About this course:
The purpose of this learning activity is to enhance clinical practice and improve patient outcomes by educating advanced practice registered nurses (APRNs) on the various types of diagnostic radiology imaging tests, ensuring an adequate understanding of the appropriate indications for ordering each exam, as well as the risks, benefits, and critical clinical considerations regarding the use of contrast media.
Course preview
Diagnostic Radiology for APRNs
The purpose of this learning activity is to enhance clinical practice and improve patient outcomes by educating advanced practice registered nurses (APRNs) on the various types of diagnostic radiology imaging tests, ensuring an adequate understanding of the appropriate indications for ordering each exam, as well as the risks, benefits, and critical clinical considerations regarding the use of contrast media.
By the completion of this module, learners should be able to:
- describe the background of radiology, outline the components of the medical imaging process, and discuss the characteristics that impact image quality
- discuss the various aspects of radiation, the essential components of radiation safety, and identify the amount of radiation in some of the most common imaging tests
- differentiate between the general risks of medical imaging tests with radiation exposure and those without radiation exposure
- describe the different types of diagnostic radiology tests and the indications for ordering, key patient teaching points, what to expect during the test, how long the test should take, as well as the associated risks and benefits
- review the different types of contrast media agents regarding their clinical considerations, including risks, contraindications, and monitoring parameters
- outline the signs and symptoms of allergic reactions to iodinated contrast, as well as the management and premedication regimens as guided by the American College of Radiology (ACR)
- recognize the components of the Appropriate Use Criteria for Advanced Diagnostic Imaging as devised by the Centers for Medicare & Medicaid Services
- discuss the guidelines for diagnostic imaging during pregnancy and lactation
Radiation
Radiation is energy in the form of particles or waves emitted by both natural and synthetic sources. Comprised of two forms, ionizing and non-ionizing, radiation surrounds us in our daily lives. As demonstrated in Figure 1, radiation exists on the electromagnetic (EM) spectrum, ranging from lower-energy microwaves to higher-energy gamma rays. EM energy travels in waves, and the strength of the radiation depends on the frequency (how rapidly the waves move up and down) and the distance the wavelength is traveling. In general, the smaller the wavelength, the higher the energy of the radiation (The Centers for Disease Control and Prevention [CDC], 2015c).
Figure 1
The Electromagnetic Spectrum
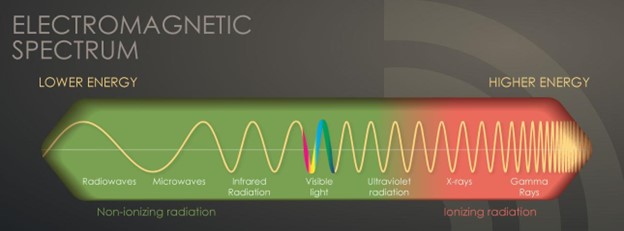
(CDC, 2015c)
Non-ionizing radiation is the most prevalent form of radiation freely available at low levels within our environment. It can heat substances but cannot remove electrons from atoms and molecules. The most common types of non-ionizing radiation include radiofrequency (RF) waves in many communication and electronic devices, kitchen microwaves, visible light, and lower energy forms of ultraviolet (UV) radiation. Intense, direct exposure to RF waves and microwaves can result in tissue damage due to heat. In contrast, overexposure to UV rays can result in skin burns, premature aging, eye damage, and skin cancer. Most skin cancers are directly related to intense, prolonged, and unprotected exposure to UV radiation from the sun and tanning beds. The line dividing ionizing and non-ionizing radiation becomes somewhat blurred in the UV section of the EM spectrum, demonstrated in Figure 1. Radiation in the UV section at lower energies is considered non-ionizing, whereas, at higher energies, extreme UV radiation becomes more harmful, rendering it ionizing radiation (CDC, 2015b, 2015c).
Ionizing radiation removes electrons from atoms and molecules, causing the atom to become ionized (or charged), enabling the wavelengths to pass through air, water, and tissue. Ionizing radiation is considered a carcinogen, or a substance capable of causing cancer, as it can penetrate the human body. When absorbed by living tissue, ionizing radiation can induce harm, especially when exposed to high levels. Most humans are exposed to low levels of ionizing radiation daily, and these radiation sources may be natural or artificial. Exposure can occur through construction and building materials, terrestrial radiation (radiation from the earth), cosmic radiation (radiation from space), and air travel. Some people are exposed to higher amounts of natural background radiation, such as those living at higher mountain elevations or engaging in frequent air travel. X-rays are the most classic example of ionizing radiation; they penetrate the body to visualize underlying bony structures. Ionizing radiation has enough energy to alter molecules within the cells of the human body, targeting genetic material or DNA. Radiation interacts with DNA directly and indirectly, as it can destroy bonds within the DNA and the water molecules surrounding the DNA. When the water molecules are broken, they generate free radicals or unstable oxygen molecules that damage cells, tissues, and organs (ACR, 2018; CDC, 2015c, 2021b). When a cell is damaged from radiation exposure, one of the following three events will occur:
- the cell will repair itself and restore its normal function
- the cell will remain altered, or only partially repair the damage, thereby heightening the risk for future cancer development
- the cell will die, and the body will recover; however, if there is widespread cell death, this can increase the risk of organ failure (CDC, 2015a)
Diagnostic Radiology
Diagnostic radiology refers to a medical sector that includes various medical imaging technologies widely utilized throughout the US healthcare system, with millions of patients undergoing imaging evaluations daily. These are noninvasive and minimally invasive medical procedures used to view the inside of the body to assess, diagnose, monitor, and treat various medical conditions. Several of these tests also serve as the backbone of preventative medicine and routine health maintenance, including cancer prevention, screening, and early detection. While diagnostic radiology is valuable and critical to properly diagnosing numerous medical conditions, there are risks. Many of these tests are linked to potential harm and a high-cost burden to patients and society that must be considered before ordering the test. To promote patient safety and ensure optimal outcomes, clinicians must understand each type of imaging modality and be aware of the clinical risks, benefits, and management of adverse events (Elsayes & Oldham, 2014).
Radiology is considered one of the most technologically advanced medical fields, dating back to 1895 when Wilhelm Conrad Rontgen first discovered the x-ray, using his wife as his patient. Subsequently, Henri Becquerel discovered radioactivity in 1896, followed by the discovery of radium in 1898 by Marie and Pierre Curie. The field has expanded exponentially over the last few centuries and relies on the collaboration of scientists, medical physicists, radiologists, and imaging technologists. Medical physicists ensure the safe and optimal use of radiological imaging modalities in patients. Diagnostic radiologists are physicians who undergo specialized training in the analysis and interpretation of medical imaging to draw conclusions helpful in diagnosing, treating, and managing acute and chronic medical conditions and injuries. Interventional radiologists are physicians who undergo specialized training in medical imaging to perform minimally invasive surgical procedures that diagnose, treat, and cure many...
...purchase below to continue the course
Imaging Process
According to the International Atomic Energy Agency (IAEA), a radiograph, or medical image, "is a pictorial representation of a measurement of an object or function of the body" (IAEA, 2014, p. 55). The overall objective of medical imaging is to make a particular area within the patient's body visible. Radiographs are created by sending an x-ray beam, generated by an x-ray tube, through a patient. As described in the most simplistic form, the image represents a shadow of the underlying structures the x-ray beam passes through. The medical imaging process has five core components: the patient, the imaging system, the system operator (the radiographer), the resultant image, and the observer (the radiologist). Each medical imaging method is devised to reveal distinctive characteristics of the body, and the variability in imaging quality and visibility of structures can differ considerably. Some of the most common factors contributing to the inconsistencies in the resulting images include the quality and characteristics of the imaging equipment, the skill of the operator (including positioning and placement of the patient), specific patient characteristics (such as body habitus, prior surgeries, presence of scar tissue, or prior radiation therapy), as well as the timing of the imaging in relation to the injury or medical condition in question (IAEA, 2014).
Imaging Quality
Obtaining high-quality images of optimal clarity is critical to draw precise conclusions. High-quality images enable radiologists to visualize the body's structures, evaluate the underlying injury or medical condition, and make accurate diagnoses. Poor-quality images hinder the ability to assess and evaluate the condition accurately, leading to inconclusive or wrongful diagnoses. This often leads to the need for additional imaging tests, increasing the risks to the patient, contributing to delays in the diagnosis and treatment of the condition, and also adding to the high-cost burden to patients and society (Elsayes & Oldham, 2014).
One key aspect of imaging quality is the signal-to-noise ratio, where signal refers to the information obtained from the part of the body being imaged, and noise refers to anything that hinders the ability to access this information. Higher-quality radiographic images have higher signal levels than noise, allowing structures within the body to be seen clearly. Images of low quality have a poor signal-to-noise ratio; in other words, the signal level is similar to or less than the noise level, which causes the structures to become obliterated. Imaging artifact is a commonly reported contributor to poor imaging quality. An artifact is a visual anomaly or any feature which appears in an image that is not present within the original imaged portion of the body. Artifacts misrepresent structures and can obscure underlying structures and simulate pathology (Holmes & Griffiths, 2016). Additional characteristics that impact the quality of medical images are highlighted in Table 1.
Table 1
Characteristics that Impact Image Quality
Characteristic | Description |
Magnification and distortion | Areas subject to radiographic imaging are larger than the actual body part being evaluated. For optimal imaging quality, the x-ray positioning should be as close to the body part in question as possible and placed in a parallel position to the body part being imaged to provide optimal magnification and minimize image distortion. |
Sharpness and blur | Sharpness is essential to producing a higher acuity image, whereas the blurring of an image reduces the image quality. Factors that affect image sharpness include:
|
Contrast and density | Contrast is the most fundamental characteristic of an image and refers to the difference in brightness (density) between two adjacent structures or between the area of interest and its surroundings. The more significant the difference between the adjacent tissue types, the easier it is to identify separate structures. Density is defined as the degree of blackening on the film. Four natural tissue densities range from dark to light shades based on their underlying structure. Gas, found in the lungs or throughout the GI tract, appears black on x-ray films. Adipose tissue appears dark gray. Fluid and soft tissues (connective tissue, muscles) appear light gray. Bone is the densest natural tissue, appearing nearly white, while anything metal appears white. |
Equipment | The resolution of the monitor on which the radiologist is viewing the images and the sophistication of the computer-based picture archiving and communication system (PACS) also impacts image quality. Better quality and more precise images are associated with higher-resolution monitors and more advanced PACS systems. |
(Elsayes & Oldham, 2014; Holmes & Griffiths, 2016; Lloyd-Jones, 2016)
Risks with Medical Imaging
Many diagnostic radiology imaging tests and procedures involve exposure to ionizing radiation, which increases the risk of harmful effects. Some of the most common examples of imaging tests associated with the highest ionizing radiation exposure include x-rays, computed tomography (CT scans), fluoroscopy, and nuclear medicine scans. Ionizing radiation can penetrate deep into the body, and repeated exposure from imaging tests increases the risk of cancer later in life. Specific populations, such as infants and young children, are more vulnerable to the harmful health effects of radiation exposure. Younger age groups have more cells dividing rapidly and tissues growing, placing them at higher risk for long-term effects. Also, young people have a longer lifespan ahead of them, which gives cancer more time to develop (CDC, 2015a, 2022b).
Pregnant women are also considered another vulnerable population due to the potential for harming the fetus at various stages of development. However, data remains uncertain and inconsistent on the suspected risks in utero and to the newborn with radiation exposure. The ACR describes radiation exposure at levels less than 100 milligrays (mGy) as having "probably no effect" or "potential effects are scientifically uncertain and probably too subtle to be clinically detectable." However, at dose levels greater than 100 mGy, there is the potential for "possible spontaneous abortion, possible malformations increasing in likelihood as the dose increases, and risk for diminished IQ or of [intellectual disability], increasing in frequency and severity with increasing dose" (ACR, 2018, p. 3). The following section will describe the current guidelines on medical imaging in pregnancy. Older adults, children, and individuals with compromised immune systems are also considered vulnerable populations concerning radiation exposure. Older adults are at heightened risk from lifelong radiation exposure, impaired organ function, and other factors of aging that already place them at increased risk for cancer development. Younger individuals, including children, are at an increased risk from radiation due to the presence of rapidly dividing cells and a longer potential lifespan for adverse effects such as cancer to develop. Further, individual sensitivity is another important factor regarding radiation, as, for unknown reasons, some individuals appear to be more sensitive to its effects than others (CDC, 2015a).
The health effects associated with ionizing radiation are strongly linked to the dose. The radiation dose or the amount of radiation is the critical factor when evaluating the risk for future unintended health consequences, as the risks are dose-dependent; the dose makes the poison. However, the amount of ionizing radiation absorbed by the body is what induces harm to health. At high doses, radiation can be lethal; lower doses can lead to serious health consequences. Other factors that influence the health effects of radiation exposure include how fast the dose was received, what body location was exposed to the radiation, and how sensitive the body is to radiation. As described in Table 2, the radiation dose is not the same as that of standard medication. There are three principal ways in which radiation doses are measured: absorbed dose, equivalent dose, and effective dose (CDC, 2015a).
Table 2
Radiation Doses
Dose | How Radiation Doses are Measured |
Absorbed dose
|
|
Equivalent dose |
|
Effective dose |
|
(RadiologyInfo.org, 2022d)
The absorbed and equivalent doses are used to evaluate the short-term effect of radiation on tissue, which ranges from weeks to months. When diagnostic imaging is performed correctly, there are generally no short-term effects; therefore, the absorbed and equivalent doses are less meaningful in clinical practice. The effective dose is the most important and valuable dose quantity for most patients since it applies to long-term effects (RadiologyInfo.org, 2022d). The absorbed radiation dose varies widely based on the different types of examinations. Nuclear medicine scans, such as the positron emission tomography-computed tomography (PET/CT) scan, have the highest associated radiation exposure among the most common medical imaging tests. A PET/CT scan is comparable to eight years of natural background radiation and poses a moderately increased (1 in 1,000 to 1 in 500) estimated lifetime risk of dying from fatal cancer. Table 3 provides a detailed overview of the radiation dose to adult patients associated with common diagnostic radiology imaging examinations and a classification of risk levels. Further, the radiation dose varies based on the operator and the facility equipment (American Cancer Society [ACS], 2018). Aside from increasing one's risk for future cancer development, ionizing radiation exposure can also contribute to the development of cataracts, skin reddening or skin burns, and hair loss. However, these health effects most commonly result from exposure to very high levels of radiation associated with radiation oncology treatment fields, which require a significantly higher dose of radiation delivered directly to a localized area of the body (CDC, 2022b).
Table 3
Radiation Dose from Common Diagnostic Imaging Tests
Imaging modality | Specific test | Effective radiation dose (approximate) | Comparison to natural background radiation in years | The estimated lifetime risk of fatal cancer* |
X-ray | X-ray spine | 1.5 mSv | 6 months | Very low |
X-ray chest | 0.1 mSv | 10 days | Minimal | |
X-ray extremity | 0.001 mSv | 3 hours | Negligible | |
X-ray upper gastrointestinal (GI) | 6 mSv | 2 years | Low | |
X-ray lower GI | 8 mSv | 3 years | Low | |
X-ray intraoral (dental) | 0.005 mSv | 1 day | Negligible | |
Intravenous pyelogram (IVP) | 3 mSv | 1 year | Low | |
CT scan | CT head | 2 mSv | 8 months | Very Low |
CT head (with contrast) | 4 mSv | 16 months | Low | |
CT spine | 6 mSv | 2 years | Low | |
CT chest | 7 mSv | 2 years | Low | |
CT chest (lung cancer screening, low-dose CT scan) | 1.5 mSv | 6 months | Very low | |
CT abdomen and pelvis | 10 mSv | 3 years | Low | |
CT abdomen and pelvis (with contrast) | 20 mSv | 7 years | Moderate | |
Coronary CT angiography (CTA) | 12 mSv | 4 years | Low | |
Cardiac CT for calcium scoring | 3 mSv | 1 year | Low | |
Nuclear medicine | PET/CT scan | 25 mSv | 8 years | Moderate |
Bone densitometry (DXA) scan | 0.001 mSv | 3 hours | Negligible | |
Multigated acquisition (MUGA) scan | 7.8 mSv | 2.5 years | Low | |
Breast imaging | Mammography | 0.4 mSv | 7 weeks | Very low |
*Estimated lifetime risk of fatal cancer from the test's radiation exposure | ||||
Negligible Less than 1 in 1,000 | Minimal 1 in 1,000,000 to 1 in 100,000 | Very Low 1 in 100,000 to 1 in 10,000 | Low 1 in 10,000 to 1 in 1,000 | Moderate 1 in 1,000 to 1 in 500 |
(Huynh, 2017; RadiologyInfo.org, Radiological Society of North America, & American College of Radiology, 2022)
Radiation Safety
While the potential for increased risk of adverse health effects from diagnostic imaging is clearly described, there is no universally recognized threshold for specific radiation dose and associated effects. Therefore, it has been argued that there is "no safe level" of radiation exposure. The priority is to ensure the potential risks of ionizing radiation are continuously weighed against the benefit derived from the imaging test or procedure. This clinical decision should be made between the ordering provider and the patient with full disclosure of the potential risks of imaging from radiation exposure compared with the predicted benefits (ACR, 2018). The US Department of Labor's Occupational Safety and Health Administration (OSHA) outlines standards for controlling ionizing radiation hazards and preventing radiation exposure to healthcare workers and patients. Diagnostic radiology departments within hospitals and free-standing diagnostic radiology facilities must implement radiation protection programs managed by a radiation safety officer (RSO), such as a radiologist or a medical physicist who is a qualified expert. The radiology equipment is housed in designated areas within hospitals, usually on the ground floor, and secured behind lead doors to reduce exposure to employees and patients. Radiologists and radiology technicians must undergo specialized training in radiation safety practices governed by state and federal law. OSHA standards require appropriate radiation caution signage to alert individuals to radiation usage or storage in designated areas, such as the bright yellow caution sign used in most medical facilities (OSHA, n.d.)
A central concept of radiation protection programs is keeping each healthcare worker's occupational radiation dose as limited as possible by implementing "As Low As Reasonably Achievable" (ALARA) programs. ALARA is premised on three chief components of time, distance, and shielding as follows:
- time: minimize the time spent in areas with elevated radiation levels; this can be accomplished with proper preparation before testing to limit the time the patient is in the exposure area and the need for unnecessary exposure due to redundant imaging
- distance: maximize the distance from sources of radiation, as a worker's radiation dose decreases as the distance from the source increases; for gamma rays and x-rays, the intensity of radiation is inversely proportional to the square of the distance from the source
- shielding: use shielding for radiation sources (i.e., placing an appropriate shield between sources of radiation and the worker), inserting the proper shielding (e.g., lead, concrete, or special plastic shields depending on the type of radiation) between a worker and a radiation source will significantly reduce or eliminate the dose received by the worker. Shielding also refers to lead doors and geographic locations within the facility to minimize exposure to other individuals who are not directly working within that environment (Frane & Bitterman, 2022; OSHA, n.d.)
