About this course:
This course examines the most common tickborne diseases throughout the US, reviewing the epidemiology, transmission, clinical presentation, diagnostic criteria, and treatment modalities from the Centers for Disease Control and Prevention (CDC).
Course preview
Tickborne Diseases (for APRNs)
This course examines the most common tickborne diseases throughout the US, reviewing the epidemiology, transmission, clinical presentation, diagnostic criteria, and treatment modalities from the Centers for Disease Control and Prevention (CDC).
Upon completion of this module, learners should be able to:
- identify the epidemiology and pathogenesis of ticks, including the most common types of ticks, pathogens, and geographic distributions of the most common tickborne diseases
- describe the risk and protective factors for tickborne diseases, proper tick removal, and tick bite prevention
- recognize the signs and symptoms of each type of tickborne disease, clinical diagnostic criteria, and treatment modalities
Tickborne diseases (TBDs) are infectious vector-borne illnesses transmitted to humans and animals through bites by infected ticks. Several infectious pathogens (e.g., viruses, bacteria, rickettsia, and protozoa) use ticks as transmission portals to inflict disease. TBDs are increasingly prevalent throughout the US, with Lyme disease (LD) being the most frequently diagnosed of these illnesses. Other common TBDs include Rocky Mountain spotted fever (RMSF), anaplasmosis, babesiosis, ehrlichiosis, tickborne relapsing fever (TBRF), tularemia, and Powassan virus disease (POW). Tick bites are painless, and many people are unaware they have been bitten by a tick, clouding the clinical suspicion for TBDs. In addition, TBDs often present in varying ways and with overlapping signs and symptoms. TBDs can lead to significant morbidity and mortality if not diagnosed and treated early in the disease trajectory. However, TBDs can be challenging to identify since diagnostic tests can be inaccurate, inconclusive, or complex to interpret, particularly during the earliest stages of the illness when treatment is likely to be the most effective. Because different types of TBDs are prevalent in specific geographic regions and seasons, advanced practice registered nurses (APRNs) must understand the defining features of each disease (CDC, 2022f).
Epidemiology
TBDs are highly prevalent throughout the US, with increasing incidence and expanding geographic distribution over the last few decades. TBDs are a serious problem and pose a threat to public health, as they have the potential to inflict significant morbidity and mortality (Eickhoff & Blaylock, 2017). In 2017, a record number of TBDs were reported to the CDC, totaling 59,350 cases. Between 2004 and 2016, the number of reported TBDs doubled, attributed partially to the discovery of 7 new infectious tickborne pathogens. The number of TBDs decreased in 2018 to 47,743 but rose again in 2019 to 50,865. However, these numbers are estimated to capture only a fraction of actual TBDs in the US. The low numbers of reported cases may be due to healthcare providers' (HCPs) lack of awareness of TBDs, limited testing practices to confirm diagnoses, and underreporting practices. Factors affecting annual TBDs include temperature, humidity, rainfall percentage, and host populations such as mice and other animals (CDC, 2018b, 2022e). Tick prevalence also varies by season and geographic region. Tick exposure can occur year-round, but ticks are most active during warmer months from April through September. However, global warming and climate change are leading to increased TBDs annually. There are several hundred tick species, with approximately 90 types in the US. The geographic distribution of the most common types of ticks that lead to TBDs will be explored in this module as they pertain to each TBD (CDC, 2020).
Pathogenesis
Ticks are arachnids and relate more closely to spiders and mites than other types of insects. They require hosts to survive and transmit infectious pathogens. Their lifecycle consists of 4 stages—egg, larval (infant), nymph (immature), and adult (mature)—as demonstrated in Figure 1.
