involves several growth factors and receptors. This process relies primarily on the HPG axis, as coordinated interactions between the granulosa and theca cells are necessary for estrogen synthesis. Granulosa cells form alongside the maturing follicle. FSH has a high affinity for granulosa cells, promoting follicle growth and maturation by synthesizing androgens (i.e., testosterone) and estrogen. Theca cells consist of connective tissue and blood vessels surrounding the ovarian follicle. They produce estradiol from androgens in response to LH. Estradiol is an estrogen steroid hormone that binds to tissues in the body, serving as the body’s primary estrogen source during reproductive years. Estradiol influences secondary sex characteristics such as breast growth, body shape, muscle mass, fat deposition, body hair, and bone health. It also regulates the menstrual cycle and serves a vital role in pregnancy. When the maturation process is complete, the oocyte becomes an ovum. The pituitary gland releases a surge of LH, stimulating the follicle’s rupture and ova release (Holesh et al., 2023; Lumen Learning, n.d.).
The ovaries typically alternate, releasing a single mature egg each month; however, if an ovary is dysfunctional or absent, the other ovary will compensate. The ovaries change in size and appearance throughout a person’s lifetime. Their size and appearance depend on the individual’s menstrual status. Menopause denotes menstruation’s permanent cessation in females with an intact uterus, defined as 12 consecutive months without a menstrual cycle (Holesh et al., 2023). According to the Office on Women’s Health (OWH), the average age for menopause in the US is 52 years. In premenopausal women (i.e., women of reproductive age who are menstruating), each ovary is comparable in size and shape to an almond, measuring 3 to 5 cm long and weighing 4 to 8 g. Normal ovarian volume starts to decrease after age 30. During menopause, reproductive hormone levels decline, egg production ceases, and the ovaries shrink as they undergo progressive involution (OWH, 2021).
The fallopian tubes are a pair of long, slim conduits on either side of the uterus that transport eggs from the ovaries to the uterus. The tubes attach to the uterus and curve up and over the ovaries, as shown in Figure 1. Each tube is 8 to 12 cm long and 1 cm in diameter; they flare into a bell shape at the ovarian end, called the infundibulum. The infundibulum contains fimbriae, which are swaying fringes that create a current to draw the ova into them. Once the ova enter the fallopian tube, cilia and peristalsis gradually propel them toward the uterus. The omentum is a layer of adipose (fatty) tissue that covers the intestines like an apron and is subdivided into the greater omentum (which drapes over the stomach) and the lesser omentum (which drapes over the liver). The peritoneum is a cellophane-like, moist sheet of tissue covering the surface of the organs in the abdomen and pelvis. It supports and protects the organs and consists of two layers: the visceral peritoneum and the parietal peritoneum. The visceral peritoneum is the inner layer of tissue lining the organs. The parietal peritoneum is the outer layer that adheres to the abdominal cavity wall. The space between the layers is called the peritoneal cavity, containing a small quantity of lubricating fluid (McCance & Heuther, 2019).
Ovarian Cancer
Many types of ovarian cancer initially develop in the distal portions of the fallopian tubes and spread to the ovary. Emerging research has found the fallopian tubes may be the source of most hereditary ovarian cancers. PPC appears to start in the cells lining the fallopian tubes, spreading to the ovaries (ACS, 2023b). Ovarian cancer is an aggressive disease characterized by rapid growth, disseminated metastasis, and malignant ascites. Ascites is the abnormal buildup of fluid within the abdominal or pelvic cavity. Without an anatomic barrier surrounding the primary tumor, cancer cells easily infiltrate the peritoneal cavity, making the peritoneum and omentum the most common sites of initial metastases. Metastases often occur by direct extension as the cancer penetrates and ruptures the ovarian capsule, permitting invasion into surrounding structures. Ovarian cancer cells can survive after detaching from the primary tumor by forming multicellular spheroids. The spheroids can float in ascitic fluid, making the intraperitoneal cavity a predominant metastasis site. The continuous circulation of free peritoneal fluid facilitates the widespread dissemination of cancerous cells, a process referred to as peritoneal seeding. As the disease advances, a higher volume of ascites accumulates, causing cancer to spread to distant sites in the abdomen (Motohara et al., 2018). Ovarian cancer cells can also travel through the lymphatics, most commonly the pelvic or aortic lymph nodes. Some of the most common sites for distant metastases include the liver, pleura (i.e., the serous membrane lining the lungs), diaphragm, intestines, spleen, brain, bladder, and skin (Mitra, 2016; Yarbro et al., 2018). Although PPC tends to spread along the pelvic and abdomen cavity surface, the precise origin is difficult to determine (ACS, 2023b).
Ovarian cancer is classified as type I or type II. Type I cancers are early-stage, low-grade tumors that generally progress from benign precursor lesions (i.e., endometriosis). These cancers tend to remain indolent or grow slowly. In contrast, most ovarian cancers are type II, which are more aggressive and grow rapidly. Type II cancers are primarily diagnosed at advanced stages and account for most ovarian cancer-related deaths. They usually originate in the fallopian tube or the surface epithelium of the ovary. Type II ovarian cancers are more commonly associated with BRCA1/2 genetic mutations (Salazar et al., 2018; Yarbro et al., 2018).
Epithelial Ovarian Cancer
Epithelial ovarian cancer accounts for at least 90% of cases and is divided into five major subtypes, as outlined in Table 2 (Salazar et al., 2018; Yarbro et al., 2018).
Table 2
Epithelial Ovarian Cancer Subtypes
Histologic Subtype | Type I or II | Clinical Features |
High-grade serous carcinoma (HGSC) | Type II |
|
Low-grade serous carcinoma (LGSC) | Type I |
|
Endometrioid carcinoma | Type I |
|
Clear cell carcinoma (CCC) | Type 1 |
|
Mucinous carcinoma | Type 1 |
|
(Koshiyama et al., 2017; Oien & Chien, 2016)
Less common types of ovarian cancer include germ cell tumors, sex-cord stromal cell tumors, and small cell carcinoma of the ovary (Koshiyama et al., 2017; Oien & Chien, 2016)
Germ Cell Tumors
Germ cell tumors start from the cells that form the ova, and most cases are benign. Mature teratomas are the most common type of benign germ cell tumors and are usually treated surgically. Malignant ovarian germ cell tumors only account for about 5% of ovarian cancers. Unlike epithelial ovarian cancers, these characteristically occur in adolescents and premenopausal women. They are often confined to an ovary and are usually curable. Immature teratomas are malignant tumors that primarily develop in females under 18. These can metastasize to other areas in the body. Dysgerminomas are the most common type of malignant germ cell tumors. They usually affect females under 30 and are cured with surgery; less than 25% of cases require additional treatment following surgery (ACS, 2023b; Ovarian Cancer Research Alliance [OCRA], n.d.; Yarbro et al., 2018).
Sex-Cord Stromal Cell Tumors
Ovarian sex-cord stromal tumors are rare, accounting for under 2% of all malignant ovarian cancers. These tumors originate in the stromal cells, which hold the ovary together and produce the female sex hormones. The most common subtype is the granulosa cell tumor. Patients with stromal tumors typically show signs of excess hormone production like hirsutism (i.e., coarse hair growth on unexpected body areas such as the face or chest) and virilization. Virilization is a condition in which a female develops features of increased masculinization. Some of the most common symptoms include baldness, acne, deepening of the voice, increased libido (i.e., sex drive), decreased size of the breasts, and menstrual irregularities, including cessation of menses (ACS, 2023b; OCRA, n.d.; Yarbro et al., 2018).
Small Cell Carcinoma of the Ovary (SCCO)
SCCO is a rare type of ovarian cancer, accounting for 0.1%. It typically affects young women, as the median age of diagnosis is 23 years, and approximately 70% of patients have hypercalcemia (OCRA, n.d.). Hypercalcemia of malignancy (HCM) is a complex metabolic disorder and oncologic emergency due to bone destruction and elevated renal absorption of calcium. Three primary mechanisms cause HCM:
- excess secretion of parathyroid hormone-related protein
- bone metastases release osteoclast activating factors, causing an imbalance between bone formation and bone resorption, leading to the excess release of calcium into the bloodstream
- overproduction of 1,25-dihydroxy vitamin D (Calcitriol)
HCM most commonly occurs in breast cancer, squamous cell lung cancer, and multiple myeloma. Symptoms can be vague, ranging from fatigue, anorexia, nausea, vomiting, constipation, polyuria, polydipsia, and lethargy to confusion, cognitive dysfunction, kidney failure, and cardiac arrhythmias. Treatment includes initial aggressive fluid administration, loop diuretics, and bisphosphonate therapy. Bisphosphonates are bone-modifying agents that lower the patient’s calcium level by inhibiting osteoclasts and stabilizing the bone matrix by binding to calcium phosphate. Following initial treatment, identifying and treating the underlying malignancy is critical for controlling the condition and preventing its recurrence (Dickens et al., 2022. Klemencic & Perkins, 2019).