Shielding patients with lead aprons and lead coverings during diagnostic imaging tests has been the standard practice since its endorsement by the US Food & Drug Administration (FDA) in the US Code of Federal Regulations in 1976. This practice originated due to concerns related to genetic fertility risks (such as germ cell mutations affecting future generations), and gonadal shielding was advised and implemented for all x-rays. Despite the previous thoughts, the focus on discontinuing patient shielding practices has become increasingly prominent in radiology literature over the last few years. On April 2, 2019, the American Association of Physicists in Medicine (AAPM) released a position statement outlining why routine fetal and gonadal shielding is unnecessary since there is no evidence that the amount of radiation exposure from diagnostic imaging has any adverse effects on reproductive cells. Even after exposure to atomic bombings, no genetic effects have been observed, even three to four generations later. It is now known that the amount of radiation exposure needed to elicit detrimental effects on fertility is 100 times higher than the dose received from diagnostic imaging, further supporting the discontinuation of patient gonadal and fetal shielding in routine practice. On May 30, 2019, the ACR submitted a letter to the AAPM, endorsing their position on patient gonadal and fetal shielding. The ACR is adapting this new change into their guidelines, with the objective that this recommendation is universally adopted and becomes the standard of care across diagnostic radiology (AAPM, n.d., 2019; National Council on Radiation Protection and Measurements [NCRP], 2021).
Some studies have even begun to establish evidence that shielding carries a substantial risk of increasing the absorbed radiation dose and compromising the diagnostic efficacy of the acquired images. Part of these risks is related to advancements in diagnostic imaging technologies. Modern x-ray imaging systems have an automatic exposure control, in which a sensor detects when the target dose is obtained. In fluoroscopy procedures, automated brightness control algorithms function as a feedback loop to constantly adjust the x-ray tube output to ensure quality images are consistently obtained. Therefore, with these new technologies, if a lead shield is applied and enters the field of view, the radiographic tube will automatically adjust and drastically increase the radiation output to try to penetrate the shield. This heightens the risk of increased absorbed radiation by the patient (AACM, 2019; NCRP, 2021).
Diagnostic Imaging Tests with Radiation Exposure
Imaging studies can be divided into two main categories: planar and cross-sectional. Planar studies produce two-dimensional (2D) images, including basic diagnostic radiology testing such as x-rays and mammography. Cross-sectional imaging techniques address the three-dimensional (3D) aspects of human anatomy by capturing more detailed images, often called "slices." This imaging technology can then create a composite analysis of 2D slices to provide a 3D visualization of the anatomy. Cross-sectional imaging includes CT scans, ultrasound, and magnetic resonance imaging (MRI; Elsayes & Oldham, 2014).
X-ray
X-ray is the most common and readily available type of diagnostic imaging test. Often referred to as a plain film, an x-ray is a quick, noninvasive, and painless imaging modality that produces images of the structures inside the body. In a standard x-ray, a beam of energy is generated by an x-ray generator and aimed at the intended body part, and a plate is placed behind the body part to capture the variations of the energy beam. This is the simplest example of how ionizing radiation produces a 2D image. For optimal imaging quality, the x-ray detector (the plate) should be as close to the body part (patient or object being imaged) as possible. The radiation beams should be perpendicular (situated at a right angle) to the body part, as this helps to minimize the magnification and enhance the sharpness of the resulting image, thereby producing a more precise result (Elsayes & Oldham, 2014).
X-rays are widely utilized throughout various healthcare domains with several indications. They help diagnose acute bone fractures and cardiopulmonary conditions, such as cardiac enlargement, pneumonia, or pleural effusion (fluid inside the lung). Dental care commonly uses x-rays to identify cavities and underlying dental pathology. They are also helpful for diagnosing arthritis and identifying foreign bodies or bowel obstructions. In addition, x-rays may assist in fluoroscopy procedures, facilitating the placement of tubes or other devices inside the body. Similarly, x-rays are also used to verify the proper placement of a device following surgery or to ensure no medical devices are left in the body (IAEA, n.d.; National Institute of Biomedical Imaging and Bioengineering [NIBIB], 2022).
Patients are required to remove any clothing or jewelry, especially metal, that may interfere with the procedure. This standard applies to all forms of diagnostic imaging tests and procedures. The patient should be educated that they will either lie, sit, or stand still while the x-ray machine takes images. The patient may be asked to move into several specific positions to obtain the highest quality imaging results. For instance, when evaluating a patient for pneumonia, the patient is usually asked to take a deep breath and hold it, which helps expand the lung fields and provide a higher-quality image. In addition, several photos may be taken from different viewpoints to facilitate a good view. The entire process should take at most 15 minutes. As described in Table 2, ionizing radiation exposure from a conventional x-ray is very low to negligible. Adverse effects are rare; however, the risk versus benefit ratio must be carefully considered before ordering any x-ray (Elsayes & Oldham, 2014).
Fluoroscopy
Fluoroscopy is a medical imaging test used to study the motion of internal body structures. According to the CDC, fluoroscopy uses an x-ray beam that passes continuously through the body to create a real-time video. The video is projected on a monitor, allowing clinicians to evaluate the movement of internal organs or devices in real time. Medical imaging tests using fluoroscopy are generally noninvasive and performed to evaluate specific body areas, determining the cause of a particular health problem. Fluoroscopy can help evaluate the bones, muscles, joints, and solid organs, such as the heart, lungs, or kidneys. It can be used alone as a diagnostic procedure or in combination with other procedures. Fluoroscopy plays an essential role in preventing health problems and diagnosing diseases. It is used in many types of diagnostic testing and procedures. Exposure to ionizing radiation during fluoroscopy depends on the test and equipment used (CDC, 2021c). Fluoroscopy is commonly used to:
- evaluate the functioning of the GI tract
- assess swallowing ability
- visualize fractures and determine if the surgical intervention has healed the injury
- perform cardiac catheterization
- locate foreign bodies
- guide medical procedures involving the placement of catheters, stents, or other devices within the body
- guide anesthesia injections into joints or the spine (CDC, 2021c)
Barium X-rays
Barium x-rays are used to diagnose underlying pathology within the GI tract, including ulcers, inflammation, tumors, hernias, or strictures. Barium is a white, chalk-like powder mixed with water to create a liquid that is either ingested by the patient or administered via an enema. The barium coats the lining of the GI tract, providing visualization of the walls of the esophagus, stomach, and intestines. This allows radiologists to evaluate these structures' contour, shape, size, and patency to identify any underlying pathology. After the barium is administered, fluoroscopy allows the radiologist to see the movement of the barium through the GI tract. There are three barium x-ray procedures: the barium enema or lower GI series, the barium small-bowel meal, and the barium swallow or upper GI series. Barium x-ray tests are typically performed as an outpatient procedure, and each test's defining features and details are described in Table 4 (Gotfried, 2023; Murphy, 2023a). More information regarding barium is discussed later in this module.
Table 4
Barium X-ray Procedures
Procedure | Description |
Barium enema (lower GI series) |
|
Barium small-bowel follow through |
|
Barium swallow (upper GI series) |
|
(Gotfried, 2023; Murphy, 2023a)
Intravenous Pyelogram
IVP, or intravenous urography, is a fluoroscopic procedure that uses iodinated contrast material to assess for abnormalities within the kidneys, ureters, and bladder. The contrast is administered intravenously (IV) and travels through the renal vasculature into the urinary collecting system. The contrast makes these areas appear bright white on the resulting x-ray images, allowing the radiologist to identify any underlying pathology. This test is commonly performed to evaluate the etiology of hematuria or flank pain generated from the kidneys (Mehta & Annamaraju, 2022). IVP is a valuable diagnostic test to assess for the following suspected conditions:
- urinary calculi
- enlarged prostate
- neoplasms of the kidney, ureter, or bladder
- congenital abnormalities of the urinary tract
- complications from surgery on the urinary tract
- scars or urinary strictures (Mehta & Annamaraju, 2022)
Typically performed as an outpatient procedure, patients must empty their bladder immediately before the scan to allow for the best quality images. Following contrast administration, the patient will lie flat on an exam table, and a series of x-ray images are taken while the kidneys are processing the contrast. Depending on the underlying issue, patients may be asked to lie on their side for enhanced images. The exam usually takes up to one hour, but in patients with impaired or sluggish renal function, the exam may take up to four hours. Following the procedure, patients are advised to increase oral hydration to flush the contrast out of the renal system (Mehta & Annamaraju, 2022).
Mammography
Mammography is a breast imaging test that uses low-dose x-rays to view the breast tissue to identify abnormalities suspicious of breast cancer. A mammogram is one of the most widely used cancer screening tools that has successfully identified early breast cancer in asymptomatic women and prevented breast cancer deaths. Approximately 33 million screening mammography exams are performed each year. Since the introduction of screening mammography in the late 1980s, the breast cancer mortality rate in the US has decreased by nearly 40% in individuals ages 40 to 84 compared to no screening (Grimm et al., 2022).
According to the ACS, the lifetime risk of a woman developing breast cancer in their life is 13%, which means that each female in the US has a 1 in 8 risk level of being diagnosed with breast cancer throughout their lifetime; and this risk increases with age. Randomized clinical trials have demonstrated that routine screening mammography can reduce the number of deaths from breast cancer in women aged 40 to 74 years, with the most substantial benefit being those aged 50 to 69. No data demonstrates a benefit to regular preventative screening in individuals under 40. Breast cancer mortality rates have decreased by 49% from 1989 through 2000 (ACS, 2023; National Cancer Institute [NCI], 2023).
In modern practice, mammography is performed for one of the following two indications: as a screening modality or as a diagnostic test. As described above, screening mammography is central to detecting precancerous and cancerous breast lesions in women with no symptoms. The recommendations for breast cancer screening are varied, with conflicting guidance from the American College of Obstetricians and Gynecologists (ACOG), ACS, the National Comprehensive Cancer Care Network (NCCN), and the US Preventative Services Task Force (USPSTF). The ACS recommends annual routine screening beginning at age 45 until 54 and then screening every two years for those 55 or older, the NCCN recommends routine screening beginning at 40, and the USPSTF recommends that screening begins at 50 and is performed every two years (for additional information regarding the specifics on breast cancer screening for early detection, please refer to the NursingCE course entitled Cancer Prevention and Early Detection). Diagnostic mammography is ordered for a patient with abnormalities, such as a palpable breast lump, nipple discharge, or skin changes. Diagnostic mammography may also be advised following an abnormal screening mammogram to obtain enhanced and dedicated images of the area of concern. In many patients, a diagnostic mammogram is performed with an ultrasound of the breast tissue (Grimm et al., 2022; NCI, 2023; Susan G. Komen, 2022).
As demonstrated in Figure 2, a mammogram is performed by compressing the breast tissue within a machine that looks like a rectangular metal box. The patient is instructed to stand facing the mammography machine during the exam, one breast is placed on the flat surface at a time, and a lever called a compression paddle is lowered to squeeze the breast tissue. Compression of the breast tissue is essential to reduce motion, even out the breast thickness, ensure that all breast tissue can be visualized, and allow the x-ray beam to penetrate the breast tissue. Further, compressing the breast tissue also allows for the use of lower doses of radiation when a thinner amount of breast tissue is imaged. The patient will be advised to remain very still during the test and, at times, may be asked to hold their breath to reduce motion and artifact when the x-ray is acquiring the images. Most women describe the exam as uncomfortable due to the pressure on the breast tissue from the compression paddle, but it is generally not considered a painful test. A mammogram usually takes about 30 minutes, and patients can resume normal activities immediately following the test. Once the images are acquired, a radiologist reviews and interprets them (RadiologyInfo.org, 2021a).
Figure 2
Mammography
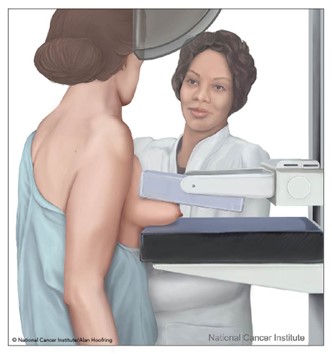
(NCI, 2007)
Standard mammography imaging has evolved significantly over the last few decades, with the development of computer-aided detection (CAD), digital mammography, and breast tomosynthesis. CAD is a form of artificial intelligence devised to search digital mammographic images to help identify possible abnormalities on the mammogram that may otherwise have been missed by highlighting them for the radiologist to examine closely. Despite evidence that the use of CAD did not increase diagnostic accuracy, it was being utilized by 92% of institutions by 2016. CAD has also been associated with an increase in false positives resulting in approximately $400 million in unnecessary healthcare costs. The clinical benefits and efficacy of CAD continue to be studied; however, its use does not replace the need for a qualified radiologist to interpret mammogram images directly (Elmore & Lee, 2022; RadiologyInfo.org, 2021a)
Some women may opt to undergo digital breast tomosynthesis (DBT), also called 3-D mammography, an advanced imaging modality that can capture several images of the breast from different angles. This modality is similar to a CT scan and produces higher-quality images in a series of thin slices combined to form a 3-D image. The radiation dose from some DBT systems is higher than conventional mammography; however, extensive population-based studies have demonstrated improved breast cancer screening detection rates and reduced need for additional views for individuals with dense breasts (Radiology.org, 2021a). Although DBT is a newer imaging modality, some of its clinical benefits include the following:
- earlier detection of small breast cancer that may not have been seen on a conventional mammogram
- particularly helpful in patients with dense breasts
- increased likelihood of detecting multiple breast tumors during one imaging test and pinpointing the size, shape, and specific location of the abnormalities
- a reduced number of unnecessary biopsies or additional imaging tests (RadiologyInfo.org, 2021a)
CT Scans
CT scans, sometimes called computed axial tomography (CAT) scans, use a series of x-rays taken from multiple angles and computer technology to create cross-sections of the inside of the body, including bones, blood vessels, organs, and soft tissues. The x-ray detector moves circulatory around the body to generate several views of the body structure being evaluated. For some scans, the table that the patient is on moves incrementally when each picture is taken; other tests require the table to move continuously. The CT scanner sends x-rays through the body during each scan rotation to form a complete picture in much greater detail than a conventional x-ray (Kocak, 2022a).
CT scans use ionizing radiation as their imaging method and may be performed with or without intravenous contrast administration. Some scans use iodine-based contrast, which may be given IV, orally (PO), or both. When contrast is used, patients are typically required to fast or remain nothing by mouth (NPO) for several hours before the scan to provide a better picture of the body area. Oral contrast is administered about two hours before the examination and is most helpful in visualizing the structures of the abdomen and pelvis. For those receiving IV contrast, patients will need to have a needle inserted into the arm for the injection. Patients should be informed that almost immediately following contrast injection, they may feel warm or flushed and may experience a metallic taste in the mouth. These symptoms are generally transient and only last seconds to minutes before they resolve (RadiologyInfo.org, 2020). Additional details regarding the multifaceted aspects of iodinated contrast administration are described later in this module.
CT scans are better than x-rays at distinguishing the different densities of soft tissue. Due to this, they are the preferred imaging method for evaluating head and neck, spinal, intra-abdominal, intrathoracic, and intracranial structures. CT scans are used to diagnose injuries from trauma, such as bone fractures, infections, internal bleeding, tumors, masses, and cancers, and are also used to guide biopsies. The CT scan is considered a first-line screening modality for patients presenting with acute head trauma or stroke since it can quickly and easily evaluate for hemorrhage or an ischemic event. In these cases, the head CT scan is ordered without contrast, as it is obtained emergently. A CT scan without contrast is also the most accurate way to detect urinary calculi. IV contrast is recommended due to improved imaging when malignancy, infection, or soft-tissue trauma is suspected (Kocak, 2022a).
CT scans are commonly used in the care of oncologic patients as part of cancer staging work-up, to evaluate response to cancer treatments, and monitor for cancer progression or recurrence. CT scans of the face, sinuses, orbits, or neck may also be ordered and are generally performed to evaluate for suspected infection (sinusitis, orbital infection) or mass (malignant or benign tumors) within these locations. A chest CT scan is commonly performed to evaluate the lung parenchyma and the mediastinum for the presence of pulmonary nodules, masses, pleural effusions, or other signs of lung disease. CT imaging of the abdomen and pelvis has many ordering indications. Contrast is generally advised when evaluating suspected appendicitis, diverticulitis, abscess, other infection, and small bowel obstruction. CT angiography (CTA) is a type of CT scan performed to evaluate the blood vessels in a particular area for narrowing, obstruction, or thrombosis. Most commonly, a CTA scan is ordered to evaluate for suspected pulmonary embolism (pulmonary CTA), aortic dissection (thoracic aorta CTA), and brain aneurysm (intracranial CTA). There are numerous additional indications for CT scans and specific imaging protocols based on the suspected injury or illness to ensure the highest quality of images are obtained, which are beyond the scope of this module (Kocak, 2022a; Patel & De Jesus, 2023; Radiologyinfo.org, 2022b).
Patients should be advised to lie flat and remain still on a table that slides into the scanning machine, which resembles a giant doughnut, as shown in Figure 3. The machine rotates around the patient to take all the necessary images, and completing the scan takes 10 to 15 minutes. As explained earlier, patients who undergo CT scans with high doses of radiation and undergo several repeated CT scans are at heightened risk for cancer later in life due to these scans. CT scans and CT-based diagnostic procedures are associated with doses of radiation 50 to 1,000 times higher than conventional x-rays (Kocak, 2022a; Patel & De Jesus, 2023; Radiologyinfo.org, 2020).
Figure 3
CT Scan
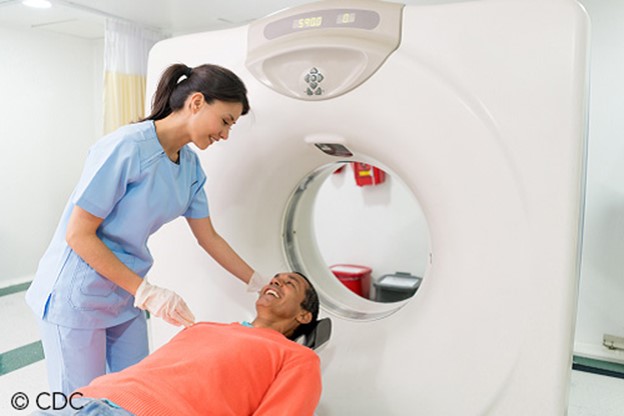
(CDC,2021a)
Nuclear Medicine Imaging
Nuclear medicine imaging differs from conventional diagnostic imaging as it can visualize how the body functions at the cellular and molecular levels. Nuclear imaging uses small quantities of radioactive tracers (radiotracers) to diagnose and treat disease. The radiotracers are most commonly injected into a vein but may also be taken orally or inhaled. The radiotracer travels through the body, releasing energy through gamma rays, which specific tissues and organs absorb. It is then detected by the external scanning device to provide information on organ function and cellular activity (Society of Nuclear Medicine & Molecular Imaging [SNMMI], n.d.).