Figure 1
Lifecycle of Ticks
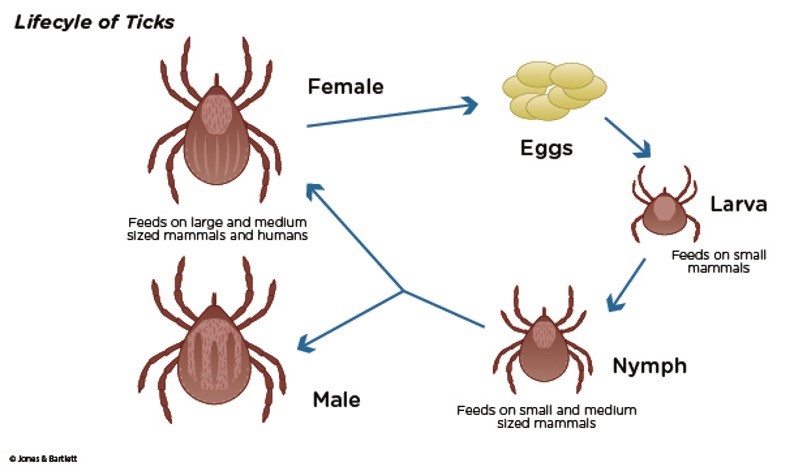
While it can take up to 3 years for a tick to mature into its adult stage, only ticks in the nymph and adult stages can attach to humans and inflict disease. Ticks are not born with a disease but acquire these infectious pathogens while feeding on mammals, reptiles, amphibians, and birds. Once a tick attaches to a host, such as a human, it inserts its hypostome (i.e., the sword-like, piercing mouthpart) into the host's skin. A hypostome is essentially the centerpiece of the tick's mouth that is enclosed within the tick's jaw-like chelicerae. Small teeth-like projections with hooks cover the exterior of the chelicerae, cut into the skin, and lance the host's blood vessels. Once blood pools at the hypostome, the tick releases a sticky, glue-like substance from its salivary glands that cements into the host's skin. This process illuminates why ticks become engorged in the host's skin and are often difficult to remove (Cowles, 2018). The CDC (2021b) recommends removing ticks as soon as possible using the proper technique, as remnants of the tick's mouthparts can be left in the wound following removal. While several tick-removal devices are available for purchase, the CDC also supports using fine-tipped tweezers. Table 1 outlines the steps of properly removing an engorged tick using fine-tipped tweezers (Benzoni & Cooper, 2022; CDC, 2021b).
Table 1
Steps for Patients to Properly Remove Ticks
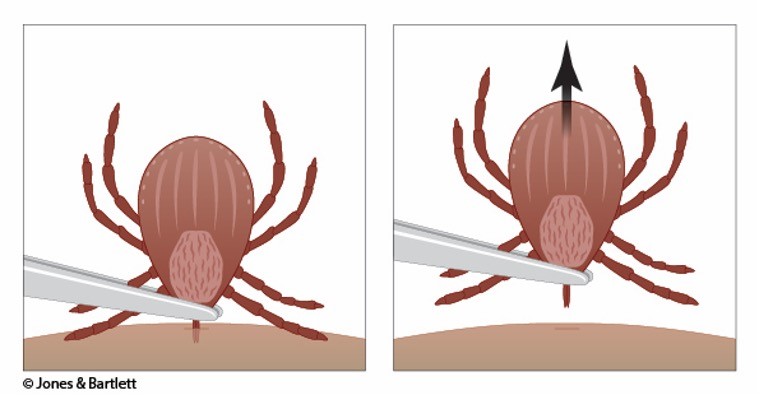
Step 1 | Use clean, fine-tipped tweezers to grasp the tick as close to the skin's surface as possible. |
Step 2 | Pull upward with steady, even pressure when removing the tick (see Figure 2). Avoid jerking or twisting the tick, as this increases the risk that the tick's mouthparts will break off and be retained within the skin. If mouthparts break, remove them individually using the tweezers. If the operator cannot remove the mouth easily with tweezers, seek help from an HCP. |
Step 3 | Once the tick is removed completely, thoroughly clean the bite area with rubbing alcohol or soap and water. The operator should clean their hands thoroughly with soap and water. |
Step 4 | Never crush a tick between the fingers. Dispose of a live tick by drowning it in alcohol, placing it in a sealed bag or container, wrapping it tightly in tape, or flushing it down the toilet. |
Step 5 | Monitor for any new or unusual symptoms for 30 days following the removal of the tick (fever, skin rash, fatigue, headache, muscle pain, or joint pain and swelling) and follow up with a licensed HCP if these symptoms appear. |
(Benzoni & Cooper, 2022; CDC, 2021b)
Figure 2
Tick Removal
Risk Factors and Protective Measures
Risk factors for tick exposure include navigating through grassy, brushy, or wooded areas where ticks are more likely to be found. Although individuals can also be exposed to ticks in their...