Signs and Symptoms
Less than 20% of ovarian cancers are detected in the early stages. Due to the ovaries’ anatomical location deep within the pelvic cavity, diagnosing the condition in an early stage remains challenging. An ovarian tumor often grows undetected until it becomes large enough to cause symptoms or be detected on a pelvic exam. Most patients with early-stage ovarian cancer are asymptomatic. When symptoms do occur, they are often ambiguous and nonspecific, such as abdominal pain, bloating, pelvic pressure, low back discomfort, constipation, and fatigue. According to the National Ovarian Cancer Coalition [NOCC], symptoms are more common in advanced disease due to ascites and pressure on the bladder or rectum. These processes can lead to persistent abdominal pain and distention, heartburn, early satiety, nausea, weight loss, urinary frequency, menstrual cycle irregularities, and dyspareunia (i.e., pain with intercourse). In some women, a palpable mass may be present. The persistence of symptoms is crucial, and nurses should consider ovarian cancer in the differential diagnosis for any patient reporting the above symptoms for longer than two weeks (NOCC, n.d.; Woo & Long, 2021).
Diagnostic Workup
Adnexa denotes the region adjoining the uterus and includes the ovary, fallopian tube, and associated structures (i.e., blood vessels, ligaments, and connective tissue). Masses in the adnexa may be symptomatic or discovered incidentally. Most are benign, some will regress spontaneously, and others require surgical removal. The differential diagnosis for an adnexal mass includes an ovarian cyst, ectopic pregnancy, abscess, endometrioma, or ovarian tumor (benign or malignant). Transvaginal ultrasound (TVUS) and serum cancer antigen-125 (CA-125) blood tests are often used to evaluate adnexal masses (Woo & Long, 2021).
CA-125
CA-125 is a tumor marker for most ovarian cancers. Tumor markers are substances or proteins produced by cancer or by the body’s response to cancer’s presence, but they are also made in smaller quantities by healthy cells. Tumor markers can be found and measured in the blood or, less commonly, in the urine. While they are considered nonspecific and are not of significant value in isolation, they can be beneficial for establishing a baseline during the diagnostic workup, evaluating response to treatment, and detecting cancer recurrence. Each tumor marker is specific to a different type of disease process (Yarbro et al., 2018).
CA-125 is present on the surface of most ovarian cancer cells and is easily measured by a blood test. However, not all women with ovarian cancer will have elevated CA-125 levels. Healthy tissues and some other cancer types produce small quantities of CA-125. Serum CA-125 is elevated in 80% of women with epithelial ovarian cancer but less than half of women with early disease. CA-125 may be an effective therapeutic tool in select patients. For example, if CA-125 levels are elevated at diagnosis and decline during treatment, the cancer is responding to the treatment. Following the completion of treatment, the CA-125 level may be evaluated periodically to monitor for cancer recurrence (OneCare Media, 2021).
CA-125 levels may be elevated in benign conditions, such as endometriosis, uterine fibroids, pregnancy, infection, pelvic inflammatory disease, colitis, pancreatitis, cirrhosis, pericarditis, and systemic lupus erythematosus. CA-125 levels can also fluctuate with menstrual cycles (Yarbro et al., 2018). According to the American Board of Internal Medicine (2023), the normal CA-125 level is below 35 U/mL. While CA-125 is the most common and sensitive tumor marker for ovarian cancers, other tumor markers such as human chorionic gonadotropin (hCG), lactate dehydrogenase (LDH), or alpha-fetoprotein (AFP) may be useful for less common types of ovarian tumors (Woo & Long, 2021).
Imaging
Ultrasound is a safe, noninvasive imaging modality that uses soundwaves to
generate images of internal body structures. Ultrasound does not use X-rays or ionizing radiation and is ordered for numerous diagnostic and therapeutic indications. Ultrasound images are obtained by placing a transducer (i.e., a small probe) and gel on the skin. The transducer produces sound waves at high frequencies, which exceed the threshold of human hearing. These high-frequency soundwaves travel from the transducer through the gel and into the body, generating images on a computer. Images are captured in real-time, allowing for the evaluation of the structure and movement of the body’s internal organs. TVUS assesses the uterus, fallopian tubes, and ovaries by placing a transducer directly into the vaginal canal, optimizing the image quality by proximity to the ovaries. While TVUS can help identify a mass in or surrounding the ovary, it cannot indicate whether it is malignant or benign (National Institute of Biomedical Imaging and Bioengineering, 2016).
If the TVUS and the CA-125 demonstrate abnormalities, the NCCN (2023b) recommends a computed tomography (CT) scan of the abdomen and pelvis—with contrast media unless contraindicated—to evaluate the extent of the disease and for signs of metastases. CT scans use a series of X-rays and computer technology to create cross-sections of the inside of the body, including bones, blood vessels, organs, and soft tissues. The X-ray beam circles the subject to generate various views of the identified structure, forming a complete picture in much greater detail than a conventional X-ray. CT scans use ionizing radiation to produce images, so patients should be counseled regarding the amount of radiation exposure. For example, a CT scan of the abdomen and pelvis with IV contrast carries a radiation dose of 20 milliSievert (mSv), which equals approximately 7 years of natural background radiation exposure (American College of Radiology, n.d.). CT scans are commonly used in the care of oncologic patients to evaluate their response to treatment and monitor for cancer progression or recurrence. Patients should be advised to lie flat and remain motionless on a table that slides into the scanning machine and resembles a large doughnut. The X-ray tube will rotate around the patient to capture all the necessary images. The test takes about 10 to 15 minutes to complete (International Atomic Energy Agency, n.d.).
Paracentesis
Malignant ascites in the abdominal cavity are a classic feature of advanced ovarian cancer. Ascites can be detected on clinical exams and radiological imaging. It typically causes significant symptoms such as shortness of breath, abdominal distension, heartburn, bloating, nausea, anorexia, and early satiety. A paracentesis is an invasive procedure performed to provide short-term relief from symptoms. A needle is inserted into the abdominal cavity to drain the fluid into a collection bag. The fluid sample undergoes morphological assessment by a pathologist to evaluate for the presence of malignant cells and features of cancer. Sometimes, patients are diagnosed with ovarian cancer based on the cytology findings from a paracentesis (Cerne & Kobal, 2017).
The NCCN (2023b) ovarian cancer guidelines also support the following components of a diagnostic workup:
- magnetic resonance imaging (MRI) scan or positron emission tomography—CT (PET/CT) scan imaging may be indicated for indeterminate lesions if the results will alter the management
- chest CT or chest X-ray as clinically indicated
- complete blood count (CBC), chemistry profile, and liver function test (LFT)
- referral to a gynecologic oncologist (gyn/onc) for clinically suspicious lesions
- genetic risk evaluation, including germline testing, for all patients with ovarian cancer, fallopian tube cancer, or PPC
Ovarian Cancer Staging
The cancer stage at diagnosis guides treatment options and strongly influences overall survival. There are detailed staging systems for ovarian cancer: the International Federation of Gynecology and Obstetrics (FIGO) system and the American Joint Committee on Cancer’s (AJCC) Tumor, Node, Metastasis (TNM) staging system, 8th edition. Both systems are essentially the same and describe specific characteristics to assign stages I through IV, as outlined in Table 3 and demonstrated in Figure 2. Since ovarian cancer is a surgically staged disease, the FIGO system has been adopted universally, and all patients with suspected ovarian malignancies should be promptly referred to a gyn/onc for surgical evaluation (AJCC, n.d., 2018; Berek et al., 2021; Bhatla & Denny, 2018; FIGO, 2014; NCCN, 2023b; Olawaiye, n.d.).