The radiotracers comprise molecules bonded tightly to a radioactive atom, and these molecules vary greatly depending on the purpose of the scan. Radiotracers must meet FDA standards for safety due to radiation exposure. The Nuclear Regulatory Commission (NRC), the FDA, and individual states regulate radioactive materials for nuclear medicine to ensure the safety of patients, healthcare professionals, and the general public. Each nuclear medicine imaging test uses a specific radioactive agent. Various nuclear medicine imaging tests are available, which are integral to caring for patients with cancer, heart disease, and bone disorders (NIBIB, 2019). Some of the most common include:
- PET/CT
- single photon emission computed tomography (SPECT)
- thyroid scintigraphy and radioactive iodine uptake (RAIU) test
- skeletal scintigraphy (Bone Scan)
- bone densitometry scan (DXA)
- multigated acquisition scan (MUGA; SNMMI, n.d.)
Positron Emission Tomography/Computed Tomography (PET/CT)
PET/CT imaging uses radiotracer fluorine-18 deoxyglucose (FDG) to create 3D images to better locate areas of abnormal cell activity. The information gathered about cell activity and function provided by PET is combined with the anatomic information provided by the CT scan. There are a few types of PET/CT scans, but the most common is a full-body PET/CT, which evaluates the internal structures from the mid-portion of the skull down to the thigh area ("eyes to thighs"), in combination with a low dose CT scan. This is a hybrid imaging modality; the resulting images acquired from each test are fused using advanced computerized technology to create enhanced images of higher quality. The functional imaging obtained by the PET scan illustrates the spatial distribution of metabolic or biochemical activity in the body, which is more precisely aligned with the anatomic imaging obtained by the CT scan. PET/CT scans provide superior information for evaluating tissues, staging and restaging cancers, and monitoring the effectiveness of cancer treatments. Since cancer cells take in glucose faster than normal tissue, the FDG tracer is a superior radiotracer for evaluating cancerous tissue since FDG is a compound similar in chemical composition to glucose. The FDG accumulates in the body's most metabolically active areas, which helps differentiate between physiologic uptake (healthy tissue) and pathologic uptake (diseased tissue; Katal et al., 2022; Kocak, 2022c).
The FDG radiotracer is administered IV, and the PET/CT scan creates images that show the distribution of the radiotracer throughout the body and determine if abnormalities are present. Highly active cancer cells show higher levels of uptake of FDG, whereas brain cells affected by dementia consume smaller amounts of glucose, indicated by lower FDG uptake. PET scans that utilize amyloid imaging agents can reveal the areas of amyloid plaque in the brain, which may help to diagnose Alzheimer's (Kocak, 2022c; SNMMI, n.d.). An example of the resultant images obtained from a PET/CT scan is displayed in Figure 4, which depicts a healthy brain scan on the left in contrast to a brain tumor depicted on the right.
Figure 4
PET/CT Scan Images of the Brain
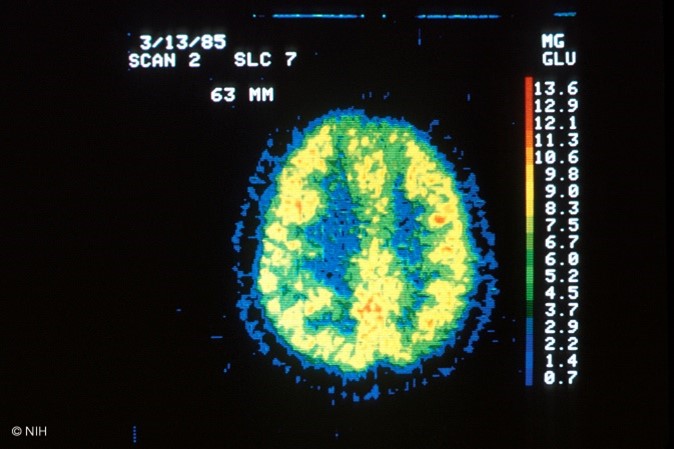
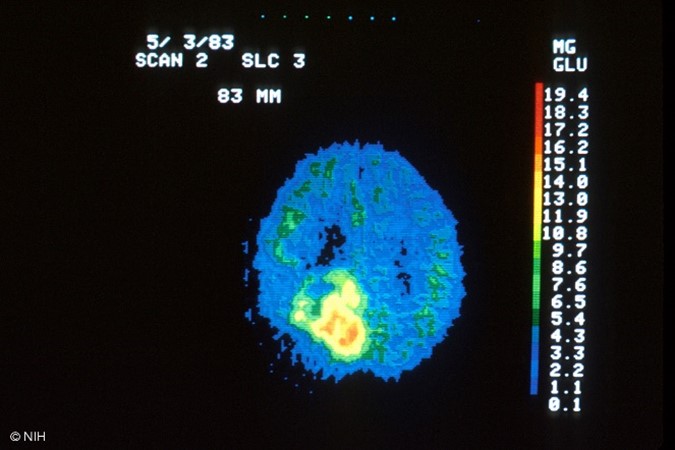
(NCI, 2001a, 2001b)
For 24 hours before the PET/CT scan, patients are advised to avoid strenuous activities such as running or cycling, as these activities can impair the quality of the images obtained. Since the PET/CT scan measures glucose uptake, strenuous activity can increase radiotracer uptake in strained and recovering muscles, increasing the likelihood of false-positive results. Patients are advised to fast for six to eight hours before the scan and follow a low glucose, low carbohydrate diet for 24 hours before the scan. From midnight until two hours before the scan, the patient can drink 12 ounces of water but nothing else. The patient's fasting blood glucose (FBG) will be obtained on the day of the scan, usually through a finger stick. The FBG level should be between 70 and 199 mg/dL for the highest-quality imaging results. If the FBG is too high, the scan will be of poor quality, thereby interfering with the clinical benefit and accuracy of the results. Typically, patients with FBG levels below 70 mg/dL or 200 mg/dL or above will be referred to the ordering provider for glucose management, and their scan will be rescheduled. Patients with underlying diabetes will be given individual instructions based on their diabetes management plan. However, all are advised to avoid taking anti-hyperglycemic medications, such as metformin (Glucophage) or insulin, within four hours of the scheduled scan. If insulin or diabetic medications are taken too close to the FDG injection time, too much FDG will collect in the muscles instead of throughout the tissues where it should flow. If a brain PET/CT is being performed, limiting brain activity before testing by avoiding reading or listening to music while waiting for the scan to begin is essential. The PET/CT scanner resembles a standard CT scanner, as seen in Figure 3. The patient will lie down on the table, usually supine, and should be advised to lay very still. The exam table will move slowly through the scanning ring. The test usually takes 25 to 40 minutes (Memorial Sloan Kettering Cancer Center [MSKCC], 2023; University of California San Francisco Department of Radiology & Biomedical Imaging, n.d.).
Following the scan, patients should be advised that some security equipment can detect radioactivity, setting off radiation alarms, such as radioactive detectors in police cars and airport security. Therefore, most patients are given a card stating they had a test performed with a radioactive tracer and the testing date. Patients should also be advised to limit close contact with infants and pregnant individuals for at least 12 hours following the scan (MSKCC, 2023). The ACOG (2017) recommendations for breastfeeding following nuclear imaging scans do not definitively state a position for or against the practice. Patients are advised to consult with a lactation specialist. In general, women who are breastfeeding may opt to stop breastfeeding for 12 hours following the scan, during which time they can continue to pump and discard the milk (MSKCC, 2023).
Single Photo Emission Computed Tomography (SPECT)
SPECT is another type of nuclear medicine imaging test very similar to PET/CT scans as it integrates CT technology and a radiotracer administered via injection. The primary distinction between PET/CT and SPECT imaging is the type of radiotracer used. For SPECT imagining, the isotopes commonly used are technetium-99m, iodine-123, and thallium-201. In SPECT imaging, the radiotracer stays within the bloodstream rather than being absorbed by tissues and organs. SPECT imaging primarily focuses on areas where blood flows; it is used to evaluate blood flow to surrounding tissues and organs and how the organs function. SPECT studies are most commonly used to diagnose or evaluate heart and brain disorders but may also be used to assess other conditions. Regarding the heart, SPECT imaging can detect blockages within the coronary arteries, damage to the myocardium (heart muscle) resulting from a heart attack, and how well the heart is pumping blood, particularly when stressed. With brain functioning, SPECT studies may be ordered to evaluate for dementia and the location and etiology of a stroke by visualizing how blood flows through veins and arteries in the brain. It can be used to diagnose areas of ischemia (blood deprivation) within the brain following a stroke or as a result of a tumor. These studies are also used in epilepsy for detecting and identifying seizure activity and localizing epileptic foci. A clinician may also order a SPECT scan to evaluate conditions that are non-cardiac or non-neurological such as parathyroid disease, pulmonary embolism, osteomyelitis, or spondylolysis (Yandrapalli & Puckett, 2022).
During a SPECT scan, the patient is usually placed in the supine position on the examination table and asked to lie very still for the duration of the examination. After injecting the radiotracer, a gamma camera rotates around the patient. The device accumulates pictures which are then used to construct 3D images of the radiotracer distribution. The resulting pictures disclose information regarding blood flow and target organ function. Patients are advised to fast for at least 3 hours before the scan, which can take up to 2.5 hours to complete. Patients are instructed to avoid caffeine for at least 12 hours before testing as caffeine can interfere with vasodilatory medications administered during the test. Patients are also advised to discontinue taking phosphodiesterase-3 inhibitors (e.g., cilostazol [Pletal]) at least 48 hours before testing due to the vasodilatory effects. Following the scan, patients are advised to maintain increased oral hydration for about 2 days to flush the radioactive material from the body. Otherwise, there are generally no special discharge instructions (SNMMI, n.d.; Yandrapalli & Puckett, 2022).
Thyroid Scintigraphy and Radioactive Iodine Uptake (RAIU) Test
There are two types of nuclear medicine imaging tests of the thyroid; thyroid scintigraphy, also called the thyroid scan, and the RAIU test. Both scans use a small amount of radioactive iodine, usually in the form of I-123, as the thyroid gland is the only tissue within the body that absorbs and holds onto iodine. The radiation emitted by I-123 is harmless to thyroid cells and can be detected externally through thyroid scanning. Rarely, I-131 may be used with RAIU scans, but I-131 destroys thyroid cells. Due to this, it is commonly reserved for treating thyroid disorders such as overactive thyroid, thyrotoxicosis, and thyroid cancers, which are beyond the scope of this module (ACR, 2019).
The thyroid scan is ordered to acquire information about the thyroid function and evaluate for abnormalities within the gland, such as nodules, masses, or inflammation. According to the ACR (2019), thyroid scans are helpful in, but not limited to, evaluation of the following:
- "size and location of thyroid tissue;
- the cause of overt and subclinical thyrotoxicosis;
- suspected focal masses or diffuse thyroid disease;
- clinical laboratory tests suggestive of abnormal thyroid function;
- function of thyroid nodules detected on clinical examination or other imaging examinations;
- congenital thyroid abnormalities, including ectopia;
- differentiating hyperthyroidism from other forms of thyrotoxicosis" (ACR, 2019, p.2).
When undergoing a thyroid scan, the I-123 is either injected into a vein within 30 to 60 minutes of the scan or administered orally as a pill or liquid. With oral administration, the I-123 must be given approximately 24 hours before the scan, allowing the radioactive iodine to reach and saturate the thyroid gland. The oral route is generally preferred for patients who undergo both the thyroid and the RAIU scans, as it can be used for both tests and does not require a second dose of the radiotracer. The thyroid scan is painless, and the patient is usually positioned lying flat (supine) on an examination table with their head tilted back to extend the neck. A gamma camera will take thyroid images from at least three different angles, and the patient will be asked to lie very still. It takes about 30 minutes to complete a thyroid scan (Iqbal & Rehman, 2022).
The RAIU scan is performed to evaluate the thyroid gland's function or determine the etiology of an overactive thyroid gland (hyperthyroidism). It may also be used to plan treatment for thyroid cancer patients. The RAIU uses a specialized probe to measure how much tracer the thyroid gland absorbs from the blood. In most cases, the RAIU scan is performed alongside the thyroid scan to show if the radiotracer is evenly spread in the gland (ACR, 2019). According to the ACR, while the RAIU scan does have overlapping indications with the thyroid scan, it is considered most useful in the following situations:
- "differentiating hyperthyroidism from other forms of thyrotoxicosis (such as subacute or chronic thyroiditis and thyrotoxicosis factitial [exogenous thyrotoxicosis]);
- assessing the necessity and calculating iodine-131 sodium iodide administered activity for patients to be treated for hyperthyroidism" (ACR, 2019, p.2).
Agents containing iodine can decrease iodine uptake in the thyroid gland, leading to inaccurate test results. Iodine is hidden in many commonly used supplements, over-the-counter agents, and certain prescription medications. Therefore, before the test, a comprehensive medication reconciliation should be performed. Patients must be informed to discontinue thyroid hormones, anti-thyroid drugs, and any other medication or dietary supplement containing iodine. Each medicine or supplement has a specified period in which it should be discontinued before the scan. For example, levothyroxine (Synthroid) is a thyroid hormone that must be stopped for four to six weeks before the scan. In contrast, iodine-containing cough syrups should be discontinued two weeks before the scan (ACR, 2019). Other iodine-based agents that need to be avoided include but are not limited to the following:
- iodized salt
- multivitamins
- amiodarone (Pacerone)
- kelp (algae seaweeds)
- IV iodinated contrast agents (ICAs)
- sulfonamides
- methimazole (Tapazole)
- high-dose corticosteroids (ACR, 2019; Iqbal & Rehman, 2022)
In the one to two weeks leading up to the radioactive iodine administration, patients are generally advised to consume a low-iodine diet, avoiding the highest sources of dietary iodine, including salt, grains, cereals, fish, poultry, and milk products (American Thyroid Association [ATA], n.d.). The RAIU scan requires the administration of radioactive iodine in liquid or capsule form. The RAIU scan occurs at two distinct time points: usually four to six hours following radiotracer administration and then again at 24 hours post-administration. During an RAIU test, the patient is generally seated upright, and a small device called a radioactive detector (uptake probe) is placed against the patient's neck. The uptake probe takes measurements of radioactive iodine uptake, and a gamma camera records pictures of the thyroid gland. Both instruments work to detect and record the distribution of the radioactive material within the thyroid. The RAIU test usually takes several minutes. Following the test, patients should be advised that most radioactive material is cleared from the body within one to two days. No special precautions must be taken as I-123 is harmless to thyroid cells. The ACR states it is safe to use radioactive iodine in patients who report iodinated contrast allergies or seafood allergies, as the reaction is to the compound containing iodine and not the iodine itself (ACR, 2019; RadiologyInfo.org, 2021b).
Skeletal Scintigraphy (Bone Scan)
Skeletal scintigraphy, also called a bone scan, is a type of nuclear medicine imaging test that uses a small amount of radioactive tracer to diagnose and evaluate various bone disorders and conditions (Adams & Banks, 2022). A bone scan is performed to assess for several types of conditions, such as:
- bone fractures, including stress, occult, accidental, and non-accidental
- primary bone masses or tumors (benign or malignant)
- metastatic bone neoplasms
- underlying bone pain that is otherwise unexplained or non-responsive to conservative treatments, such as the case with chronic low back pain or complex regional pain syndrome
- tumor-like conditions such as Paget's disease
- infections such as osteonecrosis
- complications from orthopedic hardware or prosthetic joints
- congenital or developmental anomalies
- fibrous dysplasia (ACR, 2021; Adams & Banks, 2022)
A bone scan is also routinely used to determine if cancer has spread to the bones and to monitor response to cancer treatment over time. A radiotracer such as Technetium-99m is injected into the vein and travels through the bloodstream, emitting radiation as gamma rays. These are detected by a specialized gamma camera and fused with computer images to derive an overall picture of the bones at multiple points as demonstrated in Figure 5 (Adams & Banks, 2022; RadiologyInfo.org, 2022a).
Figure 5
Bone Scan

A bone scan can detect changes at the molecular level, thereby allowing the early detection of bone disorders, even at the earliest stages of the disease. Abnormal areas within the bone will take up more or less of the radiotracer, producing brighter or darker areas in the resulting images (RadiologyInfo.org, 2022a).
In preparation for a bone scan, patients should be counseled to avoid bismuth-containing medications such as bismuth subsalicylate (Pepto-Bismol, Kaopectate) for at least four days before the scan, as these medications can interfere with the results. In addition, patients should be screened for x-ray tests utilizing barium contrast material within the last four days, which can also skew the results. The radiotracers used with bone scans take a few hours to circulate through the body and bind to the bones to produce the highest-quality images (RadiologyInfo.org, 2022a).
Patients should be advised that there will be a two- to four-hour period between the injection administration and the scan. During this time, patients will need to consume several glasses of water (usually four to eight) to facilitate the removal of any excess radiotracer from the body. Patients will also be instructed to empty their bladder before the scan, as any residual tracer in the bladder can obscure the view of the underlying pelvic bones. Patients will be asked to lie still on the examination table during the test. A total body bone scan usually takes about one hour to complete. Following the scan, patients may resume normal activities and are advised to increase oral hydration for one to two days to help facilitate the removal of any residual radiotracer circulating in their system. It generally takes 48 hours for all the radioactive tracer material to be excreted from the body (RadiologyInfo.org, 2022a).
Dual-Energy X-ray Absorptiometry (DEXA, DXA) Scan
A bone densitometry scan is also called a DXA scan. While it is a nuclear medicine test that uses a small amount of ionizing radiation to generate images of bony structures, it vastly differs from a bone scan. A DXA scan evaluates the bone mineral density (BMD), or the health and strength of the bones, and assesses osteopenia or osteoporosis and fracture risk. According to the World Health Organization (WHO), the gold standard bone density test is a DXA scan of the central skeleton, which includes the hip and lumbar spine. BMD is most commonly measured at the spine, hip, and wrist levels. The degree of bone loss is calculated and classified according to defined diagnostic criteria (CDC, 2022a).
Osteoporosis is a chronic, systemic disease characterized by low BMD, bone weakening, and deterioration of bone tissue and architecture. Globally, over 200 million people have osteoporosis leading to approximately 9 million fractures annually. Osteoporotic bones are brittle and porous, heightening the risk of fracture. It is referred to as a silent disease because the loss of bone mass is not painful, and there are generally no warning signs or symptoms preceding bone fracture. The vast majority of people do not know they have osteoporosis until they develop an acute fracture or broken bone, which is the hallmark of the disease. Fractures can occur in any bone within the body but most commonly occur in the hip bones, vertebrae, and wrist. Osteopenia is the precursor condition to osteoporosis and is characterized by a lower-than-normal BMD that is not severe enough to meet the criteria for osteoporosis. People with osteopenia are at higher risk for developing osteoporosis, but when identified early through screening with a DXA scan and appropriate action is taken, progression to osteoporosis can be successfully averted (National Institute of Arthritis and Musculoskeletal and Skin Diseases [NIAMS], 2022; Porter & Varacallo, 2022).