...purchase below to continue the course
Developed in 1946 in the US Army and registered for use by the general public in 1957, N, N-diethyl-meta-toluamide (DEET) products are the most commonly used and widely available insect and tick repellants. Approximately 120 products containing N, N-diethyl-meta-toluamide (DEET) are registered with the EPA. They are designed for direct application to the skin and are available in various formulations, including lotions, sprays, towelettes, and roll-on gels. Several safety reviews performed by the EPA over the last 25 years have continued to demonstrate that the regular use of N, N-diethyl-meta-toluamide (DEET) does not present a health concern to the general population, including children. Furthermore, the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA) is a US federal law regulating pesticide use to protect applicators, consumers, and the environment. According to the CDC and FIFRA, when N, N-diethyl-meta-toluamide (DEET) products are used as directed on the manufacturer's label, they are not considered harmful, and only in rare cases are they associated with skin irritation (CDC, n.d.; EPA, 2022).
The CDC (2020) also recommends treating clothing and other outdoor gear, such as hiking boots, sneakers, and backpacks, with products containing 0.5% permethrin. According to the US Department of Health and Human Services (USDHHS, 2018) Tickborne Disease Working Group, clothing treated with permethrin can provide extended protection from black-legged and lone star tick bites. Following outdoor exposure, individuals are encouraged to perform a full body check, examining their clothing and bodies for ticks. One strategy associated with reduced risk of acquiring LD and other common types of TBDs is to remove clothing immediately and shower within 2 hours of outdoor exposure. A full-body skin check should be performed using a hand-held or full-length mirror to inspect all body parts properly. Common hiding spots for ticks on the human body include the following areas: under the armpits, back of the knees, between the legs, around the waist, inside the belly button, and in and around the ears. Ticks may also hide throughout the scalp, in and around the hair. Individuals are advised to wash clothing in hot water immediately following outdoor exposure. Tumble drying clothing on high heat for at least 10 minutes (or until fully dry) helps kill ticks hiding on clothing. The water temperature must be hot for maximum effect, as cold and medium-temperature water will not kill ticks (Benzoni & Cooper, 2022; CDC, 2020).
Aside from clothing, household pets are another highly common portal for transmitting ticks, which can later attach themselves to humans. Since dogs are particularly vulnerable to tick bites and TBDs, owners should carefully examine dogs for ticks following each outdoor exposure. Essential areas to assess include in and around the dog's ears, around the tail and eyelids, under the collar and front legs, as well as between the back legs and toes. However, tick bites can be even more challenging to detect on dogs due to their heavy coats of hair and fur obscuring visualization of many areas of their skin. Therefore, the CDC advises owners to use tick-preventative products on their dogs to reduce the chances that a tick bite will make them ill. Similar to humans, if a tick is identified on a dog, it is essential to remove it as soon as possible (CDC, 2019c).
Also, individuals can take preventative measures to reduce the tick burden in their yards by using pesticides to aid in a tick-safe zone to minimize the risk of tick bites for humans and dogs. Before spraying pesticides, the CDC (2019b) recommends that users check with their local health or agricultural officials regarding the best time to spray pesticides in the designated geographical area, the appropriate pesticide type, and pertinent regulations regarding pesticide application on residential properties. Furthermore, additional landscape recommendations include mowing the lawn frequently, removing leaf litter, creating barriers (e.g., fences) to prevent deer or raccoons from entering the yard, and clearing tall grasses or bushes around the home (CDC, 2019b).