Figure 2
Stages of Ovarian Cancer
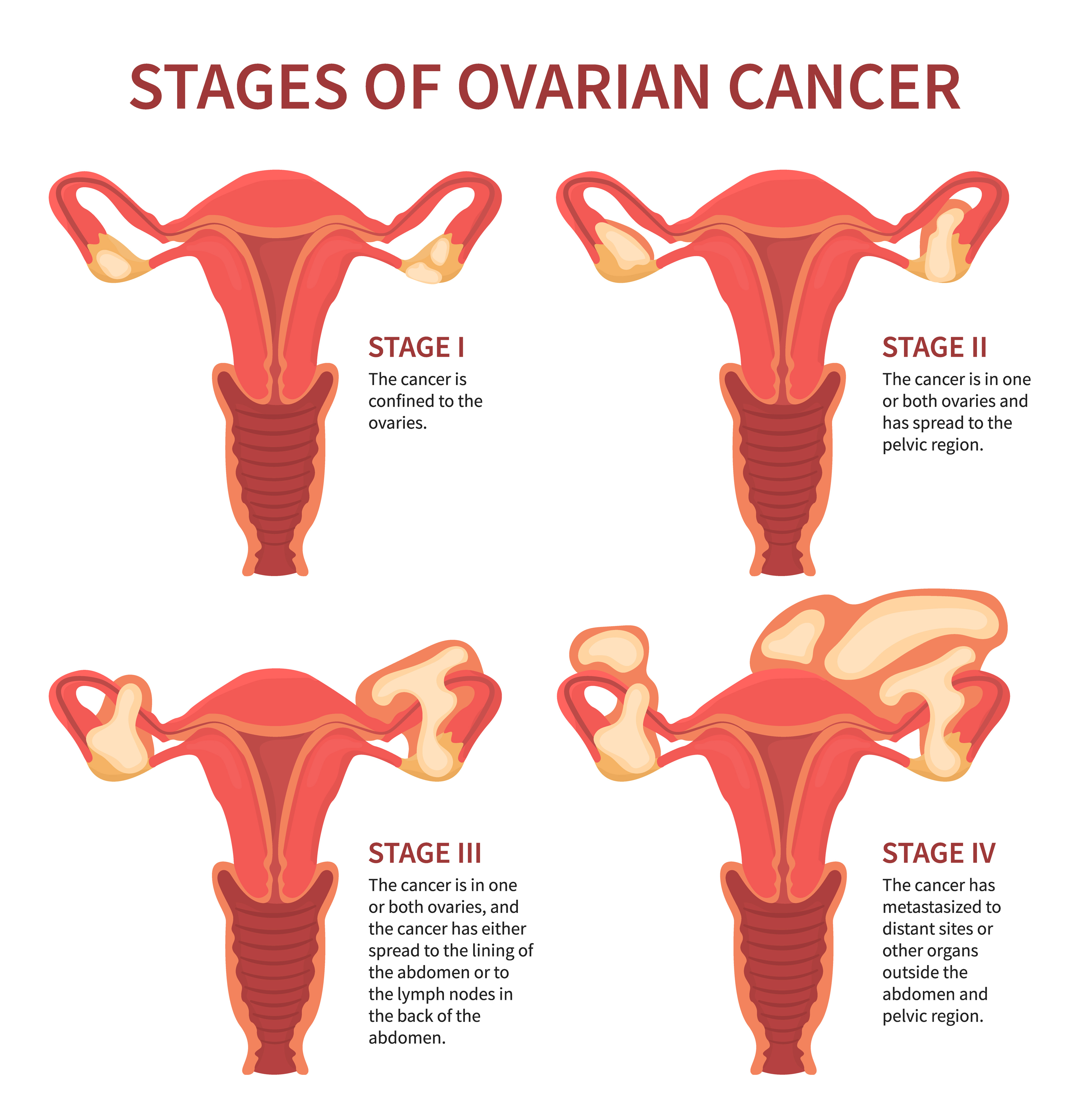
(iStock illustration ID: 1249232483)
Cancer staging reflects the cell type, tumor grade, anatomical location of the tumor, and extent of malignancy. Within the TNM staging system, T denotes the size of the tumor and whether it has grown into nearby tissue, N refers to the presence of cancer in the lymph nodes, and M indicates if cancer has metastasized to other parts of the body beyond the origin site. Tumor grade measures how different the cancer cells look compared to healthy cells under the microscope. It is based on cell differentiation and varies from low grade (i.e., grade 1) to high grade (i.e., grade 3). Grade 1 is well-differentiated and appears similar to healthy cells, whereas grade 3 is poorly differentiated (i.e., does not resemble healthy cells) and most aggressive (AJCC, n.d., 2018; FIGO, 2014; Olawaiye, n.d.; Yarbro et al., 2018).
Table 3
Cancer Staging
AJCC TNM | FIGO Stage | Description |
TX | - | primary tumor cannot be assessed |
T0 | - | no evidence of primary tumor |
T1 | I | tumor limited to 1 or both ovaries or fallopian tube(s) |
T1a | IA | tumor limited to 1 ovary (capsule intact) or fallopian tube; no tumor on ovarian or fallopian tube surface; no malignant cells in ascites or peritoneal washings |
T1b | IB | tumor limited to both ovaries (capsules intact) or fallopian tubes; no tumor on ovarian or fallopian tube surface; no malignant cells in ascites or peritoneal washings |
T1c
T1c1 T1c2 T1c3 | IC | tumor limited to 1 or both ovaries or fallopian tubes, with any of the following:
|
T2 | II | tumor limited to the pelvis; involves one or both ovaries or fallopian tubes with pelvic extension below the pelvic brim or PPC |
T2a | IIA | extension and/or implants on the uterus and/or fallopian tube(s) and/or ovaries |
T2b | IIB | extension to and/or implants on other pelvic tissues |
T3 | III | tumor involves one or both ovaries or fallopian tubes, or PPC, with microscopically confirmed peritoneal metastasis outside the pelvis and/or metastasis to the retroperitoneal (pelvic and/or para-aortic) lymph nodes |
T3a | IIIA2 | microscopic extrapelvic (above the pelvic brim) peritoneal involvement with or without positive retroperitoneal lymph nodes |
T3b | IIIB | macroscopic peritoneal metastasis beyond the pelvis that is ≤2 cm in greatest dimension with or without metastasis to the retroperitoneal lymph nodes |
T3c | IIIC | macroscopic peritoneal metastasis beyond the pelvis that is >2 cm in the greatest dimension with or without metastasis to the retroperitoneal lymph nodes (includes an extension of the tumor to the capsule of the liver and spleen without parenchymal involvement of either organ) |
M1 | IV | distant metastasis, including pleural effusion with positive cytology; liver or splenic parenchymal metastasis; metastasis to extra-abdominal organs (including inguinal lymph nodes and lymph nodes outside the abdominal cavity); and transmural involvement of intestine |
| IVA | pleural effusion with positive cytology |
| IVB | liver or splenic parenchymal metastases; metastases to extra-abdominal organs (including the inguinal lymph nodes and lymph nodes outside the abdominal cavity); transmural involvement of the intestine |
(AJCC, n.d., 2018; Berek et al., 2021; Bhatla & Denny, 2018; FIGO, 2014; Olawaiye, n.d.)
Cytoreduction
Research data demonstrate that primary assessment and cytoreduction (i.e., surgical debulking) by a gyn/onc leads to a survival advantage. Cytoreduction is a surgical procedure to remove, eliminate, or reduce the size of cancerous tumors and should be performed by a trained gyn/onc. Some tumors may be removed entirely, whereas others may be reduced in size. Unlike other cancers, which are usually biopsied before surgery, ovarian cancers are typically staged during surgery. The surgical intervention provides tissue sampling for a conclusive diagnosis and allows the surgeon to determine the extent of the disease while performing the cytoreductive procedure. Cytoreduction improves the effectiveness of subsequent chemotherapy and radiation; it is vital for women whose ovarian cancer has spread significantly throughout the abdomen at diagnosis. Optimal cytoreduction is defined as no residual cancer greater than 1 cm remaining after surgery. Studies have demonstrated survival is highest among women with no or only a small volume of residual disease, defined as less than 0.5 cm (NCCN, 2023b; Woo & Long, 2021).
Treatment Modalities
Treatment for ovarian cancer is usually multifactorial, involving combined modalities, and primarily depends on the cancer stage. The NCCN (2023b) provides evidence-based treatment guidelines for ovarian cancer according to histopathological findings, genetics, staging, inheritance patterns, and other specific features through rigorous clinical trial research, data compiled across institutions, and annual expert panel reviews. These guidelines are widely utilized in cancer care and guide medical decision-making throughout the patient’s disease trajectory. Due to the aggressive biology of ovarian cancer, treatment is chronic, and most patients experience periods of remission and relapse. Many require maintenance therapy; all patients need close monitoring and surveillance after treatment completion to monitor for recurrence. This section will overview the most common treatment strategies (NCCN, 2023b).
Surgery
Surgery is the mainstay treatment for most ovarian cancers. When ovarian cancer is diagnosed early, standard therapy includes surgical staging, cytoreduction, hysterectomy, and bilateral salpingo-oophorectomy (BSO). The procedure may or may not involve an omentectomy (i.e., removal of all or part of the omentum) and lymphadenectomy (i.e., removal of at least one lymph node to evaluate for cancer). Since most patients present with advanced disease at diagnosis, surgery is often followed by adjuvant (post-surgical) systemic therapy (NCCN, 2023b).