The degree of bone loss is calculated and classified according to defined diagnostic criteria. The DXA scan is a quick, noninvasive, and painless test. The patient is instructed to lie or sit down for less than 10 minutes while the machine scans the body. The test exposes the patient to a small amount of radiation, but less than the amount associated with a chest x-ray (ACOG, 2022). DXA test results are reported as a T-score for each site measured, comparing the patient's BMD level to that of a healthy young adult with ethnicity- and gender-matched controls. The WHO separates those T-scores into four categories: normal, low bone mass (osteopenia), osteoporosis, and severe or established osteoporosis. A T-score of 0 indicates that the BMD is equal to that of a healthy young adult, a negative T-score indicates that the bones are thinner than average, and a positive T-score denotes that the bones are stronger than average. The difference between a patient's BMD and the normal range is measured in units referred to as standard deviations. The more standard deviations below 0, denoted by negative numbers, the lower the BMD, the more severe the osteoporosis, and the higher the fracture risk. The WHO classifies osteoporosis as a BMD that is 2.5 standard deviations below normal. Treatment is usually recommended to prevent fractures when the T-score is -2.5 or lower. Table 5 defines T-scores and their corresponding BMD level (Camacho et al., 2020; NIAMS, 2018).
Table 5
WHO T-Score Interpretation
T-Score | Interpretation |
≥ -1.0 | Normal bone |
-1.0 to -2.5 | Osteopenia |
≤ -2.5 | Osteoporosis |
≤ -2.5, plus 1 or more osteoporotic fractures | Severe or established osteoporosis |
(Camacho et al., 2020; NIAMS, 2018)
Multigated Acquisition Scan (MUGA)
A MUGA scan may also be called radionuclide ventriculography (RNV), radionuclide angiography (RNA), or gated equilibrium radionucleotide angiography (ERNA), and it is a type of nuclear imaging test that evaluates how well the heart is functioning, particularly the left ventricular ejection fraction (LVEF). The LVEF measures the amount of blood pumped out of the heart with each contraction and is expressed as a percentage. The normal LVEF in an adult is 50% to 75%. This test may be performed for various reasons but is most commonly ordered as part of a cardiology work-up for chest pain, in follow-up to an abnormal electrocardiogram (EKG) or echocardiogram scan, monitoring patients with heart failure, and in evaluating the cardiac function of patients diagnosed with chronic obstructive pulmonary disease (COPD). MUGA scans are also used for monitoring patients undergoing chemotherapy that is potentially cardiotoxic, chest wall radiation, or other cancer treatment regimens that can impair heart function; however, MUGA scans are limited in the assessment of cardiac function in these individuals (Odak & Kayani, 2022).
During the MUGA scan, small electrodes are placed on the patient's chest, arms, and legs, similar to an EKG, and these are used to track the patient's heartbeat and heart rhythm during the test. This is necessary since MUGA scans require tracking R wave progression to recognize when to begin data collection. The patient's blood is obtained, mixed with the radioactive tracer, and administered IV. After allowing the radioactive tracer 15 to 20 minutes to circulate, a gamma camera takes images of the heart at designated time points during each heartbeat. A MUGA scan may be performed at rest, where the patient lies on the table while the gamma camera photographs the heart. Alternatively, it may be performed as an exercise scan or stress test, in which the patient walks on a treadmill or rides a stationary bicycle to obtain peak activity level and is subsequently asked to stop and lie on the table. At the same time, the gamma camera takes pictures of the heart. A pharmacologic agent is administered to induce cardiac stress if the individual cannot complete physical activity. The test takes approximately one to two hours, and patients can generally resume normal activities following the test. After the scan, patients are advised to drink plenty of water to help flush the radioactive materials through the renal system. MUGA scans have become less common in clinical practice since echocardiograms are more readily available (Odak & Kayani, 2022).
Diagnostic Imaging Tests without Radiation Exposure
Magnetic Resonance Imaging (MRI)
MRI is a widely utilized diagnostic imaging modality, with an estimated 108 MRIs performed per 1,000 individuals in the US annually (Organisation for Economic Co-operation and Development, 2023). MRIs are distinct from other forms of diagnostic imaging as they do not use x-rays or ionizing radiation and are considered a very safe imaging test. Instead, MRIs utilize strong EM fields, magnetic field gradients, and radio waves and are essentially giant magnets. They can vary in strength, measured in a unit called tesla (T). Most modern MRI scanners are 1.5 to 3T. In context, an MRI of 3T strength is about 60,000 times stronger than the earth's magnetic field. Over the last few years, there has been a push to improve and expand MRI imaging capabilities. The current state-of-the-art neuroimaging MRI scanners have a magnetic field strength of up to 7T. An electric current creates a temporary magnetic field within the patient's body during an MRI. Radio waves are sent from and received by a transmitter and receiving device within the machine. These signals are used to generate images of the scanned area of the body. The signal in an MRI image comes mainly from the protons in fat and water molecules within the body. MRI is at least equal but often superior to a CT scan at identifying the difference between normal and abnormal soft tissue. MRIs can be performed to image nearly any body part; each scan follows a specific protocol depending upon the clinical concern and may be performed without or without contrast administration. Gadolinium-based contrast agents (GBCAs) are rare earth metals administered IV to enhance the contrast of the MRI images. MRI contrast injections are distinct from CT scan contrast agents, and patients who have CT contrast allergies can typically safely tolerate GBCA injections (FDA, 2018; Ibrahim et al., 2022; Vachha & Huang, 2021).
MRI scans are the preferred diagnostic imaging test for numerous diseases and disorders and are considered one of the most frequently performed intracranial and spinal cord imaging tests. Different MRI types can be used to diagnose aneurysms, multiple sclerosis, strokes, herniated discs, fractures, biliary duct abnormalities, and tumors. While standard MRI scans are superior for evaluating soft tissues and organs, they also have clinical benefits regarding imaging of the heart and blood vessels, including structural abnormalities in the aorta, the thickness of the heart walls, and damage caused by heart disease. MRI is used extensively in orthopedics to evaluate bone and joint conditions, such as torn ligaments or cartilage. MRI may also be the preferred screening modality in women with dense or fibrotic breasts instead of mammography due to the superior evaluation of dense breast tissue and can outline the extent of the breast cancer after a positive biopsy (Centers for Medicare & Medicaid Services [CMS], 2018; Ibrahim et al., 2022; Kocak, 2022b).
Patients undergoing MRI scans of the abdomen or soft tissue pelvic structures are usually advised to remain NPO for six hours before the scan. Before an MRI of the prostate or to evaluate for a vaginal/rectal fistula, a bowel preparation with an enema may be necessary. However, no special instructions must be followed for all other MRIs the day before the scan. Patients are advised to lie very still on the table, as motion can create artifact and reduce the clarity of the images. The table slides into the MRI machine, which resembles a CT scanner, as demonstrated in Figure 3. However, an MRI scanner is deeper and narrower than a CT scanner. Due to this, there is a heightened risk for claustrophobia and anxiety in some patients, who may require a sedative before the scan to ensure relaxation and manage anxiety. Patients should be advised that they will hear loud thumping or banging throughout the examination. Patients should be reassured that these are harmless sounds produced by the MRI magnets, and some patients will be given earplugs or headphones to minimize the noise, which can reach up to 120 decibels. An MRI scan can take anywhere from 15 to 45 minutes, depending on the part of the body being imaged; however, some can last longer than 60 minutes (Ibrahim et al., 2022; NIBIB, n.d.).
While there is no risk of radiation exposure with MRIs, there are risks for potential injury. Due to the strong EM fields, the MRI machine can propel magnetic objects toward the machine's center at dangerous speeds, including medically implanted hardware, such as cardiac pacemakers and infusion pumps. The radiofrequency field can also cause tissue heating and burning, particularly in the presence of implanted devices, which can also heat internally. All patients must undergo screening evaluations before testing to assess for the presence of metal or any implanted hardware. The FDA has received reports of serious adverse events associated with implantable pumps in the MRI setting, such as pump malfunction, including bolus dose, overdose, underdose, or pump failure. Some of these injuries have been fatal. According to the FDA, only implantable infusion pumps labeled "MR Conditional" may be safely used within an MRI environment and only under the specified conditions of safe use as outlined by the individual device manufacturer. During the screening, patients must also be asked if they have ever welded without eye protection or had any facial injury with metal. If the patient responds yes, an orbital x-ray must be taken to ensure no hidden metal is in the orbits before the MRI (Ghadimi & Sapra, 2022; FDA, 2021).
Given the strength of the EM fields, MRI use has many contraindications. These contraindications are categorized as absolute or relative. Devices and objects are designated as MR safe (safe in all environments), MR conditional (safe if specific criteria are met based on the object or device), or MR unsafe (known safety risk). It should be noted that some of these may be compatible at 1.5T or 3T only. Clinicians must refer to a certified MRI safety website or the manufacturer's instructions for the device when determining safety and compatibility with MRI imaging (ACR, 2022; FDA, 2021; Ghadimi & Sapra, 2022). The list below comprises the most common absolute contraindications of MRI; however, these contraindications change as new advancements are made:
- cardiac implantable electronic devices (CIEDs) such as pacemakers, implantable cardioverter defibrillators (ICDs), cardiac resynchronization therapy (CRT) devices, implantable loop recorders (ILRs), and implantable cardiovascular monitors (ICMs); newer devices may be designated as MR conditional
- metallic intraocular foreign bodies
- implantable neurostimulation systems
- cochlear implants; some may be used with a 1.5T MRI scanner after the battery is removed
- catheters with metallic components (Swan-Ganz catheter)
- metallic fragments such as bullets, shotgun pellets, or shrapnel
- cerebral artery aneurysm clips
- magnetic dental implants
- tissue expanders
- medication patches, hearing aids, body piercing, external drug delivery pumps (insulin pumps), and artificial limbs are all contraindicated and must be removed before the MRI (ACR, 2020; Ghadimi & Sapra, 2022)
Several relative contraindications must be considered before any MRI scan, as the specific patient situation must be evaluated with caution to confirm it is safe to proceed with the exam (Ghadimi & Sapra, 2022). Relative contraindications include patients presenting with any of the following:
- coronary and peripheral artery stents
- non-plastic airway stents or tracheostomy (plastic tracheostomies are safe to proceed with the MRI, but if the composition of the tracheostomy is unknown or unsafe, it must be changed to a plastic source before the MRI)
- intrauterine devices (IUDs) require investigation and confirmation of the make and model; those that are unknown are only acceptable with 1.5T MRI scanners
- ocular prosthesis
- stapes implants
- surgical clips or wire sutures
- certain types of prostheses (joint, penile, etc.)
- if the patient had a colonoscopy procedure within the previous eight weeks and endoscopy clips were used or placed during the procedure, the scan must be postponed (Ghadimi & Sapra, 2022)
Additional consideration must be given to the following situations before performing the MRI scan:
- patients with programmable shunts must be informed that they have to reprogram their shunt with their provider following the MRI
- patients with inferior vena cava (IVC) filters that are of unknown composition must wait six weeks following implantation and can only be scanned with a 1.5T MRI scanner
- patients with Harrington rods (a stainless-steel surgical device) can only undergo an MRI on 1.5T scanners
- tattoos should be older than six weeks, and ice packs or padding should be used against any tattoo that is in contact with the bore of the scanner or the MRI coil, and patients must be educated to immediately report any warm sensation that develops around the tattoo site (Ghadimi & Sapra, 2022)
Ultrasound
Ultrasound is a safe, noninvasive imaging modality that uses sound waves to generate images of internal body structures. Also referred to as sonography, ultrasound does not use x-rays or ionizing radiation and can be used for diagnostic and therapeutic indications. Ultrasound images are obtained by placing a small transducer (probe) and ultrasound gel on the skin. The transducer produces sound waves at very high frequencies, which exceed the threshold of human hearing. These high-frequency sound waves travel from the probe through the gel and into the body. The reflections of the sound waves off the structure being evaluated are used to generate images on a computer. Images are captured in real-time, which allows for evaluating the structures and movement of the body's internal organs, including blood flow through vessels. Transducers may be placed external to the body on the skin, such as with a transabdominal fetal ultrasound, which is demonstrated in Figure 6. Some transducers can also be placed directly inside the body via the vagina, GI tract, or blood vessels to optimize image quality. In these instances, a transducer is placed inside the vaginal canal of a nonpregnant female to enhance the visualization of the uterus and ovaries (FDA, 2020).
Figure 6
Fetal Ultrasound
Conventional ultrasound displays images in flat sections of the body. However, technological advancements have led to the development of an ultrasound that allows formatting sound wave data into 3-D images. The most widespread use of a 3-D ultrasound is to assess a developing fetus, and the distinction between the resultant images generated from a conventional sonogram versus a 3-D sonogram is demonstrated in Figure 7. A 4-D ultrasound can also be used to assess fetal characteristics. A 4-D ultrasound is a 3-D ultrasound that is put into motion (FDA, 2020).
Figure 7
Conventional Sonogram (left) Compared to 3-D Sonogram of Fetus (right)
Ultrasound can be performed for diagnostic or therapeutic reasons. Therapeutic ultrasound interacts with tissues in the body to modify or destroy them. For example, therapeutic ultrasound may be used for ablative functions such as dissolving blood clots or delivering drugs to specific locations within the body. Diagnostic ultrasound is used to evaluate the internal organs and structures, movement, the velocity of tissue or blood, and any other abnormalities. Ultrasound may also be used during surgery to help guide the placement of a catheter or other medical device or to guide biopsies. An ultrasound-guided needle biopsy is a standard procedure for evaluating a breast mass for breast cancer (RadiologyInfo.org, 2022c).
Doppler ultrasound is a specialized technique that evaluates blood flow through the arteries and veins in the body. This type of ultrasound uses the Doppler effect to assess the movement of red blood cells (Kocak, 2022e). Doppler ultrasound is used to evaluate the following:
- blockages to blood flow (such as thrombosis, deep vein thrombosis, or other blood clots)
- narrowing of vessels (such as narrowing of the carotid arteries, which increases the risk of stroke)
- vascular tumors or masses
- congenital vascular malformations
- reduced or absent blood flow to organs, such as the testes or ovaries (Kocak, 2022e; RadiologyInfo.org, 2022c)
There is a wide array of indications for ordering a diagnostic ultrasound. Ultrasound can evaluate pain, swelling, or infection symptoms and can be performed safely on nearly every body system (Radiologyinfo.org, 2022c). Below is a list of some of the most common body parts imaged by ultrasound and possible indications for ordering a diagnostic ultrasound, but it is not a comprehensive list of indications:
- gallbladder and bile ducts (gallstones and biliary tract obstruction)
- heart and blood vessels (patency of blood blow, evaluate for abdominal aortic aneurysm (AAA), carotid arteries for narrowing)
- liver (cirrhosis, cancer or metastatic tumors, infarction)
- spleen (splenomegaly)
- pancreas (pancreatitis, tumor)
- kidneys, bladder, ureters (masses, cancer, patency, hydronephrosis)
- uterus (fibroids, fetus in pregnant women)
- ovaries (cysts, tumors)
- thyroid and parathyroid glands (masses, enlargement, nodularity)
- scrotum and testes (testicular torsion, swelling, masses, blockage in blood flow; Kocak, 2022e; RadiologyInfo, 2022c)
In some cases, ultrasound imaging may also use contrast agents for enhancement. Microbubble contrast helps assess blood perfusion in organs, thrombosis, abnormalities in the heart, liver, or kidney masses, or when evaluating inflammatory activity in IBD. Most ultrasound imaging requires little to no special preparation before the exam. However, ultrasounds ordered to assess the abdominal structures, such as the gallbladder, require patients to fast for four to eight hours before the scan. For ultrasounds ordered to evaluate the bladder and many prenatal ultrasounds, the patient may be asked to drink four to six glasses of water before their exam and avoid urinating so that the bladder is full and distended for optimal viewing (NIBIB, 2016).
While ultrasound is considered a safe and painless medical imaging modality that can be used in patients of all ages, it has limitations. Air and gas can disrupt or interfere with the ultrasound waves, obscuring images. Due to this, ultrasound does not help evaluate air-filled organs and structures such as the bowels and lungs. Ultrasound waves are less effective in obtaining high-quality images in patients with excess fatty tissue, as the sound waves weaken as they pass deeper into the body. Further, ultrasound cannot penetrate bone but helps identify infections surrounding the bones (RadiologyInfo.org, 2022c).
Echocardiogram
An echocardiogram is an ultrasound of the heart. It is a commonly used cardiac diagnostic imaging test considered safe and effective and does not risk radiation exposure. Utilizing the same sound wave technology outlined in the above section, an echocardiogram provides critical information about the heart's functioning and structures, including the valves, chambers, walls, and blood vessels. An echocardiogram may be ordered to evaluate further symptoms of dyspnea, angina (chest pain), as well as to detect any underlying congenital abnormalities. An echocardiogram can provide information regarding the size, shape, thickness, and movement of the heart, including pericarditis (inflammation of the lining of the heart), stenosis (narrowing of the heart valves), and regurgitation (leaking of blood backward through the heart valves). Like a MUGA scan, an echocardiogram provides information regarding the heart's systolic function and diastolic filling patterns. The test is usually performed with the patient lying on their back or left side. Some patients will also be connected to an EKG to monitor their heartbeat and heart rhythm during the exam. The transducer and gel are applied to the chest and provide real-time images of the heart in motion. Generally, no risks are associated with the test, and no special preparation instructions (American Heart Association [AHA], 2022; Cascino & Shea, 2022).
Transesophageal echocardiogram (TEE). A TEE is a specialized type of echocardiogram that involves the placement of a small, thin tube down into the esophagus to provide a more enhanced evaluation of the posterior heart chambers and structures than what is provided by a standard (transthoracic) echocardiogram. It is indicated for use in patients with unexplained hemodynamic disturbance and suspected valvular disease. A tiny transducer is placed at the end of the thin tube and advanced to the midline hypopharynx with the transducer facing anteriorly. The sound waves produce detailed images of the heart, which are translated to a video screen. This is usually performed when additional information is needed following a transthoracic echocardiogram. There are no associated risks of radiation exposure, as ultrasound-based technology is utilized. Patients are typically advised to remain NPO for at least six hours before the scan and are given a sedative immediately before the test to promote relaxation. Some patients report a mild sore throat for one to two days following the test, but generally, there are no reported complications (Omerovic & Jain, 2023; O'Rourke et al., 2022).