Tick Bite Prophylaxis
Providers are not advised to prescribe antibiotics to patients following a tick bite to prevent TBDs, such as anaplasmosis, babesiosis, RMSF, or other rickettsial diseases. However, according to the Infectious Disease Society of America (IDSA), in geographic areas that are highly endemic for LD, a single dose of doxycycline (Doryx) may be offered as a means of antimicrobial prophylaxis. Non-pregnant adults may be prescribed a single dose of doxycycline (Doryx) 200 mg, and children older than 8 should be prescribed 4 mg/kg up to a maximum dose of 200 mg. As per the IDSA recommendations regarding antimicrobial prophylaxis, the estimated time of attachment must be 36 hours or greater (based on estimated tick engorgement), prophylaxis must be started within 72 hours of tick removal, and LD must be common in the county or state in which the affected individual lives or recently traveled (CDC, 2021a, 2022f).
Lyme Disease
LD is the most common and frequently diagnosed TBD, with about 300,000 reported cases annually in the US. The number of counties in the US considered high incidence for LD has increased by over 300% in the Northeastern states and nearly 250% in North-Central states (USDHHS, 2018). Approximately 10% to 20% of patients with LD experience chronic and disabling symptoms, and direct costs to the US healthcare system reach $1.3 billion annually. LD is primarily transmitted by the black-legged tick (Ixodes scapularis) or the western black-legged tick (Ixodes pacificus). The black-legged tick (Ixodes scapularis) is otherwise known as the deer tick and transmits the spirochete Borrelia burgdorferi and Borrelia mayonii, both of which are known to cause LD. These ticks cause most LD infections in the US, are most prevalent in the warmer months, and, as demonstrated in Figure 3, are widely spread throughout the eastern US. The western black-legged tick (Ixodes pacificus) is found along the Pacific coast, primarily in northern California, Oregon, and Washington, although infection rates are usually low (~1%) in adults. States with the highest incidence of LD include Connecticut, Delaware, Maine, Maryland, Massachusetts, Minnesota, New York, New Jersey, New Hampshire, Pennsylvania, Rhode Island, Virginia, Vermont, Washington DC, West Virginia, and Wisconsin (CDC, 2022d, 2022f).
Figure 3
Geographic Distribution of the Blacklegged Tick (Ixodes scapularis)
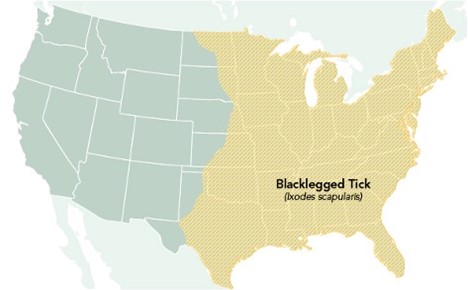