Chemotherapy
Chemotherapy, cytotoxic or antineoplastic therapy, refers to high-risk, hazardous medications that destroy cancer cells throughout the body. Chemotherapy interferes with the normal cell cycle, impairing DNA synthesis and cell replication to prevent cancer cells from dividing and multiplying. Most chemotherapy agents attack broadly (i.e., they kill normal, healthy cells in the body together with the cancer cells). Chemotherapy is classified according to biochemical activity, mechanism of action, and phase of action during the cell cycle. Chemotherapy agents are divided into two categories: cell cycle-specific and cell cycle-nonspecific. Cell cycle-specific drugs exert cytotoxic effects on cells actively dividing at specific stages within the cell cycle. These drugs do not act against cancer cells during the resting phase (G0) and are schedule-dependent, so they are most effective when administered in divided doses or by continuous infusion. Cell cycle-nonspecific drugs have a broader impact on cancer cells, as they affect cells at any phase in the cell cycle, including G0. These agents are considered dose-dependent and are most effective when administered by bolus doses, as the number of cells affected is directly proportional to the amount of drug given (Olsen et al., 2023).
Chemotherapy is prominent in treating ovarian cancer and can be used at various time points during the disease trajectory. Neoadjuvant chemotherapy intends to reduce the tumor burden in preparation for surgery. Adjuvant chemotherapy (i.e., chemotherapy after surgery) aims to decrease cancer recurrence risk, reduce micro-metastases, and eradicate any remaining cancer cells. Palliative chemotherapy treats recurrent cancer, seeking to relieve or delay cancer symptoms, enhance comfort, reduce symptom burden, and improve quality of life. IV platinum-based chemotherapy agents (i.e., carboplatin [Paraplatin] or cisplatin [Platinol]) are highly effective against ovarian cancer, making them the first-line treatment unless contraindicated. They are typically combined with a microtubule agent such as paclitaxel (Taxol) or docetaxel (Taxotere). Some of the most common chemotherapy agents for ovarian cancer include the following:
- Alkylating agents
- disrupt DNA, prevent DNA replication, and are cell cycle-nonspecific
- carboplatin (Paraplatin)
- cisplatin (Platinol)
- ifosfamide (Ifex)
- cyclophosphamide (Cytoxan)
- disrupt DNA, prevent DNA replication, and are cell cycle-nonspecific
- Microtubule agents
- inhibit cell division and are cell cycle-specific (i.e., acting on the M-phase)
- paclitaxel (Taxol)
- docetaxel (Taxotere)
- inhibit cell division and are cell cycle-specific (i.e., acting on the M-phase)
- Antitumor antibiotics
- bind to and unwind the DNA helix and are cell cycle-specific (i.e., acting on the S-phase)
- doxorubicin (Adriamycin)
- liposomal doxorubicin (Doxil; NCCN, 2023b; Nettina, 2019; Olsen et al., 2023; Yarbro et al., 2018)
- bind to and unwind the DNA helix and are cell cycle-specific (i.e., acting on the S-phase)
Chemotherapy Side Effects
The side effects of chemotherapy vary based on the drug type, dosage, duration of treatment, and specific patient factors. Since cancer cells divide rapidly, chemotherapy impacts healthy and cancerous cells that divide quickly (e.g., in the GI tract, skin/hair cells, and bone marrow). As a group, the most common side effects include bone marrow suppression (i.e., anemia, thrombocytopenia, neutropenia), fatigue, nausea, anorexia, alopecia (i.e., hair loss), diarrhea, skin changes, and peripheral neuropathy (i.e., damage to the sensory nerves). Alopecia can cause significant emotional distress for women. Chemotherapy-induced hair loss starts about 7 to 15 days following the first chemotherapy dose. It results from damage to the dividing hair matrix cells, which causes the hair shaft to break at the follicular orifice or hair bulb. While the degree of hair loss depends on the chemotherapy agent, the dose, and the administration schedule, paclitaxel (Taxol) and docetaxel (Taxotere) are well-known for inducing alopecia. Nurses should reassure patients that their hair will begin to regrow within a few weeks following the cessation of chemotherapy, as permanent alopecia following chemotherapy is extremely rare (Olsen et al., 2023).
Chemotherapy-induced peripheral neuropathy (CIPN) is a common side effect of carboplatin (Paraplatin), cisplatin (Platinol), paclitaxel (Taxol), and docetaxel (Taxotere). It is often the dose-limiting toxicity (DLT) of these agents. DLTs are severe toxicities and side effects serious enough to warrant a reduction in the dose or discontinuation of the treatment. CIPN results from the demyelination of sensory and motor axons. Patients experience reduced nerve conduction velocity, leading to the loss of deep tendon reflexes and paresthesia (numbness and tingling), weakness, and burning pain. Initially, CIPN often affects the most distal points of the extremities, such as the fingertips and toes, and progresses proximally toward the midline. In severe cases, patients may lose complete sensation in the fingers, hands, toes, and feet; this can cause significant disability, such as the inability to grasp or hold items, and gait disturbance, including imbalance and falls. CIPN is complex as no single pathophysiologic process explains the neuropathies that occur following exposure to certain chemotherapies. CIPN is dose-dependent and progressive while a patient receives treatment but can also have a cascading effect after treatment ends. During this cascading phenomenon, symptoms become more prominent after discontinuing the offending agent. Pain, sensory changes, and weakness that manifest during treatment generally lead to chemotherapy dose reductions, changes in treatment protocols, or termination of the therapeutic agent entirely. CIPN is associated with significant morbidity as it negatively impacts quality of life and impairs activities of daily living (Brown et al., 2019).
Currently, no medications or supplements are effective in preventing CIPN. Regular exercise, reducing alcohol intake, and treating preexisting medical conditions (e.g., diabetic neuropathy and vitamin B12 deficiency) may reduce the risk of CIPN. Management of CIPN is complex, and effective treatment options are limited. Pharmacologic treatment focuses on symptom relief, although many agents are not highly effective. Over-the-counter pain medications, menthol creams, capsaicin creams, or lidocaine patches may offer comfort. Some patients may be prescribed medications such as gabapentin (Neurontin), an anticonvulsant/anti-epileptic agent that also treats neuropathic pain. Some patients may find relief from selective serotonin-norepinephrine reuptake inhibitors (SNRIs) such as duloxetine (Cymbalta). Patients with CIPN must be counseled on ways to avoid injury (e.g., wearing supportive shoes) and promote home safety (e.g., using handrails on stairs and removing throw rugs). Patients must also be mindful of water temperatures due to decreased sensitivity to hot water, increasing their risk for burns when bathing or washing dishes. Improvement in function and resolution of symptoms often occurs gradually over time, but nerve damage may be permanent (Brown et al., 2019).
Hypersensitivity Reactions to Chemotherapy
A hypersensitivity reaction (HSR) happens when the immune system becomes overstimulated by a foreign substance and creates antibodies, provoking an immune response. HSRs are commonly associated with several chemotherapy agents that are used widely in ovarian cancer treatment, such as paclitaxel (Taxol), docetaxel (Taxotere), and carboplatin (Paraplatin). HSR risk can be reduced by premedicating patients with corticosteroids, antihistamines, and/or acetaminophen (Tylenol). HSRs can occur during the initial chemotherapy infusion or after subsequent administrations of the same agent. Paclitaxel (Taxol) is well-known for its risk of a nearly immediate, acute HSR, whereas carboplatin (Paraplatin) more commonly induces an HSR after several cycles. Most HSRs occur during the first 15 minutes of the infusion. Initial signs and symptoms can include hives, urticaria, pruritis, swelling, back pain, facial flushing, rhinitis, abdominal cramping, chills, hypotension, and anxiety. Patients may require supplemental oxygen, fluid resuscitation, or other emergency medications as indicated. For life-threatening symptoms like bronchospasm, angioedema (i.e., swelling of the oral cavity, lips, or tongue), or anaphylaxis, epinephrine 0.1-0.5 mg (1:10,000 solution for adult patients) may be required (Nettina, 2019; Olsen et al., 2023).
Intraperitoneal (IP) Chemotherapy
Chemotherapy for ovarian cancer may also be administered through the IP route. While IV chemotherapy is the standard of care for most patients with ovarian cancer, a select subset of patients may benefit from IP chemotherapy. IP chemotherapy is given through an implanted catheter and access port inserted into the peritoneal space. The medication is instilled directly into the port to treat cancer within the peritoneal cavity. Several studies have demonstrated that the frequency and severity of toxic side effects are significantly higher in patients receiving IP chemotherapy. Common side effects include abdominal pressure, bloating, urinary frequency, nausea, vomiting, and anorexia. IP chemotherapy carries a risk of peritonitis (i.e., inflammation of the peritoneum), which can be life-threatening and lead to sepsis. IP chemotherapy is associated with prolonged bone marrow suppression and higher renal toxicity than IV chemotherapy. IP chemotherapy is not widely used, primarily due to its higher toxicity profile and lack of survival benefit over IV chemotherapy (Eoh et al., 2017). However, the NCCN (2023b) lists IP chemotherapy as an option for patients with optimally cytoreduced stage II to III disease. Based on expert consensus across various evidence-based guidelines, IP therapy is appropriate for patients with no or minimal residual disease following surgical cytoreduction (Eoh et al., 2017).