Contrast Media in Medical Imaging
For various types of diagnostic radiology tests, substances called contrast media are administered to temporarily change the appearance of body structures on radiographic images to improve the visibility of specific body structures. Contrast media alters the absorption of the x-rays to enhance the contrast (or difference) between distinct anatomical structures, thereby generating a higher-quality image of the underlying pathology. Several types of contrast agents are generally categorized as either positive or negative, as shown in Table 6. Positive contrast media agents include barium compounds, gadolinium, and iodinated agents, which absorb more x-rays than surrounding tissue due to their high density. Positive contrast media leads to increased attenuation of x-rays and appears white on the resulting images. Negative contrast media agents, such as air, oxygen, carbon dioxide, and nitrous oxide, are primarily gaseous, which absorb fewer x-rays than surrounding tissue due to their low density. Negative agents lead to decreased attenuation of x-rays and appear black or gray on the resulting images (ACR, 2022; RadiologyInfo.org, 2022b). In 2022, Congress added Section 3621 to the Consolidated Appropriations Act of 2023, requiring the FDA to regulate all contrast agents and radioactive drugs as drugs, not medical devices (117th Congress, 2022).
Table 6
Comparison of Positive and Negative Contrast Media
| Positive contrast media (Radiopaque) | Negative contrast media (Radiolucent) |
Attenuation | Increased | Decreased |
Color on imaging | White | Black or gray |
Common types | Iodinated agents, barium compounds, gadolinium | Gaseous agents (air, oxygen, carbon dioxide, and nitrous oxide) |
(ACR, 2022; RadiologyInfo.org, 2022b)
Contrast agents can be administered orally, IV, intra-arterially (IA), or instilled directly into body cavities such as the urinary or GI tracts, including rectally, via an enema. Contrast can also be administered intrathecally, although this administration route is less common. While contrast agents are used to enhance medical images, and their value is widely recognized, they are still considered pharmaceutical agents and are not without risks. Each type of contrast agent has unique properties, side effect profiles, and risks and is subject to specific monitoring and clinical decision-making regarding individual patient factors. Adverse effects from contrast media agents can range from minor side effects to life-threatening clinical emergencies. Therefore, clinicians must be well versed on the potential risks these agents pose before prescribing them and ascertain a keen awareness of the signs of impending adverse reactions as a means to respond promptly and effectively to reduce morbidity and mortality (ACR, 2022; RadiologyInfor.org, 2022b).
Iodinated Contrast Agents
Iodine is a naturally occurring chemical element that is found in many types of contrast agents. Iodine-based contrast agents (ICAs) enhance x-ray and CT images and are most commonly administered by injection through veins, arteries, or other body cavities. ICAs are the most widely utilized category of contrast agents, with millions of doses of ICAs administered each year in the US. Dating back to their initial development in the 1920s, their use has expanded significantly over the last several decades, as well as their composition and chemical structuring. There are a few different types of ICAs available, and they are grouped according to their osmolarity (high, low, or iso-), ionicity (ionic or nonionic), and the number of benzene rings (monomer or dimer; ACR, 2022; Cutler, 2016; Spampinato et al., 2017).
Ionic compounds dissociate (dissolve) into positively and negatively charged particles when entering a solution, whereas nonionic compounds do not dissociate. Osmolality is the concentration of particles dissolved in a fluid, where higher osmolality denotes more particles in the serum. Lower osmolality means the serum is more diluted and contains fewer particles. Osmolarity is the concentration of a solution expressed as the total number of solute particles per liter. The main difference between osmolarity and osmolality is that osmolarity is a measure calculated considering the volume of a solution, whereas osmolality is computed considering the mass of a solution. These concepts are essential to understanding contrast agents, as the osmolality of the agent affects the incidence of side effects. Initial (first-generation) ICAs were ionic compounds with high osmolality. They had toxic profiles and were accompanied by a high rate of adverse events and allergic reactions in patients. This led to the development of safer, nonionic, and lower osmolality ICAs. Despite this, high osmolality ICAs are still used for GI and cystourethral administration. Currently, nonionic low or iso-osmolar preparations are considered the gold standard for IV contrast injections and are used nearly exclusively. Nonionic contrast media cause less discomfort (burning) with intravascular administration and are associated with fewer adverse reactions when compared with ionic agents. Further, low-osmolar contrast is associated with significantly lower acute allergic and adverse reaction rates than high-osmolar agents (McCance & Huether, 2019; Murphy, 2023b; Spampinato et al., 2017). The four classes of iodinated contrast media are grouped according to ionization, and an example of each type is listed in Figure 8.
Figure 8
Four Groups of Iodinated Contrast Media

- (Murphy, 2023b; Spaminato et al., 2017)
While the currently utilized nonionic and low-osmolar or iso-osmolar ICAs are considered safe and effective when administered correctly, significant risks and clinical considerations must be contemplated before prescribing (ACR, 2022; Davenport et al., 2020). Clinicians must balance the potential risks of contrast media with the diagnostic benefits, which are premised on a multitude of factors, including:
- the probability and necessity of an accurate diagnosis
- alternative methods of diagnosis
- risk of misdiagnosis
- expectations about kidney function recovery
- allergic reaction risk and risk for other adverse reactions (Davenport et al., 2020)
One of the most concerning and well-established risks of ICAs is contrast-induced acute kidney injury (CI-AKI) in patients with impaired renal function. This condition is characterized by worsening renal function within 48 hours of IV contrast administration. The ACR and National Kidney Foundation (NKF) published a consensus statement on using IV contrast media in patients with kidney disease. The statement reveals that the risk for CI-AKI is uncertain primarily due to a lack of control groups sufficient to separate CI-AKI (AKI caused by the contrast media) and contrast-associated AKI (CA-AKI) or post-contrast AKI (AKI coincident to contrast media administration). The consensus statement discloses concerns that the risk for CI-AKI has been overstated and does not apply to all populations; however, it offers specific recommendations based on risk factors and the staging of the patient's underlying renal disease (Davenport et al., 2020; Rudnick & Davenport, 2023). Table 7 displays the Kidney Disease Improving Global Outcomes (KDIGO) staging criteria for AKI and chronic kidney disease (CKD) for clinical decision-making regarding IV ICAs. These guidelines are being updated, and the newest version will be released in 2023 at the end of quarter 2.
Table 7
KDIGO AKI and CKD Staging
KDIGO AKI Staging | ||
Stage | Serum creatinine (sCr) criteria | Urine output |
1 | 1.5 – 1.9 times baseline serum creatinine OR Increase in sCr ³ 0.3 mg/dL (³ 26.5 mmol/l) | < 0.5 mL/kg/hr for 6-12 hours |
2 | 2.0-2.9 times baseline sCr | < 5.0 mL/kg/hr for ≥ 12 hours |
3 | 3.0 times baseline sCr OR Increase in sCr ³ 4.0 mg/dL (³ 353.6 mmol/l) OR Initiation of kidney replacement therapy OR Decrease in eGFR to < 35 mL/min/1.73 m2 (for patients < 18 years old) | < 0.3 mL/kg/hr for ≥ 24 hours OR Anuria for ≥ 12 hours |
KDIGO CKD Staging | ||
Grade | GFR criteria | |
Grade 1 | ³ 90 mL/min/1.73 m2 with markers of kidney damage | |
Grade 2 | 60-89 mL/min/1.73 m2 with markers of kidney damage | |
Grade 3a | 45-59 mL/min/1.73 m2 | |
Grade 3b | 30-44 mL/min/1.73 m2 | |
Grade 4 | 15-29 mL/min/1.73 m2 | |
Grade 5 | < 15 mL/min/1.73 m2 | |
(Davenport et al., 2020; Kidney International, 2012)
Following a thorough review of several extensive, well-controlled research studies, the ACR and NKF report the following findings:
- CA-AKI
- the risk for CA-AKI increases with each stepwise increase in the CKD stage, with risk percentages as follows:
- 5% risk with eGFR ³ 60
- 10% risk with eGFR 45-59
- 15% risk with eGFR 30-44
- 30% risk with eGFR less than 30 mL/min/1.73 m2
- risk factors for CA-AKI include: eGFR, diabetes mellitus, exposure to other nephrotoxic agents, hypotension, hypovolemia, congestive heart failure, and impaired kidney perfusion
- there was no clinically significant difference in the risk of CA-AKI between types of contrast administered
- the risk for CA-AKI increases with each stepwise increase in the CKD stage, with risk percentages as follows:
- CI-AKI
- the risk of CI-AKI is cited as substantially less than CA-AKI; the authors caution that the actual risk remains undetermined in those patients with severe kidney disease; several large controlled observational studies have demonstrated no evidence of CI-AKI regardless of the CKD stage
- the risk percentages of CI-AKI are as follows:
- 0% risk with eGFR ³ 45
- 0-2% risk with eGFR 30-44
- 0-17% risk with eGFR less than 30 mL/min/1.73 m2
- there are limited studies linking patient-related factors to CI-AKI, and the primary risk factor was found to be eGFR, without any other risk factors confirmed
- no studies have directly compared the risk for CI-AKI between the various types of contrast, and there is no clinically significant difference in risk based on the type of contrast use (Davenport et al., 2020)
Table 8 summarizes some of the key recommendations for clinicians, as put forth in the consensus statement for IV ICA in patients with CKD and those at risk for AKI.
Table 8
Guidelines for IV ICA in Patients with AKI/CKD
Recommendations |
|
|
|
|
|
|
Prophylaxis |
|
(Davenport et al., 2020; Rudnick & Davenport, 2023)
Patients with renal disease are not the only ones at risk for complications from ICAs. Patients with type II diabetes, or non-insulin-dependent diabetes mellitus (NIDDM), taking metformin (Glucophage) and other biguanides are at risk for lactic acidosis. Lactic acidosis is characterized by a buildup of lactic acid in the bloodstream (greater than 2 mmol/L), which can lead to severe symptoms, including extreme exhaustion, muscle cramps, body weakness, abdominal pain, diarrhea, headache, jaundice, and breathing changes. Since the kidneys are primarily responsible for excreting metformin (Glucophage), patients taking this medication with underlying renal insufficiency are at heightened risk for this severe complication (ACR, 2022; FDA, 2016). Therefore, the FDA recommends the discontinuation of metformin (Glucophage) and metformin-containing products 24 hours before ICA administration in the following patient situations:
- in patients with an eGFR less than 30 mL/minute/1.73 m2 not undergoing hemodialysis
- patients on hemodialysis that make more than 100 mL of urine per day
- in patients with a history of liver disease, alcoholism, or heart failure
- in patients who will be administered intra-arterial ICA (ACR, 2022; FDA, 2016)
According to the ACR (2022), patients taking metformin (Glucophage) who have no evidence of AKI and have an eGFR of 30 mL/min/1.73 m2 or less do not need to discontinue metformin (Glucophage) before or after ICA administration. There is no obligatory need to re-evaluate the patient's renal function following the test or procedure. However, for patients with AKI or severe CKD (stage 4 or 5), metformin (Glucophage) should be temporarily discontinued at the time of or before the procedure. The FDA (2016) and ACR (2022) similarly concur that for patients who meet the criteria to withhold the metformin (Glucophage), their renal function (eGFR) must be reassessed 48 hours after the imaging procedure and before resuming the metformin (Glucophage). Further, patients with severe cardiac disease, such as symptomatic congestive heart failure, angina, pulmonary hypertension, cardiac arrhythmias, or severe but compensated cardiomyopathy, may also be at risk of a cardiac event. While the ACR (2022) determines that these patients are at modestly increased risk, they do not advise restricting ICAs solely based on the patient's cardiac status. However, the risks and benefits should be considered individually, and the ACR clarifies several common misconceptions regarding the use of ICAs in the following patient groups:
- beta-blockers: patients on beta-blockers do not need to discontinue their medication(s) before contrast administration
- sickle-cell trait/disease: contrast should not be restricted in patients with sickle-cell trait/disease, as there is no evidence that iodinated contrast increases the risk for acute sickle crisis
- pheochromocytoma: there is no evidence that contrast use increases the risk of hypertensive crisis in patients with pheochromocytoma
- myasthenia gravis: there is a questionable relationship between the use of iodinated contrast and exacerbations of myasthenia gravis due to limited studies in a small number of patients; it remains controversial whether iodinated contrast should be considered a relative contraindication in these patients
- hyperthyroidism: patients with hyperthyroidism can develop thyrotoxicosis following exposure to iodinated contrast, although it is a rare complication. The ACR (2022) does not recommend the restriction of contrast medium solely based on a history of hyperthyroidism; however, it makes the following recommendations for two particular circumstances:
- "In patients with acute thyroid storm, iodinated contrast medium exposure can potentiate thyrotoxicosis; in such patients, iodinated contrast medium should be avoided. Corticosteroid premedication in this setting is unlikely to be helpful;
- In patients considering radioactive iodine therapy or patients undergoing radioactive iodine imaging of the thyroid gland, administration of an iodinated contrast medium can interfere with the treatment and diagnostic dose uptake. If iodinated contrast medium is administered, a washout period is suggested to minimize this interaction. The washout period is ideally 3-4 weeks for patients with hyperthyroidism, and 6 weeks for patients with hypothyroidism" (ACR, 2022, p.7).
Allergic Reactions
ICAs pose a risk for allergic reactions, which can have variable presentations ranging from acute to delayed, as outlined in Table 9. Acute reactions are further categorized by severity from mild to severe. The frequency of allergic reactions following the administration of iodinated contrast media has decreased with the change from utilizing high-osmolality contrast medical to nonionic low-osmolality contrast media. Patients can have an allergic reaction at initial exposure to the ICA, mediated by anaphylaxis mechanisms, in which symptoms develop within minutes of exposure. Anaphylactic reactions are usually unpredictable but are the most clinically significant reactions to ICAs, as they involve the release of histamine and other biologic mediators. Most reactions occur within the first 5 to 60 minutes following ICA administration. However, delayed reactions can occur up to one week following administration. Delayed reactions are more common in young adults, women, and patients with a history of a contrast allergy, and they tend to be cutaneous (ACR, 2022; Kocak, 2022d; Rogers & Tadi, 2023).
Table 9
Allergic Reactions to Iodinated Contrast Media
Severity | Description |
Mild
| Symptoms are self-limited without evidence of progression. |
| |
Moderate
| Symptoms are more pronounced and usually require medical management; they may have the potential to become severe if not treated. |
| |
Severe
| Symptoms are often life-threatening and can result in permanent morbidity or death if not managed appropriately. |
| |
Delayed | Rash that develops hours to 7 days following ICA administration Delayed reactions are usually limited to dermatologic manifestations. |
(ACR, 2022; Kocak, 2022d; Rogers & Tadi, 2023).
While ICA reactions are sporadic and often unpredictable, some factors are associated with an increased risk of reaction to ICA. The ACR (2022) advises that prior allergic-like or unknown-type reactions to the same class of contrast medium are considered the most significant risk factor for predicting future adverse events. Additional risk factors for ICA allergic-like reactions include the following:
- patients with a prior allergic reaction (or an unknown type of reaction) to an ICA are at an approximately 5-fold increased risk of developing a future allergic-like reaction with subsequent exposure to contrast
- patients with unrelated allergies are at a 2- to 3-fold increased risk for an allergic-like reaction
- patients with a history of asthma are at increased risk for allergy and may be more likely to develop bronchospasm, but those with well-controlled asthma may not be at increased risk
- patients with multiple allergies (such as dermatitis and urticaria) have at least a 3-fold increased risk for a severe reaction to contrast media
- patients with mild allergies (such as seasonal allergies) are at unclear risk (ACR, 2022; Beckett et al., 2015; Kocak, 2022d)
Despite popular misconception, it is now well-established and confirmed that there is no link between shellfish allergy and allergy to ICAs. Patients with shellfish or povidone-iodine (Betadine) allergies are at no higher risk from iodinated contrast than patients with other allergies. Furthermore, there is no cross-reactivity between the different classes of contrast; therefore, a patient with an ICA allergy is not at heightened risk for a gadolinium-based allergy (ACR, 2023; Beckett et al., 2015).
Treatment of Allergic Reactions
According to ACR (2022), all facilities in which ICAs are injected must be equipped with emergency supplies, and staff must be trained in basic life support in the event of a life-threatening allergic reaction. While most allergic reactions are mild, self-limiting, and do not progress in severity, they can sometimes become more severe. Therefore, it is advised that any patient who is displaying signs of reaction should be observed for a minimum of 20 to 30 minutes, or longer if needed, to ensure clinical stability and recovery. Mild reactions may require symptomatic treatment but often do not require any treatment at all. Moderate reactions are usually not life-threatening; severe reactions are rare but potentially fatal. Severe reactions require prompt recognition and action by qualified healthcare providers to prevent morbidity and death (ACR, 2022; Beckett et al., 2015).
Clinicians should maintain IV access, observe vital signs, and assess the patient's appearance for all reactions. The ACR (2022) lists the following five immediate assessments that should be performed upon recognition of a potential allergic reaction:
- What is the patient's general appearance?
- Can the patient speak? How does their voice sound?
- What is the quality of the patient's breathing?
- What is the patient's pulse?
- What is the patient's blood pressure?
Clinicians responding to these patients are advised to prioritize a focused assessment and evaluate the patient's level of consciousness, skin appearance, phonation quality, lung auscultation, blood pressure, and heart rate. These key assessment points help quickly determine the severity of the reaction so that effective treatment can be promptly and successfully administered. In free-standing outpatient facilities, staff should be trained to activate emergency response systems and understand when to elevate the care, such as by calling 911. These facilities should also be equipped with the necessary equipment, including but not limited to oxygen, an automated external defibrillator (AED), suction equipment, and medications. In addition to preserving IV access, monitoring vital signs and delivering supplemental oxygen by mask as needed (at a rate of 6 to 10 L/min) is essential. Table 10 concisely explains the guidelines for additional interventions for managing acute reactions to ICAs in adult patients (ACR, 2022).
Table 10
Management of Acute Reactions to ICAs in Adults
Clinical Manifestation | Treatment |
HIVES (URTICARIA) | |
Mild Scattered or transient | No treatment is needed, but if symptomatic, may use:
|
Moderate Numerous or symptomatic |
|
Severe Widespread or progressive |
|
* Note: Second-generation antihistamines may be beneficial for patients that need to drive themselves home | |
DIFFUSE ERYTHEMA | |
With hypotension | Administer 1,000 mL of IV fluids rapidly:
|
With profound hypotension (unresponsive to fluids) |
|
*Note: For the remainder of this treatment algorithm, follow the epinephrine administration dosing guidelines and instructions as outlined in this section, based on the administration route (IV, IM, or auto-injector); IV administration is preferred in hypotensive patients due to poor perfusion to the extremities | |
BRONCHOSPASM | |
Mild |
|
Moderate |
|
Severe |
|
LARYNGEAL EDEMA | |
|
|
HYPOTENSION Systolic blood pressure (SBP) < 90 mm Hg | |
|
|
Severe hypotension with bradycardia (pulse < 60 bpm) Vasovagal Reaction |
|
Severe hypotension with tachycardia (pulse > 100 bpm) Anaphylactoid Reaction |
|
HYPERTENSIVE CRISIS Diastolic blood pressure (DBP) > 120 mmHg; SBP > 200 mm Hg Symptoms of end-organ compromise | |
|
|
PULMONARY EDEMA | |
|
|
SEIZURES/CONVULSIONS | |
|
|
HYPOGLYCEMIA | |
If the patient can swallow safely: |
|
If the patient is unable to swallow safely and IV access IS available: |
|
If the patient is unable to swallow safely and IV access is NOT available: |
|
(ACR, 2022)
Premedication Regimen for Patients with an ICA Allergy
While well-controlled studies and definitive data are not available on the efficacy of premedication in preventing an allergic reaction in high-risk patients, the ACR (2022) believes that premedication does reduce the likelihood of a reaction in high-risk patients receiving low-osmolality iodinated contrast medium. While there has yet to be a formal consensus on the exact premedication regimen, oral premedication is preferable to IV premedication due to lower cost, increased convenience, and greater evidence support in the literature. The majority of data exists on the use of oral methylprednisolone (Medrol) or oral prednisone (Deltasone), with or without oral diphenhydramine (Benadryl). The ACR (2022) supports using the below premedication regimens in patients at high risk and at the discretion of the ordering clinicians.