(CDC, 2022c)
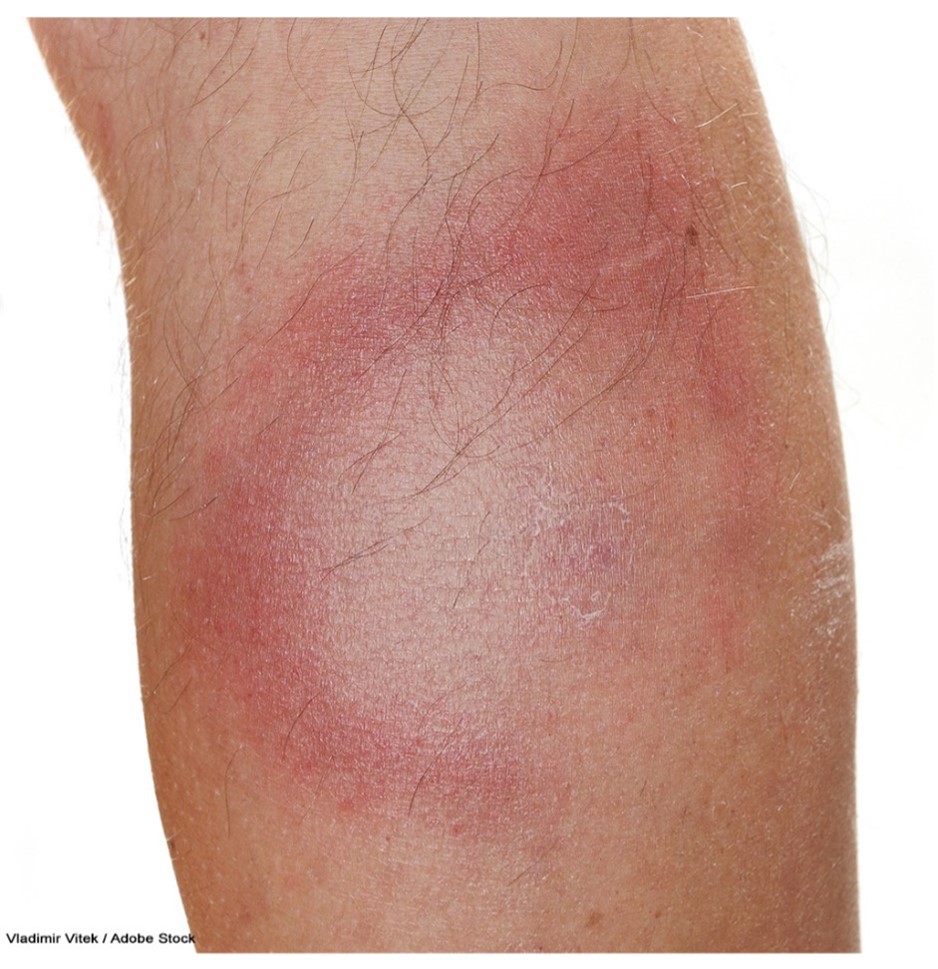
The incubation period of LD can range from 3 to 30 days following a bite by an infected tick. Early-stage symptoms may include erythema migrans (EM), the classic bullseye rash. This ring-like rash with a central clearing slowly expands its perimeter, as demonstrated in Figure 4. The rash occurs in up to 80% of infected persons, begins at the tick bite site about 7 days following a bite, and expands gradually over several days. It may be warm to the touch but is rarely associated with pruritus or skin lesions. An EM-like rash has also been found on humans following a lone star tick bite. This condition, Southern Tick-Associated Rash Illness (STARI), resembles early LD, complicating the diagnosis. Lone star ticks can be found eastward from central Texas and Oklahoma across the southern states and along the Atlantic coast. Systemic symptoms may accompany STARI, but severe disease has not been found. The cause of STARI is unknown, and diagnostic tests are not available. It is not currently known if antibiotic treatment is necessary for patients with STARI; however, many providers treat this condition with the same antibiotics as LD (CDC, 2022f).
Other early symptoms may include flu-like syndromes such as fatigue, headaches, fever, arthralgia, myalgias, and lymphadenopathy. These symptoms may develop, persist, and progress for days or months following the tick bite, leading to the condition's disseminated (i.e., later) stage. The disseminated stage can be characterized by multiple secondary annular rashes and progressive symptoms affecting various body systems, especially the musculoskeletal, neurologic, and cardiac systems. Rheumatologic and musculoskeletal manifestations of LD may include Baker's cysts, severe joint pain, redness, and swelling that most prominently affect the large joints, as well as migratory arthritis and effusion in one or more joints. If these symptoms are left untreated, patients can experience chronic or recurrent episodes of arthritis in the joints. Cardiac manifestations include conduction abnormalities, myocarditis, pericarditis, shortness of breath, and dizziness. Neurologic manifestations may consist of Bell's palsy (i.e., paralysis of one side of the face), inflammation of the brain and spinal cord (meningitis, encephalopathy, encephalitis), and nerve pain, including peripheral neuropathy (CDC, 2022f).