Targeted Therapy
Targeted therapies refer to novel treatment modalities that attack specific parts of cancer cells to prevent tumor development or shrink existing tumors. Numerous proteins are located on the cellular membranes; these growth factor receptors connect the external and internal cellular environments and are essential for cell growth and development. Targeted agents can block or turn off chemical signals that tell cancer cells to grow and divide. They can also starve the tumor by cutting off the blood supply and preventing the formation of new blood vessels or by carrying toxins or poison directly to the cancer cells, thereby killing them without harming healthy cells (Sengupta, 2017). Most ovarian cancer research throughout the last decade has focused on developing specialized drugs that block growth factor receptors and identifying new targets. These drugs have revolutionized treatment options for patients with advanced and metastatic ovarian cancer by fighting against drug resistance and increasing survival. The next section provides an overview of the most common types of targeted agents that treat ovarian cancer (Masoud & Pages, 2017; NCCN, 2023b).
Anti-Angiogenesis Agents
Vascular endothelial growth factor (VEGF) is a signaling protein that stimulates angiogenesis (i.e., new blood vessel formation) in healthy and cancerous cells. Blood vessels carry oxygen and nutrients to the tissue for growth and survival. Tumors need blood vessels to grow and spread. Anti-angiogenesis agents (i.e., VEGF inhibitors) cut off blood supply to cancer cells by interfering with the VEGF receptor; tumors stay small and eventually starve (Olsen et al., 2023). Bevacizumab (Avastin) is a humanized monoclonal antibody that binds to and inhibits the activity of human VEGF to its receptors. It has tremendously impacted the management of ovarian cancer. Ferriss and colleagues (2015) found that women with malignant ascites who received bevacizumab (Avastin) had significantly improved PFS and overall survival compared to those who received chemotherapy alone. The NCCN (2023b) added bevacizumab (Avastin) to their upfront chemotherapy guideline with carboplatin (Paraplatin) and paclitaxel (Taxol) as a category 2B recommendation, reflecting the consensus that the intervention is appropriate.
The side effects of bevacizumab (Avastin) include bleeding, headaches, hypertension, and proteinuria (i.e., protein spilling into urine due to increased pressure within the kidneys). Many patients require concurrent treatment with antihypertensives due to the medication’s elevation of blood pressure levels (Olsen et al., 2023). VEGF inhibitors are contraindicated within 28 days of major surgery (preoperatively or postoperatively) due to the increased risk of major bleeding events, delayed wound healing, and fistula development (i.e., an abnormal connection between hollow spaces within the body). VEGF inhibitors have a black-box warning for bowel perforation (i.e., a hole in the intestines). Patients should be counseled to report the sudden onset of severe and diffuse abdominal pain, bloating, nausea, vomiting, or rectal bleeding (Olsen et al., 2023).
Poly ADP-Ribose Polymerase (PARP) Inhibitors
The PARP enzyme serves a critical role in cell growth, cell regulation, and the repair of healthy cells and cancer cells. It fixes DNA damage in cancer cells, helping them repair themselves and survive (Olsen et al., 2023). PARP inhibitors interfere with the PARP enzyme, preventing cancer cells with a BRCA1/2 mutation from repairing DNA damage and inducing cell death. PARP inhibitors have transformed the treatment of BRCA–mutant cancers (Ring & Modesitt, 2018). Olaparib (Lynparza), rucaparib (Rubraca), and niraparib (Zejula) were initially approved to treat BRCA1/2-mutant ovarian cancer. Due to emerging research demonstrating the effectiveness of oral PARP inhibitors in patients without a BRCA1/2 mutation, the American Society of Clinical Oncology (ASCO) released a new recommendation: all women with epithelial ovarian cancer without a germline BRCA1/2 mutation should undergo somatic tumor testing for BRCA1/2 (i.e., the tumor itself should be tested; Konstantinopoulos et al., 2019).
Olaparib (Lynparza) is administered at 300 mg PO twice daily, rucaparib (Rubraca) is administered at 600 mg PO twice daily, and niraparib (Zejula) is given at 300 mg PO once daily. The most common side effects of PARP inhibitors include anemia, neutropenia, fatigue, nausea, diarrhea or constipation, anorexia, and arthralgias. They are also associated with a rare risk (under 1.5%) of myelodysplastic syndrome (MDS, a bone marrow failure disorder) or acute myeloid leukemia (AML, a type of blood cancer). They carry a slight risk (under 1%) of pneumonitis and embryo-fetal toxicity. Niraparib (Zejula) has demonstrated more significant hematologic toxicities, including anemia and thrombocytopenia requiring transfusion support. PARP inhibitors have several considerable drug interactions, particularly antifungal medications and certain antibiotics. Patients should alert their clinician before starting any new medications. Patients should avoid grapefruit and Seville oranges, as they can increase PARP inhibitors’ effects and toxicity (NCCN, 2023b; Olsen et al., 2023).
Immunotherapy
Immunotherapy stimulates the immune system to recognize and destroy cancer cells. Immunotherapy can produce antitumor effects by modifying the body’s natural host defense mechanisms, making them more sensitive to cancer cells. Immune-based treatments work differently than chemotherapy via highly specialized activity. Immune checkpoint inhibitors block the receptors that cancer cells use to inactivate immune cells (specifically T-cells). When this signal is blocked, T-cells can better differentiate between healthy and cancer cells, thereby augmenting the cancer cells’ immune response. Checkpoint inhibitors occur in two categories: programmed cell death-1 (PD-1)/PD-ligand 1 (PD-L1) inhibitors and cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) inhibitors. PD-1 is a transmembrane checkpoint protein expressed on the surface of circulating immune cells. PD-1 usually acts as an “off switch” to keep the immune cells from attacking other cells in the body. When PD-1 binds to PD-L1, it signals the T-cell to leave the neighboring cells alone. Some cancer cells have large amounts of PD-L1, which helps them evade immune attacks. PD-1 and PD-L1 inhibitors, monoclonal antibodies that target PD-1 or PD-L1, prevent this complex formation and boost the immune response against cancer cells. Pembrolizumab (Keytruda) is a humanized monoclonal antibody that binds to PD-1, preventing its interaction with PD-L1 and PD-L2 (Sasikumar & Ramachandra, 2018).
Immunotherapy's role in treating ovarian cancers is less advanced than other cancers, and clinical research is ongoing. Microsatellite instability (MSI) occurs in about 20% of endometrioid ovarian cancers. Therefore, microsatellite testing should be performed in all newly diagnosed endometrioid ovarian cancers (Fraune et al., 2020). ASCO recommends testing all women with clear cell, endometrioid, or mucinous ovarian cancer for mismatch repair deficiency (dMMR). MMR deficiency can be reported as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), but these groups have the same meaning (Konstantinopoulos et al., 2019). Initially, pembrolizumab (Keytruda) was the only agent used for metastatic ovarian tumors in this setting. It is approved by the US Food & Drug Administration (FDA) for MSI-H or MMR-deficient (dMMR) ovarian tumors and unresectable or metastatic tumors with tumor mutational burden-high (TMB-H) status (i.e., ≥10 mutations/megabase (mut/Mb) who have progressed following prior treatment and who have no satisfactory alternative treatment options (Merck & Co., 2023). More recently, dostarlimab-gxly (Jemperli) was also approved for dMMR/MSI-H recurrent or advanced tumors and is similarly listed in the NCCN (2023b) guidelines alongside pembrolizumab (Keytruda) as an immunotherapy option for these patients (GlaxoSmithKline [GSK], 2023).
Pembrolizumab (Keytruda) and dostarlimab-gxly (Jemperli) have comparable side effect profiles. They are generally well-tolerated. Common side effects include fatigue, nausea, anorexia, coughing, diarrhea, skin rash, and itching. The most common adverse reactions (ARs) of pembrolizumab (Keytruda) include fatigue, musculoskeletal pain, rash, diarrhea, pyrexia, cough, decreased appetite, pruritus, dyspnea, constipation, pain, abdominal pain, nausea, and hypothyroidism (Merck & Co., 2023). The most common ARs with dostarlimab-gxly (Jemperli) include fatigue/asthenia, anemia, diarrhea, and nausea (GSK, 2023). However, both agents carry a risk for severe and possibly fatal autoimmune-related ARs. Although any organ system can be affected, the most commonly observed reactions consist of colitis, hepatitis, endocrinopathies (i.e., thyroid and adrenal dysfunction), pneumonitis, and skin-rash progression to Stevens-Johnson syndrome (Sasikumar & Ramachandra, 2018; Sengupta, 2017).