Standard 12- or 13-hour premedication regimen:
- For use in outpatient, emergency department, and inpatient settings for patients at high risk in whom medication use is not anticipated to delay care decisions or treatment adversely.
- Regimen #1: prednisone (Deltasone) 50 mg PO at 13 hours, 7 hours, and 1 hour before contrast administration, plus diphenhydramine (Benadryl) 50 mg IV, IM, or PO 1 hour before contrast administration, OR
- Regimen #2: methylprednisolone (Medrol) 32 mg PO 12 hours and 2 hours before contrast administration, and diphenhydramine (Benadryl) 50 mg may be added as in regimen #1, OR
- Regimen #3: (for patients who are unable to take oral medication) is the same as regimen #1, but hydrocortisone (Cortef) 200 mg is given as a replacement for each dose of prednisone (Deltasone), plus diphenhydramine (Benadryl) 50 mg IV 1 hour before contrast administration (ACR, 2022)
Accelerated IV premedication regimen:
- For use in outpatient, emergency department, and inpatient settings for patients at high risk in whom the use of 12- or 13-hour premedication is anticipated to delay care or treatment adversely.
- Regimen #1: (usually 4-5 hours in duration) methylprednisolone sodium succinate (Solu-Medrol) 40 mg IV or hydrocortisone sodium succinate (Solu-Cortef) 200 mg IV immediately, and then every 4 hours until contrast administration, plus diphenhydramine (Benadryl) 50 mg IV 1 hour before contrast administration, OR
- Regimen #2: (usually 4-5 hours in duration) dexamethasone sodium sulfate (Decadron) 7.5 mg IV immediately, and then every 4 hours until contrast administration, plus diphenhydramine (Benadryl) 50 mg IV 1 hour before contrast administration, OR
- this is the preferred regimen for patients with an allergy to methylprednisolone (Solu-Medrol)
- Regimen #3: methylprednisolone sodium succinate (Solu-Medrol) 40 mg IV or hydrocortisone sodium succinate (Solu-Cortef) 200 mg IV, plus diphenhydramine (Benadryl) 50 mg IV, each 1 hour before contrast administration
- Any regimen less than 4-5 hours in duration (including this regimen) has no evidence of efficacy but may be considered in emergent situations where there are no alternatives (ACR, 2022)
Special Considerations for Patients who are Pregnant or Breastfeeding
The effects of ICAs on the human embryo and fetus remain incompletely understood. Studies have demonstrated that ICAs can cross the placenta and enter the fetus in measurable amounts, but the effects are unknown, and long-term data still need to be gathered. The ACR (2022) does not recommend routine screening for pregnancy before ICA administration. The FDA supports this recommendation, and most ICAs are category B medications except for diatrizoate meglumine and diatrizoate meglumine sodium, classified as category C. The literature base regarding the safe use of ICA in breastfeeding patients is also limited; however, many studies have demonstrated that the routine dose of ICA absorbed by the infant's gut from ingested breast milk is extremely low. While the ACR states that available data suggest it is safe for the lactating individual and infant to continue breastfeeding after receiving ICA, the individual should be allowed to make an informed decision after fully disclosing the available information. For individuals who abstain from breastfeeding temporarily, the recommendation is to express and discard breastmilk for 12 to 24 hours following ICA administration. There is no benefit to refraining from breastfeeding beyond 24 hours (ACR, 2022; Perelli et al., 2022). The ACOG Committee on Obstetric Practice recommends that iodinated contrast should only be used if required to obtain essential additional diagnostic information that will affect the care of the fetus or pregnant individual during the pregnancy. The ACOG also supports the continuation of breastfeeding without interruption following the use of iodinated contrast materials (ACOG, 2017).
Gadolinium
GBCAs are the primary form of contrast used to enhance the quality of MRI scans. Administered IV, GBCAs are paramagnetic and work by altering the magnetic properties of neighboring water molecules, thereby enhancing the quality of the MRI images. The first GBCA was approved for use by the FDA in 1988. Currently, there are nine types of MRI contrast agents available for use. Similar to ICAs reviewed in the prior section, the osmolarity and viscosity of the GBCAs can also vary widely and are generally higher for the ionic than the nonionic agents. Gadolinium is one of the metals included in the Lanthanide series, and if used alone, it is toxic to humans. Gadolinium in GBCAs has undergone chelation, in which other chemical ions are mixed with gadolinium to mitigate its harmful effects on the body. The majority of GBCAs used in the US are chelates of gadolinium, have a positive magnetic susceptibility, and function to produce an increased signal on the resulting images. The GBCA is injected into the patient before the MRI and is primarily eliminated from the body via the kidneys, with a small amount of liver excretion (Ibrahim et al., 2022; Rogers & Tadi, 2023).
GBCAs are generally well-tolerated and are much less likely to produce an allergic reaction than ICAs. The incidence of adverse events when administered at clinical doses of 0.1-0.2 mmol/kg is low, ranging from 0.07% to 2.4%. Most reactions are mild and physiologic, including coldness, warmth, pain at the injection site, nausea, vomiting, headache, paresthesia, and dizziness. Allergic-like reactions are uncommon, ranging from 0.004% to 0.7%, and symptoms are generally very mild and transient, usually limited to hives, other skin reactions, or itchy eyes. While manifestations of allergic-like reactions to GBCA can be similar to those of ICAs, they are rare, with life-threatening anaphylactic reactions occurring at a low rate of 0.001% or 0.01%. For example, in a survey of 20 million administered doses of GBCA, there were only 55 severe reactions. While fatal reactions to GBCAs are possible, they are rare and are often due to cardiovascular or respiratory effects. When managing GBCA-related allergic reactions, clinicians should follow the same ICA guidelines outlined in Table 6 (ACR, 2022).
In 2015, the FDA published a safety alert as residual gadolinium had been found within the brain of patients who received multiple doses of GBCAs throughout their lifetime. Research has demonstrated that gadolinium deposits appear to occur preferentially within specific areas of the brain, even in the absence of clinically evident disease, and in the setting of intact blood-brain barriers. The etiology of this remains unclear and is a relatively undefined clinical phenomenon. Legal suits have been brought due to this phenomenon in recent years despite a lack of clinical correlation (ACR, 2022). Clinicians must balance the potential risks of GBCAs with the diagnostic benefits, which are also premised on a multitude of patient-specific factors such as:
- the probability and necessity of an accurate diagnosis
- alternative methods of diagnosis
- risk of misdiagnosis
- the clinical benefit of the diagnostic information against the unknown, yet the potential risk of gadolinium deposition in the brain
- expectations regarding the patient's risk for the development of nephrogenic systemic fibrosis (NSF; Davenport et al., 2020)
NSF is a rare systemic but potentially life-threatening complication with an unknown etiology unique to GBCAs. It is characterized by thickening skin, organs, and other tissues in patients with severe preexisting renal disease. The initial symptoms typically include skin thickening and pruritus, which can then rapidly progress to contractures and joint immobility. In extreme cases, the condition can be fatal. The time between GBCA administration and the onset of NSF is usually 2 to 10 weeks following GBCA administration. However, rare case reports of symptoms have appeared years after the last GBCA exposure. NSF seems to have first appeared around 2000, but radiology guideline changes did not take place until 2006, at which time clinicians began to withhold GBCAs in patients with AKI or severe CKD when the GFR is <30mL/min/1.73m2. While this led to a decline in the incidence of NSF cases, further investigation demonstrated that the risk for NSF was higher in specific GBCAs. As a result, modern-day radiology algorithms group GBCAs based on their reported associations with NSF in vulnerable patient populations, as outlined in Table 11 (ACR, 2022; Ibrahim et al., 2022).
Table 11
Classification of GBCAs in Relation to Reported Cases of NSF with ACR Recommendations for Use
Group | Risk Interpretation | Agents |
Group I
Associated with the highest number of NSF cases | The ACR considers patients exposed to Group 1 GBCAs to be at risk of developing NSF if any of the following conditions apply: patients on dialysis (any form), those with CKD grade 4 or 5 without dialysis, or those with AKI. |
|
Group II
Associated with few (if any) unconfounded cases of NSF | The ACR considers the risk of NSF among patients exposed to standard or lower than-standard doses of Group II GBCAs to be sufficiently low or possibly nonexistent. |
|
Group III Data are insufficient regarding the risk for NSF, but few (if any) unconfounded cases of NSF have been reported | There is insufficient real-life data to determine the likelihood of NSF from administering group III GBCAs. Therefore, the ACR views Group III agents as carrying the same risk level as Group I GBCAs. |
|
Summary ACR Recommendations for GBCA Use in Patients at Risk for NSF | ||
| ||
(ACR, 2022)
The ACR (2020) advises clinicians to follow the algorithm demonstrated in Figure 9 regarding the evaluation of renal function before GBCA administration. Regarding this clinical decision-making algorithm, a history of renal disease, including AKI, is considered a risk factor, as well as any of the following:
- prior or current dialysis
- renal transplantation
- single kidney
- kidney surgery
- renal cancer
- hypertension requiring medical therapy
- diabetes mellitus (ACR, 2022)
Figure 9
Clinical Decision Algorithm for Evaluating Renal Function Before Group I or Group III GBCA Administration
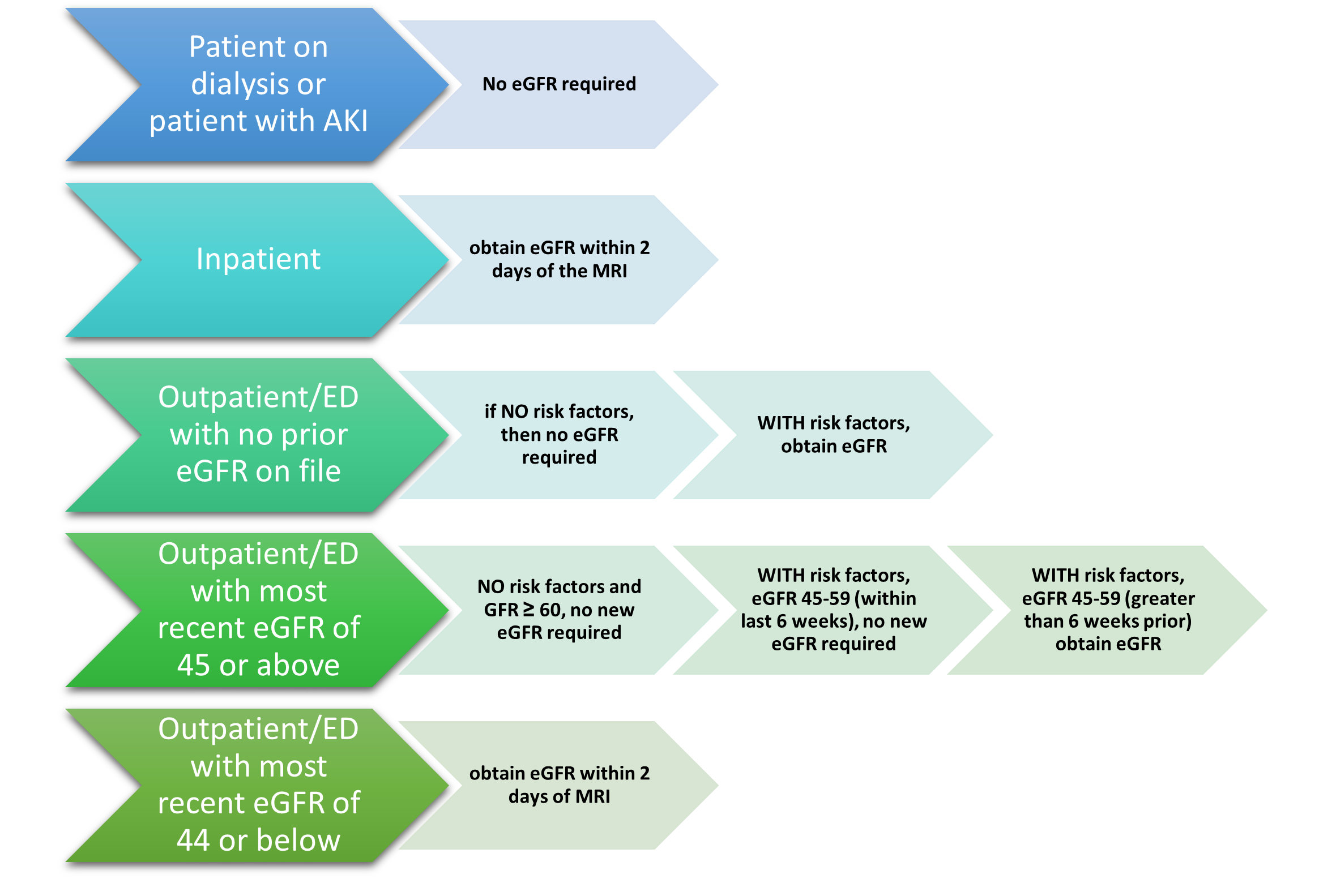
(ACR, 2022)
The ACR (2022) makes several recommendations regarding the use of GBCAs in the following patient groups:
- patients with CKD 4 of 5 not on dialysis: group I GBCAs are contraindicated, and group II GBCAs should be used instead
- patients with AKI: group I GBCAs should be avoided if possible, and group II agents are preferred
- patients on metformin (Glucophage): it is not necessary to discontinue metformin (Glucophage) before GBCA administration when the standard dose of gadolinium (0.1 to 0.3 mmol/kg) is being used
- sickle cell disease/trait: the risk to patients with sickle cell disease at approved dosages is very low or nonexistent, and withholding contrast or premedicating patients with no other risk factors is not recommended (ACR, 2022)
Special Considerations for Patients who are Pregnant or Breastfeeding
No known adverse effects have been reported in fetuses when standard doses of GBCAs were administered to pregnant women; however, similar to ICAs, the data is limited, and long-term studies are lacking. There are no reported cases of NSF related to GBCAs in pregnant women. However, gadolinium chelates can accumulate in the amniotic fluid, which suggests a potential risk for NSF in the pregnant individual or fetus. Since it remains unclear how GBCAs can affect the fetus, it is recommended that these agents be administered with caution in pregnant patients. GBCAs should only be used if considered critical, and the potential benefits justify the potentially unknown risks to the fetus. The lowest possible dosage for diagnostic results should be used in these cases. The ACOG Committee on Obstetric Practice acknowledges that the use of gadolinium contrast is limited and should not be withheld if it significantly improves the predicted diagnostic performance and outcomes for the fetus or pregnant individual. The ACOG also recommends that breastfeeding not be interrupted following gadolinium administration, as less than 0.04% of the administered dose is excreted into breast milk. However, the individual should be allowed to make an informed decision after fully disclosing the available information. For individuals who abstain from breastfeeding temporarily, the recommendation is to express and discard breast milk for 12 to 24 hours following GBCA administration. There is no benefit to refraining from breastfeeding beyond 24 hours (ACOG, 2017; ACR, 2022).
A study conducted in 2019 showed an increased prevalence of GBCA administration during the first trimester of pregnancy. This was attributed to a lack of pregnancy screening before diagnostic testing. To avoid GBCA administration during pregnancy, the ACR recommends that facilities that conduct imaging with GBCA administration have a pregnancy screening protocol. Unfortunately, no pregnancy screening technique is 100% effective, and false negatives may result if testing occurs early in the pregnancy; therefore, all individuals able to become pregnant should be counseled on the potential risks and lack of complete understanding of the ramifications of fetal exposure to GBCA (ACR, 2022).
Barium Sulfate
Barium sulfate contrast agents (BSCAs) are preferred for evaluating and visualizing the GI tract. They can only be administered orally or rectally. They are composed of a suspension of insoluble barium sulfate particles not absorbed by the gut. They are ideal for illuminating the GI tract since they have excellent coating properties of the GI mucosa, as demonstrated in Figure 10. They are the most common type of orally administered contrast material and enhance the visualization of the GI tract in CT scans, x-rays of the abdomen and pelvis, and specific MRI scans. They are also commonly used for fluoroscopic studies, such as an upper gastrointestinal (UGI) examination, small bowel meal, or enteroclysis (small bowel enema; ACR, 2022; Gotfried, 2023).
Figure 10
Visualization of the Intestinal Tract Following Administration of Barium Contrast
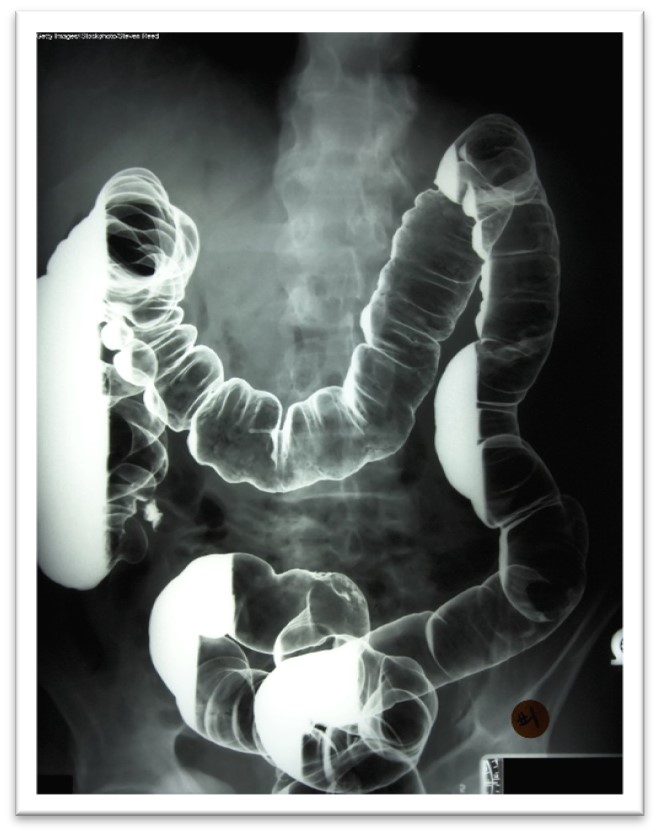
Patients scheduled to undergo a scan using an oral BSCA should be advised to remain NPO for four to eight hours before the exam, depending on the type of scan and the institution's policy. If the contrast material is to be administered rectally, the patient will usually be instructed to follow a clear liquid diet the day before and require a colon preparation with bisacodyl (Dulcolax), polyethylene glycol (Miralax), or magnesium citrate to clean the colon before the exam (ACR, 2022; Zaccarini et al., 2022).