Figure 4
Erythema Migrans Rash in Lyme Disease
Several types of testing are available for LD; however, the CDC endorses a two-step antibody testing protocol, which can be performed using the same blood sample. The testing evaluates for immunoglobulin M (IgM) or immunoglobulin G (IgG) LD antibodies in a serum sample. Current recommendations include using a sensitive enzyme immunoassay (EIA) or immunofluorescence assay, followed by a western immunoblot assay for specimens yielding positive or equivocal results. If the EIA is negative, no further testing is advised. If the EIA is indeterminate (or equivocal), the Western immunoblot (Western blot) test should be performed. Western blot testing can detect antibodies to several proteins of B. burgdorferi and is usually performed to confirm the diagnosis. However, clinicians must be aware that serologic tests are not sensitive during the first few weeks of infection, so the EIA test may not be positive during the early stage of the disease. While this has been the standard testing protocol for more than a decade, the US Food and Drug Administration (FDA) developed several LD serologic assays with new indications for use in July 2019, allowing for an EIA (rather than Western immunoblot assay) to serve as the second step in LD testing (Mead et al., 2019). On routine laboratory testing, patients with LD may demonstrate an elevated erythrocyte sedimentation rate (ESR) and mild hepatic transaminases (CDC, 2022f).
The preferred first-line treatment for LD in adults is antibiotic therapy with doxycycline (Doryx). Doxycycline (Doryx) is a broad-spectrum antibiotic derived from the tetracycline class, a group of antibiotics associated with staining permanent teeth in young children. However, when doxycycline (Doryx) is prescribed at the recommended dose and duration required to treat anaplasmosis, there has been no evidence that it causes staining of permanent teeth when given before the age of 8. Patients usually recover completely within 2 to 4 weeks when treatment is initiated early in the disease. When different durations of antibiotics are shown, a shorter duration is preferred to minimize adverse effects, including antimicrobial resistance and diarrhea. The standard treatment regimens for uncomplicated LD are as follows:
- Pharmacological management of adults
- doxycycline (Doryx) 100 mg by mouth twice daily for 10 to 14 days (preferred regimen)
- cefuroxime axetil (Ceftin) 500 mg by mouth twice daily for 14 days
- amoxicillin (Amoxil) 500 mg by mouth 3 times per day for 14 days
- Pharmacological management of children
- amoxicillin (Amoxil) 50 mg/kg/day by mouth (maximum 500 mg per dose), divided into 3 doses for 14 days
- doxycycline (Doryx) 4.4 mg/kg/day by mouth (maximum 100 mg per dose), divided into 2 doses for 10 to 14 days
- cefuroxime axetil (Ceftin) 30 mg/kg/day by mouth (maximum 500 mg per dose), divided into 2 doses for 14 days (CDC, 2022f)
For more complex LD cases, such as those with facial palsy or suspected meningitis, a lumbar puncture may be required to evaluate the cerebral spinal fluid (CSF) for infection. With Lyme meningitis, the CSF will demonstrate lymphocytic pleocytosis (i.e., abnormal lymphocyte increase), normal glucose, and elevated protein. Intravenous antibiotic therapy may be required for these patients, as well as those with other severe symptoms (e.g., cardiac involvement) or those with manifestations of late LD, such as Lyme arthritis that does not respond to oral antibiotic therapy. Intravenous antibiotics are only recommended if arthritis does not improve with oral treatment and is typically prescribed for up to 28 days. Ceftriaxone (Rocephin) is the preferred antibiotic for neurologic LD in the US and is administered at 2 g IV daily for 14–28 days (CDC, 2022c).