Antibody Drug Conjugates (ADCs)
ADCs are an emerging class of targeted agents that deliver chemotherapy directly to cancer cells via a linker attached to a monoclonal antibody (i.e., a protein). Once the linker binds to a specific target on the cancer cell, it releases a powerful cytotoxic drug into the cancer cell. ADCs aim to improve efficacy while minimizing systemic toxicity to normal tissue using a targeted delivery mechanism (Pettinato, 2021). Mirvetuximab soravtansine (Elahere) is an ADC targeting folate receptor alpha (FRa). FRa is a tumor-associated antigen overexpressed in over 80% of ovarian carcinomas and is associated with more poorly differentiated, aggressive tumors. FRa binds to folic acid, and its derivatives mediate cellular processes, including cell division, proliferation, and tumor growth. Since FRa is highly expressed in ovarian cancer and is readily measurable, it has become a novel therapeutic target for ovarian cancer patients (Birrer et al., 2019; Mai et al., 2023). In the phase III MIRASOL trial, Mirvetuximab soravtansine (Elahere) showed a 35% reduction in the risk of disease progression or death compared to chemotherapy in patients with FRa-high platinum-resistant ovarian cancer (Moore et al., 2023). Mirvetuximab soravtansine (Elahere) was granted accelerated approval by the FDA in November 2022. It is the first biomarker-directed treatment to demonstrate an overall survival benefit in patients with platinum-resistant ovarian cancer (FDA, 2023).
Mirvetuximab soravtansine (Elahere) is administered as an IV infusion every three weeks. Before each dose, patients must be premedicated with the following medications to reduce the risk and severity of infusion reactions.
- corticosteroid (dexamethasone [Decadron])
- antihistamine (diphenhydramine [Benadryl])
- acetaminophen (Tylenol)
- antiemetic (to prevent nausea and vomiting)
The most common ARs include ocular toxicity (i.e., vision impairment, dry eye, keratopathy), fatigue, nausea, increased liver function enzymes,
abdominal pain, peripheral neuropathy, diarrhea, constipation, hypomagnesemia, and bone marrow suppression (i.e., decreased leukocytes, neutrophils, and hemoglobin). Due to the risk for ocular toxicity, patients must undergo an ophthalmologic examination before starting treatment, before every other dose, and promptly if eye problems arise. Treatment may need to be withheld or permanently discontinued based on the severity and potential improvement of the eye problems. Further, starting the day before each mirvetuximab soravtansine (Elahere) infusion, on the day of the infusion, and for three days after that, patients should administer eye drops containing corticosteroids into each eye six times daily. On days 5 to 8, drops should be administered four times daily. Patients should also use preservative-free lubricant eye drops at least four times a day and avoid using contact lenses during treatment with mirvetuximab soravtansine (Elahere). The lubricant eye drops should be administered at least ten minutes after administering the corticosteroid eye drops. While mirvetuximab soravtansine (Elahere) has demonstrated a therapeutic breakthrough for patients with platinum-resistant ovarian cancer, they require a great deal of support, monitoring, and education to prevent, manage, and immediately treat ocular toxicities (ImmunoGen, 2022; Moore et al., 2023).
Hormonal Therapy
Hormonal treatments block certain hormones from reaching cancer cells or prevent the body from producing these hormones (Nettina, 2019). Estrogen and estrogen receptors drive many types of ovarian cancers, making them amenable to hormone-blocking treatments to shrink or slow their growth. Hormonal therapy is also prescribed as maintenance therapy to reduce the risk of ovarian cancer recurrence. Currently, there are four types of hormonal therapy used in ovarian cancer treatment: selective estrogen receptor modulators (SERM), selective estrogen receptor down-regulators (SERD), luteinizing hormone-releasing hormones (LHRH), and aromatase inhibitors (AI). These agents are outlined in Table 4 (Nettina, 2019; NCCN, 2023b).
Table 4
Hormonal Treatments for Ovarian Cancer
Drug & Dosing | Mechanism | Warnings & Precautions |
SERM Tamoxifen (Soltamax) 20 mg po daily |
|
|
LHRH Leuprolide acetate (Lupron) 7.5 mg IM injection once monthly |
|
|
AI Anastrozole (Arimidex) 1 mg po daily Letrozole (Femara) 2.5 mg po daily Exemestane (Aromasin) 25 mg po daily |
|
|
(Nettina, 2019; Olsen et al., 2023)
The most common adverse effects of hormone treatment depend on the type of drug. They generally include hot flashes, night sweats, loss of libido, weight gain, joint aches or pains, mood changes, thinning or weakening of the bones (i.e., osteopenia or osteoporosis), and atrophic vaginitis (i.e., atrophy, loss of elasticity, dryness, and resulting irritation of the vaginal mucosa). Due to the impact of hormonal therapy on bone thinning, patients should be counseled on the importance of a calcium-rich diet with at least 1,200 mg of calcium daily. Nurses should counsel patients on the importance of weight-bearing exercises to preserve bone health. Exercise can also help reduce the severity of joint aches and pains associated with these medications (Olsen et al., 2023).
For a more detailed review of the principles of chemotherapy, immunotherapy, and specific oncology nursing implications, refer to the following NursingCE courses:
- Oncology Nursing CE Course Part 1
- Oncology Nursing CE Course Part 2
- Oncology Medication Administration Nursing CE Course for RNs and LPNs
- Oncology Medication Management Nursing CE Course for APRNs
Screening and Early Detection
Extensive research has focused on identifying screening tests for ovarian cancer without success. The need for a reliable method for the early detection of ovarian cancer among asymptomatic women remains a prominent issue and the subject of ongoing research. TVUS and CA-125 are not sensitive and specific enough to detect ovarian cancer at early, curable stages. Neither test has been shown to reduce morbidity or mortality associated with the disease. Ovarian cancer screening recommendations include women at average risk and those at high risk (Yarbro et al., 2018).
Average-Risk Women
The US Preventive Services Task Force (USPSTF, 2018), ACS (2020b), and Society of Gynecologic Oncology (SGO, 2017) all recommend against screening for ovarian cancer in asymptomatic women without a known high-risk hereditary cancer syndrome. This recommendation is based on a higher risk-to-benefit ratio; harms associated with screening include false-positive results leading to unnecessary diagnostic testing, psychological damage, and surgical interventions in women who do not have cancer. The ACS (2020b) recommends that all women undergo routine health maintenance with an annual physical exam, including a pelvic examination with a bimanual rectovaginal exam. The USPSTF (2018) contrasts this recommendation with the following statement: “Current evidence is insufficient to assess the balance of benefits and harms of screening with a pelvic examination to detect a range of gynecologic conditions in asymptomatic, nonpregnant women” (USPSTF, 2018, p. 590).
High-Risk Women
Ovarian cancer screening guidelines for high-risk women are more complex than those for average-risk women. The USPSTF (2018) classifies high-risk women as those with specific hereditary cancer syndromes or those with a strong family history of ovarian or breast cancer who may be at risk for a hereditary cancer syndrome. The USPSTF does not make specific recommendations regarding managing and screening women in this high-risk category. The ACS (2020b), USPSTF (2018), SGO (2017), and the American College of Obstetricians and Gynecologists (ACOG, 2017) collectively endorse that all women with a family history that is suspicious of an inherited BRCA1/2 gene mutation should be referred to a genetic counselor. Women with a family history of ovarian cancer who test negative for inherited gene mutations may still be at increased risk relative to the general population (Peshkin & Isaacs, 2023).
Although it remains inconclusive whether screening leads to a decreased number of deaths in women with inherited risk, the NCCN (2023a) endorses the use of TVUS combined with serum CA-125 as screening modalities starting at 30 to 35 years in women who have not undergone RRSO. They do not provide further instructions regarding the frequency of these screening tests (NCCN, 2023a). Patients should be counseled on the limited effectiveness of these tests and potential harmful screening outcomes (Peshkin & Isaacs, 2023). The ACOG (2017) recommends that all women with BRCA1/2 mutations or those with another actionable deleterious mutation predisposing them to ovarian cancer should be counseled on risk-reduction strategies, particularly RRSO (FORCE, 2023). The ACOG (2017) and NCCN (2023a) recommend RRSO between 35 and 40 years for BRCA1 carriers (or whenever childbearing is complete). Women with BRCA1 mutations tend to be diagnosed with ovarian cancer approximately a decade younger than those without an identifiable genetic mutation. Women with BRCA2 may consider delaying RRSO until 40 and 45 years due to the later onset of ovarian cancer. Salpingectomy alone remains under investigation as a risk-reduction method and is not the standard of care (NCCN, 2023a). The ACOG (2017) and NCCN (2023a) also recommend that women with BRCA1/2 mutations or those who carry another actionable deleterious mutation that predisposes to breast cancer should be offered risk-reducing bilateral mastectomy due to their heightened risk of breast cancer (Temkin et al., 2018).