While the ACR (2022) states explicitly that there are no absolute contraindications for the use of BSCAs, they recommend avoiding their use in patients who are suspected or known to have bowel perforations or prior allergies to barium. Older adults or those unable to change positions may have difficulty turning themselves to distribute the barium properly. Patients who have a suspected fistula, paralytic ileus, severe dehydration, or a history of cystic fibrosis (which is associated with an increased risk for blockage in the small bowel) are considered to be at greater risk for having an adverse reaction to or complication from BSCA administration. Barium sulfate is generally well tolerated orally, although some patients describe a mildly unpleasant taste. When barium sulfate is administered through the rectum using an enema, patients may experience temporary abdominal fullness and mild discomfort. It is generally recommended to ensure the BSCA is warmed to room temperature, which helps enhance patient tolerability and reduce colon spasms (ACR, 2022; Cutler, 2016; Gotfried, 2023; RadiologyInfo.org, 2022b).
Adverse reactions to BSCAs are rare and are almost always mild in presentation. The most common symptoms include nausea, vomiting, abdominal cramping, or discomfort, which can occur during or after the examination. Patients can have vasovagal reactions, more common with rectal contrast administration after the colon is distended. Following the completion of a BSCA procedure, patients should be instructed to increase oral fluid intake to help facilitate and expedite the removal of the contrast material from the body, which is expelled through the feces. Patients should be educated that their stool may be white for a few days until all the contrast material is eliminated. It is common for some patients to experience changes in their routine bowel habits for the first 12 to 48 hours, such as diarrhea or constipation. Barium enemas are also used therapeutically in some instances to remove feces from the bowel. This procedure may be recommended for patients with severe constipation or blockages to prevent surgical intervention and reduce the risk of bowel perforation. There have been rare reports of appendicitis following the use of a barium enema. This phenomenon is known as barium-induced appendicitis and is thought to result from barium retention and subsequent obstruction of the appendiceal lumen. Perforation may also occur due to barium administration; this adverse effect is most commonly seen in patients with toxic megacolon after administering a barium enema (Gotfried, 2023; RadiologyInfo.org, 2022b; Zaccarini et al., 2022).
Allergic-like reactions to BSCA are uncommon, with an incidence rate of 1 in 750,000 examinations. If an allergic-like reaction does occur, the manifestations are usually very mild and limited to transient rashes, urticaria, pruritus, or mild bronchospasm. Moderate to severe allergic reactions are rare, occurring in about 1 out of 2.5 million patient exposures. Clinical manifestations of moderate to severe allergic reactions may include more extensive dermatologic changes, respiratory symptoms, and vascular events such as hypotension. Rarely, angioedema of the stomach and small bowel and toxic epidermal necrolysis (TEN) have been described. Although some believe there is a correlation between asthma and allergic-type reactions to barium, no conclusive evidence supports this claim (ACR, 2022).
Though this is infrequent, direct barium toxicity has been reported in patients receiving oral and rectal BSCAs. The etiology is based on the concept that any barium dissociating from the stable barium compound may be absorbed through the bloodstream, leading to toxicity. Symptoms of barium toxicity usually present rapidly and include watery diarrhea, nausea, and vomiting. Absorption of barium can cause electrolyte imbalances, including rapid hypokalemia, which, if left untreated, can cause muscle weakness, respiratory or cardiac arrest, and death. The treatment is limited to supportive therapy, rehydration, and managing electrolyte disturbances (ACR, 2022).
Microbubble Contrast Agents
Microbubble contrast agents (MCAs) are most commonly used to enhance ultrasonography images, particularly heart exams. Currently, microbubbles are the only approved contrast agent for use with ultrasounds. MCAs are microscopic, typically 1 to 8 µm, highly compressible gas-filled, and coated with a phospholipid or protein outer layer. MCAs have a high degree of "echogenicity," or the ability to reflect ultrasound waves; structures with higher echogenicity will appear brighter than surrounding areas on the resulting ultrasound images. These tiny gas bubbles can be safely injected IV through a peripheral or central access line or instilled into hollow structures, such as the urinary bladder. Once injected, they allow for transient improvement in ultrasound visualization, capturing differences between the gas in the microbubbles and the surrounding tissues. They enhance the ultrasound signal from blood and improve the visualization of blood flow without subjecting the patient to several risks associated with other forms of contrast media (ACR, 2022; RadiologyInfo.org, 2022b; Yusefi & Helfield, 2022).
These agents are approved for administration via slow IV infusion or bolus injection. Since the microbubbles dissolve rapidly, usually within 15 minutes, image acquisition should be completed within 10 minutes post-injection before the microbubbles spontaneously rupture and dissolve. As the microbubbles dissolve, they release a gas eliminated mainly by the lungs through exhalation. This is a valuable and practical alternative to ICAs or GBCAs for patients with AKI, CKD, or allergies (RadiologyInfo.org, 2022b; Yusefi & Helfield, 2022).
According to the ACR (2020), there are currently three MCAs that are FDA-approved for IV administration in adults undergoing echocardiography to improve visualization of the left ventricular cavity and the endocardial borders. These three agents include perflutren lipid microspheres (Definity), sulfur hexafluoride lipid-type A microspheres (Lumason; SonoVue), and perflutren protein-type A (Optison). Sulfur hexafluoride lipid-type A microspheres (Lumason) is additionally approved for liver imaging and imaging of the pediatric urinary tract during voiding ultrasonography (ACR, 2022; Yusefi & Helfield, 2022). While their current FDA-approved indications are limited to the above, these agents have also been successfully used off-label for the following assessments and procedures:
- blood flow in tumors
- benign cysts and solid tumors in the kidney
- blood perfusion in organs
- abnormalities in the heart
- thrombosis, such as in myocardial infarction
- bowel wall inflammation or inflammatory activity in inflammatory bowel disease as well as liver and kidney masses
- endoleaks following AAA repair
- abscesses and phlegmons
- ultrasound-guided interventions and ablative therapies (ACR, 2022; RadiologyInfo.org, 2022b; Yusefi & Helfield, 2022)
MCAs have a reasonably favorable safety profile, as the rate of adverse events is less than that of ICAs or GBCAs. MCAs have been administered millions of times worldwide and have been found to have an excellent safety profile. A meta-analysis of microbubble tolerance indicated that most severe adverse effects are caused by pseudoanaphylaxis with a rate of 0.004-0.009%. Adverse events are usually mild and physiologic. The most common symptoms include headaches, flushing, warmth, nausea, and altered taste, with most severe reactions occurring within 30 minutes of MCA administration. The risk for severe cardiopulmonary reactions increases in patients with a preexisting and unstable cardiac condition, such as an acute myocardial infarction or congestive heart failure. There is no reported renal toxicity with MCAs. MCAs should not be used intra-arterially and are contraindicated in patients with prior allergic reactions to these agents (ACR, 2022; Yusefi & Helfield, 2022).
Special Considerations for Patients who are Pregnant or Breastfeeding
No well-controlled studies evaluating the risks of MCAs when administered to pregnant women are available. Therefore, the ACR (2020) recommends only using these agents in pregnant women when necessary and when the prospective benefits outweigh the unknown but potential risks for the fetus. Breastfeeding individuals should be counseled on temporarily pumping and discarding breastmilk for up to 24 hours following administration (ACR, 2022).
Saline Contrast Media
Agitated saline contrast (ASC) is considered a distinct type of contrast within the MCA family. It is administered intravenously and provides air microbubble contrast to the right side of the heart. Like the above MCAs, ASC is transient and diffuses into the lungs. This technique helps examine the right side of the heart and identify intracardiac or extracardiac (pulmonary arteriovenous) shunts.; particularly right to left shunts. The primary safety concern following the administration of ASC involves cerebral air embolism. There have been reports of ischemic complications, including transient ischemic attack and stroke. This extremely low risk has been attributed to a technical error where the ASC was not mixed or injected correctly. When ischemic events occur, the effects are often mild and short-lived (Ahmed et al., 2021; Millington et al., 2023).
Gaseous Contrast Agents
Gaseous agents (such as oxygen [O2] and carbon dioxide [CO2]) can also be used as contrast agents in specific types of imaging tests. Similar to MCAs used in ultrasonography, these agents are absorbed rapidly by the body and eliminated through exhalation. Research is limited on these contrast agents, and the ACR manual of contrast media omits any reference to or discussion of these agents entirely. While air and oxygen can be dangerous due to the risk of gas emboli during specific procedures, CO2 does not pose a risk of gas emboli and can be used with relative safety. CO2 has the most research data supporting its clinical benefits and use as an intravascular contrast agent. It is used as an alternative to ICA or GBCAs in patients with renal dysfunction since it has no adverse effects ok the kidneys or allergies to ICA. It is the least expensive contrast medium, is readily available, has low molecular weight compared to ICAs, and is less viscous than blood. These characteristics make it an ideal agent for imaging small collateral vessels and various vascular beds and chambers. It displaces the blood in the vessels and acts as a negative contrast agent. However, administering CO2 as a contrast agent requires extreme care as it is a colorless, odorless, and significantly compressible gas. It is also less sensitive and specific than ICAs or GBCAs (Gupta et al., 2020; Young & Mohan, 2022). CO2 has been successfully used as a contrast agent in the following clinical situations:
- angiography in patients with peripheral arterial disease who are at risk for contrast-induced nephropathy
- aortic aneurysm repairs
- aortography
- renal artery angiography
- inferior vena cava imaging and filter placement
- portal vein imaging
- splenoportography
- oncologic tumor embolization procedures
- upper extremity venography
- dialysis fistula or graft repair
- assessment of gastrointestinal bleeding (Gupta et al., 2020; Young & Mohan, 2022)
Contraindications to the use of CO2 include the concurrent use of nitrous oxide as a sedating agent. This is due to the decreased solubility of CO2 when mixed with nitrous oxide. CO2 arteriography should not be performed above the diaphragm due to an increased risk of cerebral air embolism leading to stroke or death. When possible, the patient should be placed in the Trendelenburg position to decrease the risk of embolism. CO2 can also become trapped in the pulmonary artery or right side of the heart leading to obstruction of venous return and subsequent hypotension and bradycardia. This phenomenon can be alleviated by placing the patient in a left lateral decubitus position to float the gas in a layer above the blood until the gas molecules dissolve in the bloodstream entirely (Young & Mohan, 2022).
Diagnostic Imaging During Pregnancy and Lactation Overview
According to the ACOG Committee on Obstetric Practice, diagnostic imaging during pregnancy and lactation is controversial with various opinions. The confusion regarding the safety of diagnostic imaging often results in unnecessary avoidance of needed testing and interruption of breastfeeding. The most common documented adverse effects from high-dose radiation exposure on the developing fetus include growth restriction, microcephaly, and intellectual disability. The ACOG Committee on Obstetric Practice (2017) makes the following recommendations regarding diagnostic imaging procedures during pregnancy and lactation:
- Ultrasound and MRI are the preferred imaging techniques for patients. Still, they should be used only when use is strongly predicted to provide medical benefit to the mother or fetus.
- Most radiation exposure through CT scans or nuclear medicine imaging is provided at a dose much lower than the exposure associated with fetal harm. If these techniques are deemed medically necessary, they should not be withheld solely based on pregnancy.
- It generally is recommended that iodinated contrast only be used if required to obtain essential additional diagnostic information that will affect the care of the fetus or mother during the pregnancy. Breastfeeding can be continued without interruption following the use of iodinated contrast materials. For individuals who abstain from breastfeeding temporarily, the recommendation is to express and discard breastmilk for 12 to 24 hours following administration.
- The use of gadolinium contrast with MRI should be limited and used only if it significantly improves the predicted diagnostic outcomes for the fetus or mother. Breastfeeding should not be interrupted following gadolinium administration.
- Radioactive iodine-123 (I-123) and iodine-131 (I-131) readily cross the placenta, and use should be avoided in pregnancy. If a thyroid scan is essential, it is recommended that the radioactive isotope technetium 99m is used.
- Since specific nuclear materials excreted into breast milk can have deleterious effects on the infant, consultation with experts in breastfeeding and nuclear medicine is recommended when these compounds are used in lactating women.
The Burden of Overuse and Misuse of Medical Imaging
The rate of diagnostic imaging and associated radiation exposure has expanded tremendously since the 1990s. Medicare enrollees were the highest utilizers of diagnostic imaging, followed by individuals between the ages of 45 and 64 that were commercially insured. Between 2000 and 2006, the use of CT, MRI, and ultrasound for diagnostic testing increased rapidly in the US. Compared to other developed countries, the US is ranked as the highest utilizer of CT and MRI scans per 1,000 individuals. Between 2000 and 2016, the rate of CT scans per 1,000 individuals increased from 204 to 428 for those 65 or older and from 63 to 134 for those aged 18-64. The rate of MRI imagining between 2000 and 2016 increased from 62 per 1,000 individuals to 139 in those aged 65 or older and 42 to 85 in those aged 18-64. During the same time frame, the rate of ultrasound testing increased from 324 to 495 per 1,000 individuals among individuals 65 or older and from 174 to 278 for those aged 18-64 (Smith-Bindman et al., 2019)
There has been a rise in radiation exposure secondary to the increased amount of CT scans performed and the increase in the radiation dose in each scan. Due to advancements in technology, modern CT scans are multiphasic, which means they are performed using repeated scanning before and after injecting a contrast dye agent. The multiphasic scanning process generates more precise and significantly higher-resolution images, allowing for increased diagnostic accuracy. However, they expose patients to 30-50% more radiation than older CT scanners (Agency for Healthcare Research and Quality [AHRQ], 2019).
The increased diagnostic imaging has led to increased adverse effects, including incidental findings, overdiagnosis, overtreatment, and healthcare costs. It is estimated that unnecessary diagnostic imagining accounts for between $75.7 and $101.2 billion annually in wasted healthcare spending in the US. While radiation exposure is a widely recognized and well-described health risk of medical imaging, NCI also describes the risk of false-positive and false-negative test results. False-positive results occur when an abnormality is identified on the scan, but no pathology or cancer is present within the patient. False positives often lead to additional diagnostic work-up, including further radiology imaging tests carrying additional radiation exposure, or invasive tests, such as a biopsy. False-negative results mean no abnormality was identified on the imaging test, despite some underlying pathologic process within the patient. False-negative results lead to delays in treatment and a false sense of security for the affected patient. Incidental findings refer to identifying a previously undiagnosed medical condition discovered unintentionally during evaluation for another disease or ailment. Incidental findings pose several medical and ethical dilemmas for clinicians and patients, often leading to additional diagnostic work-up and monitoring, contributing to further unnecessary radiation exposure, depending upon the identified condition. There are times when incidental findings can be life-saving, such as when a pancreatic tumor is identified during imaging for another reason. However, most incidental findings contribute to the overuse of medical imaging services and inflate the cost burden on the patient, the healthcare system, and the economy. Similarly, overdiagnosis and overtreatment are well-described in medical imaging literature. One of the most prominent and controversial examples of overdiagnosis and overtreatment is ductal carcinoma in situ (DCIS) of the breast, which is commonly identified on screening mammography. DCIS is a noninvasive tumor of atypical cells that have not spread outside the ducts or lobules into the surrounding breast tissue. DCIS has been the topic of tremendous debate over the last few decades regarding how to classify the condition and its definitive treatment pathway (Chalmers et al., 2021; NCI, 2023).
To reduce the burden of unnecessary medical imaging, the American Board of Internal Medicine (ABIM) Foundation launched the Choosing Wisely campaign in 2012. The choosing wisely campaign has compiled evidence-based recommendations from various medical specialty societies about potentially unnecessary medical testing and procedures. It is intended to guide clinicians on the proper ordering of diagnostic imaging and promote conversations between clinicians and patients; it helps patients to choose care that is "supported by evidence, not duplicative of other tests or procedures already received, free from harm, and truly necessary" (ABIM Foundation, n.d., para. 2).
Expanding on the Choosing Wisely campaign, CMS established the Protecting Access to Medicare Act (PAMA) of 2014, Section 218(b), to increase the rate of appropriate advanced diagnostic imaging services provided to Medicare beneficiaries. This legislation led to the development of the Appropriate Use Criteria (AUC) for Advanced Diagnostic Imaging, which serves as a resource modality to increase the rate of appropriate advanced diagnostic imaging services while decreasing the risk of medically unnecessary imaging. The AUC information is available to clinicians through an electronic, interactive tool called a Clinical Decision Support Mechanism (CDSM). The CDSM tool helps with clinical decision-making and can be accessed through electronic health record (EHR) technology, private-sector mechanisms, or CMS-established ones. The objective is to ensure clinicians order the most patient-appropriate test based on the specific clinical condition. Initially, participation was voluntary, but as of January 1, 2023, the payment penalty phase was implemented. CMS released a statement removing the voluntary participation clause, informing providers ordering Medicare Part B advanced diagnostic imaging services to access CDSM and review the AUC before ordering the test. This requirement applies to CT, MRI, nuclear medicine, and PET scans, for all Medicare patients to be rendered in outpatient settings and emergency departments. Decisions about the appropriateness of the test are based on a 1-9 scale, with ratings of 4-9 considered appropriate use of the potential diagnostic test. CMS uses the data collected from the AUC program to identify any outlier ordering clinicians, who then become subject to a prior authorization requirement (Advanced Diagnostic Radiology [ADR], n.d.; CMS, 2022). The recommendations when ordering medical imaging tests are summarized as follows (IAEA, n.d.):
- Justification: Each diagnostic radiology imaging test and procedure must be justified and only performed when necessary. The benefits and risks of the intended test must be considered, and alternatives that pose less risk should be contemplated.
- Optimization and ALARA: Tests should be performed using the minimal effective radiation dose required to achieve quality images, thereby reducing the radiation exposure as much as possible.
- Avoid unnecessary repeat testing: Repeat testing is often necessary to monitor disease status and evaluate for treatment response, such as with cancer; however, all unnecessary repeat testing should be avoided.