Post-treatment Lyme disease syndrome (PTLDS) manifests as a persistent constellation of symptoms, including fatigue, headaches, joint pain, musculoskeletal aches, and cognitive difficulties that can last a significant period, usually more than 6 months following the completion of treatment. Symptoms can range from mild to severe, and the etiology and management of these symptoms are poorly understood. Unfortunately, no treatment is proven effective for PTLDS, and efforts are focused on managing symptoms (Hu, 2021).
Anaplasmosis
The pathogen Anaplasma phagocytophilum causes anaplasmosis. This condition is primarily transmitted through the Western black-legged tick (Ixodes pacificus) and the black-legged tick (Ixodes scapularis), both known to transmit LD. Thus, coinfection with anaplasmosis and LD is not uncommon. Anaplasmosis is more common in the areas where LD is endemic, such as the US Pacific coast—mainly northern California, Oregon, and Washington—and throughout the eastern US (CDC, 2022e). The incubation period is 5 to 14 days, and symptoms usually begin within 1 to 2 weeks following an infected tick bite. Early signs and symptoms are generally mild to moderate and commonly include any or several of the following: fever, chills, rigors, severe headache, myalgias, malaise, nausea, vomiting, diarrhea, and anorexia. Although this is relatively rare, under 10% of patients may experience a skin rash. Therefore, a rash in a patient with suspected anaplasmosis may indicate a coinfection with LD. Clinical manifestations of late-stage illness may include hypoxia, respiratory failure, organ failure, bleeding problems, and death. Given the heightened risk for morbidity and mortality with late-stage illness, treatment with appropriate antibiotic therapy should not be delayed for patients with clinical signs suggestive of anaplasmosis. A presumptive diagnosis is often made based on the signs and symptoms of the condition and later confirmed through laboratory testing. Early recognition and timely initiation of treatment are essential (CDC, 2022a). The diagnostic workup and findings of anaplasmosis are listed in Table 2.
Table 2
Anaplasmosis Diagnostic Testing
Diagnostic Test | Probable Results |
Routine laboratory work |
|
Blood-smear microscopy
|
|
PCR |
|
Serology |
|
(CDC, 2022a)
Doxycycline (Doryx) is the recommended treatment for all patients with suspected anaplasmosis infections, including adults and children. It is the most effective at preventing complications when started early in the illness. Adults should receive a dose of 100 mg twice daily, either orally or intravenously. Children under 100 pounds should receive a dose of 2.2 mg/kg twice daily, up to a maximum of 100 mg per dose. Treatment should be prescribed for 10 to 14 days to provide an appropriate length of therapy for possible coinfection with LD. The use of antibiotics other than doxycycline (Doryx) increases the risk of patient morbidity and mortality (CDC, 2022f).
Rocky Mountain Spotted Fever
RMSF belongs to a group of related conditions known as spotted fever group rickettsioses. RMSF is caused by the pathogen Rickettsia rickettsii and is transmitted by the Rocky Mountain wood tick (Dermacentor andersoni), the American dog tick (Dermacentor variabilis), and the Brown dog tick (Rhipicephalus sanguineus). The Rocky Mountain wood tick (Dermacentor andersoni) is found within the Rocky Mountain states and southwestern Canada at elevations of 4,000 to 10,500 feet. The American dog tick (Dermacentor variabilis), sometimes called the wood tick, is widely dispersed in areas east of the Rocky Mountains and isolated areas along the Pacific Coast. The Brown dog tick (Rhipicephalus sanguineus) is found across the US, as demonstrated in Figure 5. However, it primarily transmits RMSF in the southwestern portion of the US and along the US-Mexico border. While dogs are the primary host of Brown dog ticks, they can also directly bite humans or other mammals. RMSF has become more common in areas of Arizona over the last decade, with more than 500 cases and 28 fatalities between 2002 and 2021 (CDC, 2022e).