References
American Board of Internal Medicine. (2023). ABIM laboratory test reference ranges – July 2023. https://www.abim.org/Media/bfijryql/laboratory-reference-ranges.pdf
American Cancer Society. (2023a). Key statistics for ovarian cancer. https://www.cancer.org/cancer/ovarian-cancer/about/key-statistics.html
American Cancer Society. (2023b). Ovarian cancer. https://www.cancer.org/cancer/ovarian-cancer.html
American College of Obstetricians and Gynecologists. (2017). Clinical management guidelines for obstetricians-gynecologists: Hereditary breast and ovarian cancer syndrome. Obstetrics & Gynecology, 130(3), e110-e126. https://www.sgo.org/wp-content/uploads/2012/09/PB-182.pdf
American College of Radiology. (n.d.). Radiation dose to adults from common imaging examinations. Retrieved September 17, 2023, from https://www.acr.org/-/media/ACR/Files/Radiology-Safety/Radiation-Safety/Dose-Reference-Card.pdf
American Joint Committee on Cancer. (n.d.). Physician education / Cancer staging system webinars. Retrieved September 17, 2023, from https://www.facs.org/quality-programs/cancer-programs/american-joint-committee-on-cancer/staging-education/physician/
American Joint Committee on Cancer. (2018). AJCC 8th edition staging. https://www.facs.org/media/2zvlrdmi/ajcc_staging_rules_8th_ed.pdf
Babic, A., Sasamoto, N., Rosner, B. A., Tworoger, S. S., Jordan, S. J., Risch, H. A., Harris, H. R., Rossing, M. A., Doherty, J. A., Fortner, R. T., Chang-Claude, J., Goodman, M. T., Thompson, P. J., Moysich, K. B., Ness, R. B., Kjaer, S. K., Jenson, A., Schildkraut, J. M., Titus, L., . . . & Cramer, D. W. (2020). Association between breastfeeding and ovarian cancer risk. JAMA Oncol, 6(6), e200421. https://doi.org/10.1001/jamaoncol.2020.0421
Berek, J. S., Renz, M., Kehoe, S., Kumar, L., & Friedlander, M. (2021). Cancer of the ovary, fallopian tube, and peritoneum: 2021 update. International Journal of Gynecology & Obstetrics, 155(S1), 61-68. https://doi.org/10.1002/ijgo.13878
Bhatla, N., & Denny, L. (2018). FIGO cancer report 2018. International Journal of Gynecology & Obstetrics, 143(S2), 2-3. https://doi.org/10.1002/ijgo.12608
Birrer, M. J., Betella, I., Martin, L. P., & Moore, K. N. (2019). Is targeting the folate receptor in ovarian cancer coming of age? The Oncologist, 24(4), 425-429. https://doi.org/10.1634/theoncologist.2018-0459
Brown, T. J., Sedhom, R., & Gupta, A. (2019). Chemotherapy-induced peripheral neuropathy. JAMA Oncology, 5(5),750. https://doi.org/10.1001/jamaoncol.2018.6771
Centers for Disease Control and Prevention. (2023a). BRCA gene mutations. https://www.cdc.gov/cancer/breast/young_women/bringyourbrave/hereditary_breast_cancer/brca_gene_mutations.htm
Centers for Disease Control and Prevention. (2023b). Ovarian cancer. https://www.cdc.gov/cancer/ovarian/index.htm
Centers for Disease Control and Prevention. (2023c). The BRCA1 and BRCA2 genes. https://www.cdc.gov/genomics/disease/breast_ovarian_cancer/genes_hboc.htm
Cerne, K., & Kobal, B. (2017). Ascites in advanced ovarian cancer. https://www.intechopen.com/books/ovarian-cancer-from-pathogenesis-to-treatment/ascites-in-advanced-ovarian-cancer
Dickens, L. T., Derman, B., & Alexander, J. T. (2022). Endocrine Society hypercalcemia of malignancy guidelines. JAMA Oncology Clinical Guidelines Synopsis, 9(3), 430-431. https://doi.org/10.1001/jamaoncol.2022.7941
Eoh, K. J., Lee, J., Nan, E. J., Kim, S., Kim, Y. T., & Kim, S. W. (2017). Long-term survival analysis of intraperitoneal versus intravenous chemotherapy for primary ovarian cancer and comparison between carboplatin- and cisplatin-based intraperitoneal chemotherapy. Journal of Korean Medical Science, 32(12), 2021-2028. https://doi.org/10.3346/jkms.2017.32.12.2021
Facing Our Risk of Cancer Empowered. (2023). Ovarian, fallopian tube, and primary peritoneal cancer risk management. https://www.facingourrisk.org/info/risk-management-and-treatment/ovarian-cancer-risk-factors
Ferriss, J. S., Java, J. J., Bookman, M. A., Fleming, G. F., Monk, B. J., Walker, J. L., Homeslay, H. D., Fowler, J., Greer, B. E., Boente, M. P., & Burger, R. A. (2015). Ascites predicts treatment benefit of bevacizumab in front-line therapy of advanced epithelial ovarian, fallopian tube, and peritoneal cancers: An NRG oncology/GOG study. Gynecologic Oncology, 139(1), 17-22. https://doi.org/10.1016/j.ygyno.2015.07.103
Fraune, C., Rosebrock, J., Simon, R., Hube-Magg, C., Makrypidi-Fraune, G., Kluth, M., Buscheck, F., Hoflmayer, D., Schmalfeldt, B., Muller, V., Wolber, L., Witzel, I., Paluchowski, P, Wilke, C., Heilenkotter, U., von Leffern, I., Clauditz, T. S., Wilczak, W., Sauter, S., & Burandt, E. (2020). High homogeneity of MMR deficiency in ovarian cancer. Gynecologic Oncology, 156(3), 669-675. https://doi.org/10.1016/j.ygyno.2019.12.031
GlaxoSmithKline. (2023). Highlights of prescribing information: Jemperli (dostarlimab-gxly) injection, for intravenous use. https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Jemperli/pdf/JEMPERLI-PI-MG.PDF
Holesh, J. E., Bass, A. N., & Lord, M. (2023). Physiology, ovulation. StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK441996/
ImmunoGen. (2022). Highlights of prescribing information: Elahere (mirvetuximab soravtansine-gynx) injection, for intravenous use. https://www.elahere.com/pdf/prescribing-information.pdf
International Atomic Energy Agency. (n.d.). Computed tomography – what patients need to know. Retrieved September 17, 2023, from https://www.iaea.org/resources/rpop/patients-and-public/computed-tomography
International Federation of Gynecology and Obstetrics. (2014). FIGO ovarian cancer staging. https://www.sgo.org/wp-content/uploads/2012/09/FIGO-Ovarian-Cancer-Staging_1.10.14.pdf
Klemencic, S., & Perkins, J. (2019). Diagnosis and management of oncologic emergencies. West J Emerg Medicine, 20(2), 316-322. https://doi.org/10.5811/westjem.2018.12.37335
Konstantinopoulos, P. A., Norquist, B., Lacchetti, C., Armstrong, D., Grisham, R. N., Goodfellow, P. J., Kohn, E. C., Levine, D. A., Liu, J. Y., Lu, K. H., Sparacio, D., & Annunziata, C. M. (2019). Germline and somatic tumor testing in epithelial ovarian cancer: ASCO guidelines. Journal of Clinical Oncology, 38(11), 1222-1245. https://doi.org/10.1200/JC0.19.02960
Kurian, A. W., Hughes, E., Handorf, E. A., Gutin, A., Allen, B., Hartman, A., & Hall, M. J. (2017). Breast and ovarian cancer penetrance estimates derived from germline multiple-gene sequences results in women. JCO Precision Oncology, 1, 1-12. https://doi.org/10.1200/PO.16.00066
Lumen Learning. (n.d.). Anatomy of the female reproductive system. Retrieved September 17, 2023, from https://courses.lumenlearning.com/boundless-ap/chapter/the-female-reproductive-system/
Mai, J., Wi, L., Yang, L., Sun, T., Liu, X., Yin, R., Jiang, Y., Li, J., & Li, Q. (2023). Therapeutic strategies targeting folate receptor a for ovarian cancer. Frontiers in Immunology, 14, 1254532. https://doi.org/10.3389/fimmu.2023.1254532
Masoud, V., & Pages, G. (2017). Targeted therapies in breast cancer: New challenges to fight against resistance. World Journal of Clinical Oncology, 8(2), 120-134. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5385433/
Mayo Clinic. (2022). Oophorectomy (ovary removal surgery). https://www.mayoclinic.org/tests-procedures/oophorectomy/in-depth/breast-cancer/art-20047337
McCance, K. L., & Heuther, S. E. (2019). Pathophysiology: The biologic basis for disease in adults and children (8th ed.). Elsevier.