References
117th Congress. (2022). H.R.2617 - Consolidated Appropriations Act, 2023. https://www.congress.gov/bill/117th-congress/house-bill/2617
Adams, C., & Banks, K. P. (2022). Bone scan. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK531486
Advanced Diagnostic Radiology. (n.d.). Appropriate use criteria (AUC). Retrieved April 24, 2023, from https://adronline.org/auc
Agency for Healthcare Research and Quality. (2019). Radiation safety. https://psnet.ahrq.gov/primer/radiation-safety#Radiation-Risks-Associated-With-Diagnostic-Imaging
Ahmed, H., Manning, W. J., & Downey, B. C. (2021). Contrast echocardiography: Contrast agents, safety, and imaging techniques. UpToDate. Retrieved April 12, 2023, from https://www.uptodate.com/contents/contrast-echocardiography-contrast-agents-safety-and-imaging-technique
American Association of Physicists in Medicine. (n.d.). Patient gonadal and fetal shielding in diagnostic imaging: Frequently asked questions. Retrieved April 20, 2023, from https://www.aapm.org/org/policies/documents/CARES_FAQs_Patient_Shielding.pdf
American Association of Physicists in Medicine. (2019). AAPM position statement on the use of patient gonadal and fetal shielding (PS 8-A). https://www.aapm.org/org/policies/details.asp?id=2552
American Board of Internal Medicine Foundation. (n.d.). Choosing wisely: Our mission. Retrieved April 12, 2023, from https://www.choosingwisely.org/our-mission
American Cancer Society. (2018). Understanding radiation risk from imaging tests. https://www.cancer.org/treatment/understanding-your-diagnosis/tests/understanding-radiation-risk-from-imaging-tests.html
American Cancer Society. (2023). Key statistics for breast cancer. https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html
American College of Obstetricians and Gynecologists. (2017). ACOG committee
opinion: Guidelines for diagnostic imaging during pregnancy. Obstetrics & Gynecology, 130(4), e210-e216. https://www.acog.org/-/media/project/acog/acogorg/clinical/files/committee-opinion/articles/2017/10/guidelines-for-diagnostic-imaging-during-pregnancy-and-lactation.pdf
American College of Obstetricians and Gynecologists. (2022). Osteoporosis: Frequently asked questions. https://www.acog.org/womens-health/faqs/osteoporosis
American College of Radiology. (2018). ACR-SPR practice parameters for imaging pregnant or potentially pregnant adolescents and women with ionizing radiation. https://www.acr.org/-/media/ACR/Files/Practice-Parameters/Pregnant-Pts.pdf
American College of Radiology. (2019). ACR-SNMMI-SPR practice parameter for the performance of scintigraphy and uptake measurements for benign and malignant thyroid disease. https://www.acr.org/-/media/ACR/Files/Practice-Parameters/Thy-Scint.pdf
American College of Radiology. (2020). ACR manual on MR safety. https://www.acr.org/-/media/ACR/Files/Radiology-Safety/MR-Safety/Manual-on-MR-Safety.pdf
American College of Radiology. (2021). ACR–CANM-SNMMI-SPR practice parameters for the performance of skeletal scintigraphy (bone scan). https://www.acr.org/-/media/ACR/Files/Practice-Parameters/Skeletal-Scint.pdf
American College of Radiology. (2022). ACR manual on contrast media. https://www.acr.org/-/media/ACR/Files/Clinical-Resources/Contrast_Media.pdf
American Heart Association. (2022). Echocardiogram (echo). https://www.heart.org/en/health-topics/heart-attack/diagnosing-a-heart-attack/echocardiogram-echo
American Thyroid Association. (n.d.). Low iodine diet. Retrieved May 9, 2023, from https://www.thyroid.org/low-iodine-diet
Beckett, K. R., Moriarity, A. K., & Langer, J. M. (2015). Safe use of contrast media: What the radiologist needs to know. Radiographics, 35(6), 1738-1750. https://doi.org/10.1148/rg.2015150033
Camacho, P. M., Petak, S. M., Binkley, N., Diab, D. L., Eldeiry, L. S., Farooki, A., Harris, S. T., Hurley, D. L., Kelly, J., Lewiecki, M., Pessah-Pollack, R., McClung, M., Wimalawansa, S. J., & Watts, N. B. (2020). American Association of Clinical Endocrinologists/American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis - 2020 update. Endocrine Practice, 26(Suppl 1), 1-46. https://doi.org/10.4158/GL-2020-0524
Cascino, T., & Shea, M. J. (2022). Echocardiography. Merck Manual Professional Version. https://www.merckmanuals.com/professional/cardiovascular-disorders/cardiovascular-tests-and-procedures/echocardiography
Centers for Disease Control and Prevention. (2015a). Health effects of radiation: Health effects depend on the dose. https://www.cdc.gov/nceh/radiation/dose.html
Centers for Disease Control and Prevention. (2015b). The electromagnetic spectrum: Non-ionizing radiation. https://www.cdc.gov/nceh/radiation/nonionizing_radiation.html
Centers for Disease Control and Prevention. (2015c). What is radiation? The electromagnetic spectrum [image]. https://www.cdc.gov/nceh/radiation/spectrum.html
Centers for Disease Control and Prevention. (2021a). Computed tomography (CT) scans [image]. https://www.cdc.gov/nceh/radiation/ct_scans.html
Centers for Disease Control and Prevention. (2021b). The electromagnetic spectrum: Ionizing radiation. https://www.cdc.gov/nceh/radiation/ionizing_radiation.html
Centers for Disease Control and Prevention. (2021c). Radiation in healthcare: Fluoroscopy. https://www.cdc.gov/nceh/radiation/fluoroscopy.html
Centers for Disease Control and Prevention. (2022a). Does osteoporosis run in your family? https://www.cdc.gov/genomics/disease/osteoporosis.htm
Centers for Disease Control and Prevention. (2022b). Radiation in healthcare: Imaging procedures. https://www.cdc.gov/nceh/features/medical-imaging-procedures/index.html
Centers for Medicare & Medicaid Services. (2018). Magnetic resonance imaging. https://www.cms.gov/medicare-coverage-database/view/ncd.aspx?ncdid=177
Centers for Medicare & Medicaid Services. (2022). Appropriate use criteria program. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Appropriate-Use-Criteria-Program
Chalmers, K., Smith, P., Garber, J., Gopinath, V., Brownlee, S., Schwartz, A. L., Elshaug, A. G., & Saini, V. (2021). Assessment of overuse of medical tests and treatments at US hospitals using Medicare claims. JAMA Network Open, 4(4), e218075. https://doi.org/10.1001/jamanetworkopen.2021.8075
Cutler, S. (2016). Chapter 28: Contrast media. https://radiologykey.com/contrast-media-2
Davenport, M. S., Perazella, M. A., Yee, J., Dillman, J. R., Fine, D., McDonald, R. J., Rodby, R. A., Wang, C. L., & Weinreb, J. C. (2020). Use of intravenous iodinated contrast media in patients with kidney disease: Consensus statements from the American College of Radiology with the National Kidney Foundation. Radiology, 294(3), 660-668. https://doi.org/10.1148/radiol.2019192094
Elmore, J. G., & Lee, C. I. (2022). Artificial intelligence in medical imaging - learning from past mistakes in mammography. JAMA Health Forum, 3(2), e215207. https://doi.org/10.1001/jamahealthforum.2021.5207
Elsayes, K. M., & Oldham, S. A. A. (2014). Introduction to diagnostic radiology. McGraw-Hill Education. https://accessmedicine.mhmedical.com/book.aspx?bookID=1562
Frane, N., & Bitterman, A. (2022). Radiation safety and protection. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK557499
Ghadimi, M., & Sapra, A. (2022). Magnetic resonance imaging contraindications. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK551669
Gotfried, J. (2023). X-ray and other imaging contrast studies of the gastrointestinal tract. Merck Manual Professional Version. https://www.merckmanuals.com/professional/gastrointestinal-disorders/diagnostic-and-therapeutic-gastrointestinal-procedures/x-ray-and-other-imaging-contrast-studies-of-the-gastrointestinal-tract
Grimm, L. J., Avery, C. S., Hendrick, E., & Baker, J. A. (2022). Benefits and risks of mammography screening in women ages 40 to 49 years. Journal of Primary Care & Community Health, 13. https://doi.org/10.1177/21501327211058322
Gupta, A., Dosekun, A. K., & Kumar, V. (2020). Carbon dioxide-angiography for patients with peripheral arterial disease at risk of contrast-induced nephropathy. World Journal of Cardiology, 12(2), 76-90. https://doi.org/10.4330/wjc.v12.i2.76
Holmes, K., & Griffiths, M. (2016). Chapter 14: Image quality. https://radiologykey.com/image-quality
Huynh, C. (2017). The role of radiology and imaging in chronic lymphocytic leukemia. https://cllsociety.org/2017/12/the-role-of-radiology-and-imaging-in-chronic-lymphocytic-leukemia
Ibrahim, M. A., Hazhirkarzar, B., & Dublin, A. B. (2022). Gadolinium magnetic resonance imaging. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK482487
International Atomic Energy Agency. (n.d.). X-rays: What patients need to know. Retrieved April 12, 2023, from https://www.iaea.org/resources/rpop/patients-and-public/x-rays
International Atomic Energy Agency. (2014). Diagnostic radiology physics: A handbook for teachers and students. IAEA Publications. https://www-pub.iaea.org/MTCD/Publications/PDF/Pub1564webNew-74666420.pdf
Iqbal, A., & Rehman, A. (2022). Thyroid uptake and scan. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK555978
Katal, S., Eibschutz, L. S., Saboury, B., Gholamrezanezhad, A., & Alavi, A. (2022). Advantages and applications of total-body PET scanning. Diagnostics, 12(2), 426. https://doi.org/10.3390/diagnostics12020426
Kidney International. (2012). KDIGO clinical practice guideline for acute kidney injury. Kidney International Supplements, 2(1), 124-138. https://kdigo.org/wp-content/uploads/2016/10/KDIGO-2012-AKI-Guideline-English.pdf
Kocak, M. (2022a). Computed tomography. Merck Manual Professional Version. https://www.merckmanuals.com/professional/special-subjects/principles-of-radiologic-imaging/computed-tomography
Kocak, M. (2022b). Magnetic resonance imaging. Merck Manual Professional Version. https://www.merckmanuals.com/professional/special-subjects/principles-of-radiologic-imaging/magnetic-resonance-imaging
Kocak, M. (2022c). Positron emission tomography (PET). Merck Manual Professional Version. https://www.merckmanuals.com/professional/special-subjects/principles-of-radiologic-imaging/positron-emission-tomography-pet
Kocak, M. (2022d). Radiographic contrast agents and contrast reactions. Merck Manual Professional Version. https://www.merckmanuals.com/professional/special-subjects/principles-of-radiologic-imaging/radiographic-contrast-agents-and-contrast-reactions
Kocak, M. (2022e). Ultrasonography. Merck Manual Professional Version. https://www.merckmanuals.com/professional/special-subjects/principles-of-radiologic-imaging/ultrasonography
McCance, K. L., & Huether, S. E. (2019). Pathophysiology: The biologic basis for disease in adults and children (8th ed.). Elsevier.
Mehta, S. R., & Annamaraju, P. (2022). Intravenous pyelogram. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK559034
Memorial Sloan Kettering Cancer Center. (2023). About your PET-CT with FDG tracer. https://www.mskcc.org/cancer-care/patient-education/pet-ct-fdg
Millington, S. J., Mayo-Malasky, H., & Koenig, S. (2023). Agitated saline contrast injection in patients with severe hypoxemia. Journal of Intensive Care Medicine, 38(5). https://doi.org/10.1177/08850666231159019
Murphy, A. (2023a). Barium swallow. https://radiopaedia.org/articles/barium-swallow
Murphy, A. (2023b). Iodinated contrast media. https://radiopaedia.org/articles/iodinated-contrast-media-1
National Cancer Institute. (2001a). PET scan of a brain tumor [image]. https://visualsonline.cancer.gov/details.cfm?imageid=1755
National Cancer Institute. (2001b). PET scan of a normal brain [image]. https://visualsonline.cancer.gov/details.cfm?imageid=1753
National Cancer Institute. (2007). Mammography in progress [Image]. https://visualsonline.cancer.gov/details.cfm?imageid=4361
National Cancer Institute. (2023). Mammograms. https://www.cancer.gov/types/breast/mammograms-fact-sheet#what-are-the-benefits-and-potential-harms-of-screening-mammograms
National Council on Radiation Protection and Measurements. (2021). NCRP recommendations for ending routine gonadal shielding during abdominal and pelvic radiography. https://ncrponline.org/wp-content/themes/ncrp/PDFs/Statement13.pdf
National Institute of Arthritis and Musculoskeletal and Skin Diseases. (2018). Bone mass measurement: What the numbers mean. https://www.bones.nih.gov/health-info/bone/bone-health/bone-mass-measure
National Institute of Arthritis and Musculoskeletal and Skin Diseases. (2022). Osteoporosis. https://www.niams.nih.gov/health-topics/osteoporosis#tab-overview
National Institute of Biomedical Imaging and Bioengineering. (n.d.). Magnetic resonance imaging (MRI). Retrieved April 24, 2023, from https://www.nibib.nih.gov/science-education/science-topics/magnetic-resonance-imaging-mri
National Institute of Biomedical Imaging and Bioengineering. (2016). Ultrasound. https://www.nibib.nih.gov/science-education/science-topics/ultrasound
National Institute of Biomedical Imaging and Bioengineering. (2019). Nuclear medicine. https://www.nibib.nih.gov/sites/default/files/Nuclear%20Medicine.pdf
National Institute of Biomedical Imaging and Bioengineering. (2022). X-rays. https://www.nibib.nih.gov/science-education/science-topics/x-rays
Occupational Safety and Health Administration. (n.d.). Ionizing radiation. US Department of Labor. Retrieved April 13, 2023, from https://www.osha.gov/ionizing-radiation/control-prevention
Odak, M., & Kayani, W. T. (2022). MUGA scan. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK564365
Omerovic, S., & Jain, A. (2023). Echocardiogram. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK558940
Organisation for Economic Co-operation and Development. (2023). Magnetic resonance imaging (MRI) exams. https://doi.org/10.1787/1d89353f-en
O'Rourke, M. C., Goldstein, S., & Mendenhall, B. R. (2023). Transesophageal echocardiogram. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK442026
Patel, P. R., & De Jesus, O. (2023). CT scan. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK567796
Perelli, F., Turrini, I., Giorgi, M. G., Renda, I., Vidiri, A., Straface, G., Scatena, E., D'Indinosante, M., Marchi, L., Giusti, M., Oliva, A., Grassi, S., De Luca, C., Catania, F., Vizzielli, G., Restaino, S., Gullo, G., Eleftheriou, G., Mattei, A … Cavaliere, A. F. (2022). Contrast agents during pregnancy: Pros and cons when really needed. International Journal of Environmental Research and Public Health, 19(24), 16699. https://doi.org/10.3390/ijerph192416699
Porter, J. L., & Varacallo, M. (2022). Osteoporosis. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK441901
Radiologyinfo.org. (2020). Body CT. https://www.radiologyinfo.org/en/info/bodyct
RadiologyInfo.org. (2021a). Mammography. https://www.radiologyinfo.org/en/info/mammo
RadiologyInfo.org. (2021b). Thyroid scan and uptake. https://www.radiologyinfo.org/en/info/thyroiduptake
RadiologyInfo.org. (2022a). Bone scan. https://www.radiologyinfo.org/en/info/bone-scan
RadiologyInfo.org. (2022b). Contrast materials. https://www.radiologyinfo.org/en/pdf/safety-contrast.pdf
RadiologyInfo.org. (2022c). General ultrasound. https://www.radiologyinfo.org/en/info/genus
RadiologyInfo.org. (2022d). What is radiation dose? https://www.radiologyinfo.org/en/info/safety-hiw_09
RadiologyInfo.org, Radiological Society of North America, & American College of Radiology. (2022). Radiation dose to adults from common imaging examinations. https://www.acr.org/-/media/ACR/Files/Radiology-Safety/Radiation-Safety/Dose-Reference-Card.pdf
Rogers, D. C., & Tadi, P. (2023). Intravenous contrast. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK557794
Rudnick, M. R., & Davenport, M. S. (2023). Prevention of contrast-induced acute kidney injury associated with computed tomography. UpToDate. Retrieved April 14, 2023, from https://www.uptodate.com/contents/prevention-of-contrast-induced-acute-kidney-injury-associated-with-computed-tomography#H1762264800
Smith-Bindman, R., Kwan, M. L., Marlow, E. C., Theis, M. K., Bolch, W., Cheng, S. Y., Bowles, E. J. A., Duncan, J. R., Greenlee, R. T., Kushi, L. H., Pole, J. D., Rahm, A. K., Stout, N. K., Weinmann, S., & Miglioretti, D. L. (2019). Trends in use of medical imaging in US healthcare systems and in Ontario, Canada, 2000-2016. JAMA, 322(9), 843-856. https://doi.org/10.1001/jama.2019.11456
Society of Nuclear Medicine & Molecular Imaging. (n.d.). Fact sheet: What is nuclear medicine and molecular imaging? Retrieved on April 13, 2023, from https://www.snmmi.org/AboutSNMMI/Content.aspx?ItemNumber=5648
Spampinato, M. V., Abid, A., & Matheus, M. G. (2017). Current radiographic iodinated contrast agents. Magnetic Resonance Imaging Clinics of North America, 25(4), 697-704. https://doi.org/10.1016/j.mric.2017.06.003
Susan G. Komen. (2022). Breast cancer screening for women at average risk. https://www.komen.org/breast-cancer/screening/when-to-screen/average-risk-women
UCSF Department of Radiology and Biomedical Imaging. (n.d.). PET/CT scan: how to prepare, what to expect & safety tips. Retrieved April 25, 2023, from https://radiology.ucsf.edu/patient-care/prepare/pet-ct
US Food & Drug Administration. (2016). FDA revises warnings regarding the use of the diabetes medicine metformin in certain patients with reduced kidney function. https://www.fda.gov/media/96771/download
US Food & Drug Administration. (2018). MRI (magnetic resonance imaging). https://www.fda.gov/radiation-emitting-products/medical-imaging/mri-magnetic-resonance-imaging
US Food & Drug Administration. (2020). Ultrasound imaging. https://www.fda.gov/radiation-emitting-products/medical-imaging/ultrasound-imaging
US Food & Drug Administration. (2021). Testing and labeling medical devices for safety in the magnetic resonance (MR) environment: Guidance for industry and food and drug administration staff. https://www.fda.gov/media/74201/download
Vachha, B., & Huang, S. Y. (2021). MRI with ultrahigh field strength and high-performance gradients: Challenges and opportunities for clinical neuroimaging at 7T and beyond. European Radiology Experiment, 5(35). https://doi.org/10.1186/s41747-021-00216-2
Yandrapalli, S., & Puckett, Y. (2022). SPECT imaging. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK564426
Young, M., & Mohan, J. (2022). Carbon dioxide angiography. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK534244
Yusefi, H., & Helfield, B. (2022). Ultrasound contrast imaging: Fundamentals and emerging technology. Frontiers in Physics, 17(10). https://doi.org/10.3389/fphy.2022.791145
Zaccarini, D. J., Lubin, D., Sanyal, S., & Abraham, J. L. (2022). Barium sulfate deposition in the gastrointestinal tract: Review of the literature. Diagnostic Pathology, 27(99). https://doi.org/10.1186/s13000-022-01283-8