Merck & Co. (2023). Highlights of prescribing information: Keytruda (pembrolizumab) injection, for intravenous use. https://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf
Mitra, A. K. (2016). Ovarian cancer metastasis: A unique mechanism of dissemination. https://www.intechopen.com/books/tumor-metastasis/ovarian-cancer-metastasis-a-unique-mechanism-of-dissemination
Moore, K. N., Angelergues, A., Konecny, G. E., Banerjee, S. N., Pignata, S., Colombo, N., Moroney, J. W., Cosgrove, C., Lee, J., Roszak, A., Breuer, S., Tromp, J. M., Bello-Roufai, D., Gilbert, L., Miller, R., Myers, T. K., Wang, Y., Berkenbilt, A., Lorusso, D., & Van Gorp, T. (2023). Phase III MIRASOL (GOG 3045/ENGOT-ov55) study: Initial report of mirvetuximab soravtansine vs. investigator’s choice of chemotherapy in platinum-resistant, advanced high-grade epithelial ovarian, primary peritoneal, or fallopian tube cancers with high folate receptor-alpha expression. Journal of Clinical Oncology, 41 (no. 17_ suppl), LBA5507-LBA5507. https://ascopubs.org/doi/pdf/10.1200/JCO.2023.41.17_suppl.LBA5507?role=tab
Motohara, T., Masuda, K., Morotti, M., Zheng, Y., E-Sahhar, S., Chong, K. Y., Wietek, N., Alsaadi, A., Karaminejadranjbar, M., Hu, Z., Artibani, M., Gonzalez, L. S., Katabuchi, H., Saya, H., & Ahmed, A. A. (2018). An evolving story of the metastatic voyage of ovarian cancer cells: Cellular and molecular orchestration of the adipose-rich metastatic microenvironment. Oncogene, 38, 2885-2898. https://doi.org/10.1038/s41388-018-0637-x
National Cancer Institute. (2018). Oral contraceptives and cancer risk. https://www.cancer.gov/about-cancer/causes-prevention/risk/hormones/oral-contraceptives-fact-sheet
National Cancer Institute. (2023a). Genetics of breast and gynecologic cancers (PDQ®)-Health professional version. https://www.cancer.gov/types/breast/hp/breast-ovarian-genetics-pdq#_88
National Cancer Institute. (2023b). Ovarian, fallopian tube, and primary peritoneal cancer prevention (PDQ®). https://www.ncbi.nlm.nih.gov/books/NBK65937
National Cancer Institute. (2023c). SEER cancer stat facts: Ovarian cancer. https://seer.cancer.gov/statfacts/html/ovary.html
National Comprehensive Cancer Network. (2023a). NCCN clinical practice guidelines in oncology (NCCN guidelines®) genetic/familial high-risk assessment: Breast, ovarian, and pancreatic: Version 1.2021. https://www.nccn.org/professionals/physician_gls/pdf/genetics_bop.pdf
National Comprehensive Cancer Network. (2023b). NCCN clinical practice guidelines in oncology (NCCN guidelines®) ovarian cancer including fallopian tube cancer and primary peritoneal cancer: Version 2.2023 – June 2, 2023. https://www.nccn.org/professionals/physician_gls/pdf/ovarian.pdf
National Institute of Biomedical Imaging and Bioengineering. (2016). Ultrasound. https://www.nibib.nih.gov/science-education/science-topics/ultrasound
National Ovarian Cancer Coalition. (n.d.). What are the signs & symptoms of ovarian cancer? Retrieved October 1, 2023, from https://ovarian.org/about-ovarian-cancer/signs-and-symptoms/
Nettina, S. M. (2019). Lippincott manual of nursing practice (11th ed.). Wolters Kluwer.
Nielson, M., Infante, E., & Brand, R. (2021). MUTYH polyposis. GeneReviews [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK107219/#
Office on Women’s Health. (2021). Menopause. https://www.womenshealth.gov/menopause
Oien, D. B., & Chien, J. (2016). TP53 mutations as a biomarker for high-grade serous ovarian carcinoma: Are we there yet? Translational Cancer Research, 5(Suppl 2), S264-S268. https://doi.org/10.21037/tcr.2016.07.45
Olawaiye, A. B. (n.d.). AJCC cancer staging 8th edition gynecologic cancers. Retrieved September 17, 2023, from https://www.facs.org/media/iomn5exr/gynecologic-cancers-8th-ed.pdf
Olsen, M., LeFebvre, K., Walker, S., & Dunphy, E. P. (2023). Chemotherapy and immunotherapy guidelines and recommendations for practice (2nd ed.). Oncology Nursing Society.
OneCare Media. (2021). CA-125. https://labtestsonline.org/tests/ca-125#
Ovarian Cancer Research Alliance. (n.d.). What is ovarian cancer? Retrieved September 19, 2023, from https://ocrahope.org/patients/about-ovarian-cancer/types-ovarian-cancer/#epithelial-ovarian-tumors-primary-peritoneal-tumors-fallopian-tube-tumors
Peshkin, B. N., & Isaacs, C. (2023). Genetic testing and management of individuals at risk of hereditary breast and ovarian cancer syndromes. UpToDate. Retrieved September 17, 2023, from https://www.uptodate.com/contents/genetic-testing-and-management-of-individuals-at-risk-of-hereditary-breast-and-ovarian-cancer-syndromes
Pettinato, M. C. (2021). Introduction to antibody-drug conjugates. Antibodies (Basel), 10(4), 42. https://doi.org/10.3390/antib10040042
Ring, K. L., & Modesitt, S. C. (2018). Hereditary cancers in gynecology. Obstetrics and Gynecology Clinics, 45(1), 155-173. https://doi.org/10.1016/j.ogc.2017.10.011
Salazar, C., Campbell, I. G., & Gorringe, K. L. (2018). When is “type I” ovarian cancer not “type I”? Indications of an out-dated dichotomy. Frontiers in Oncology, 8(654), 1-9. https://doi.org/10.3389/fonc.2018.00654
Sasikumar, P. G. & Ramachandra, M. (2018). Small-molecule immune checkpoint inhibitors targeting PD-1/PDL1 and other emerging checkpoint pathways. BioDrugs, 35(5), 481-497. https://doi.org/10.1007/s40259-018-0303-4
Sengupta, S. (2017). Cancer nanomedicine: Lessons for immune-oncology. Trends Cancer, 3(8), 551-560. https://doi.org/10.1016/j.trecan.2017.06.006
Society of Gynecologic Oncology. (2017). Committee opinion no. 716: The role of the obstetrician and gynecologist in the early detection of epithelial ovarian cancer in women at average risk. Obstetric Gynecol, 130(3), e149-e149. https://doi.org/10.1097/AOG.0000000000002299
Temkin, S. M., Bergstrom, J., Samimi, G., & Minasian, L. (2018). Ovarian cancer prevention in high-risk women. Clin Obstet Gynecol, 60(4), 738-757. https://doi.org/10.1097/GRF.0000000000000318
US Food & Drug Administration. (2023). FDA DISCO burst edition: FDA approval of elahere (mirvetuximab soravtansine-gynx) for FRa positive, platinum-resistant epithelial ovarian, fallopian tube, or peritoneal cancer. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-disco-burst-edition-fda-approval-elahere-mirvetuximab-soravtansine-gynx-fra-positive-platinum
US National Library of Medicine. (2020). TP53 gene: Tumor protein p53. https://medlineplus.gov/genetics/gene/tp53/#conditions
US National Library of Medicine. (2021). Lynch syndrome. https://ghr.nlm.nih.gov/condition/lynch-syndrome
US Preventive Services Task Force. (2018). Screening for ovarian cancer: US Preventive Task Force recommendation statement. JAMA, 319(6), 558-594. https://doi.org/10.1001/jama.2017.21926
Woo, J., & Long, J. (2021). Ovarian cancer & ovarian tumors. In Papadakis M. A., McPhee S. J., & Rabow M. W. (Eds.), Current Medical Diagnosis & Treatment 2021. McGraw-Hill. https://accessmedicine-mhmedical-com.gshremote.senylrc.org/content.aspx?bookid=2957§ionid=249381517
Yarbro, C. H., Wujcik, D., & Gobel, B. H. (Eds.). (2018). Cancer nursing: Principles and practice (8th ed.). Jones & Bartlett Learning.