Figure 5
Geographic Distribution of the Brown Dog Tick
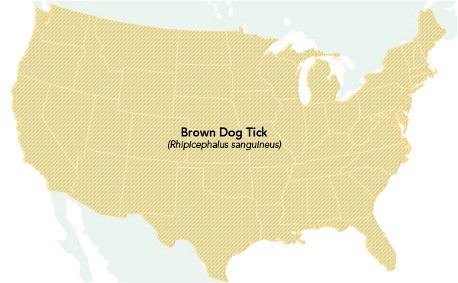
(CDC, 2022e)
RMSF can be transmitted after only 2 to 20 hours of being bitten by an infected tick, with symptoms developing within 3 to 12 days. Early symptoms include high fevers, severe headaches, malaise, myalgias, nausea, vomiting, anorexia, and edema around the eyes and on the back of the hands. A rash usually appears between 2 and 4 days following the onset of the fever, although nearly 10% of patients with RMSF never develop a rash. The rash is initially maculopapular, with small, flat, pink, non-pruritic macules on the wrists, forearms, and ankles, but eventually spreads to the trunk, palm, and soles. Around day 6 or later, the rash usually changes to a petechial rash, a sign of progression to severe disease. Late symptoms, occurring around a week after the initial onset, may include an altered mental status and respiratory compromise. In the most severe cases, damage to blood vessels may lead to extremity necrosis requiring amputation, cerebral edema, coma, and multiorgan system damage (CDC, 2019e).
A definitive diagnosis of RMSF is based on laboratory testing, as portrayed in Table 3. However, due to the potential for rapid disease progression and the severity of the condition, antibiotic therapy should not be delayed for patients presenting with clinical signs indicating RMSF. The goal is to start antibiotic treatment before the petechial rash develops, as this is more likely to prevent fatal outcomes, particularly when treatment is started within the first 5 days of symptom onset (CDC, 2019e, 2022f).
Table 3
Rocky Mountain Spotted Fever Diagnostic Testing
Diagnostic Test | Probable Results |
Routine laboratory work |
|
PCR |
|
Serology |
|
IHC |
|
(CDC, 2022f)
Like anaplasmosis and LD, doxycycline (Doryx) is the first-line, mainstay treatment for RMSF in patients of all ages. Adults should receive a dose of 100 mg twice daily, either orally or intravenously. Children under 100 pounds should receive a dose of 2.2 mg/kg twice daily, up to a maximum of 100 mg per dose. The duration of therapy should continue for at least 3 days after the fever subsides with evidence of clinical improvement. The CDC (2022f) recommends that doxycycline (Doryx) should be continued for at least 5 to 7 days, as the use of antibiotics other than doxycycline (Doryx) increases the risk of patient death. Patients with RMSF usually respond well to treatment, and long-term health complications or persistent infections are rare. However, a small subset of patients who recover from the illness may have permanent damage to the extremities, manifesting as neuropathy, paralysis, or amputation (CDC, 2019e).
Babesiosis
Babesiosis is caused by the microscopic parasite Babesia microti, which infects red blood cells (RBCs) and can cause severe disease. Babesia microti is most commonly carried by the black-legged tick (Ixodes scapularis) and is found in the northeast and upper areas of the midwest, particularly New England, New York, New Jersey, Wisconsin, and Minnesota (CDC, 2022c).
The incubation period of babesiosis can range from 1 to 9 weeks. While the clinical manifestations may develop several weeks following exposure, some individuals may develop symptoms many months later. Babesiosis can vary dramatically in severity, ranging from asymptomatic infection to a life-threatening condition. Some individuals may experience mild flu-like symptoms such as fevers, chills, headaches, body aches, anorexia, abdominal pain, nausea, or generalized fatigue. In contrast, others may endure more serious effects, including significant RBC destruction, causing hemolytic anemia, jaundice, hepatosplenomegaly, and dark urine. Less commonly, patients may develop a sore throat, coughing, photophobia, and emotional lability. Those at heightened risk for becoming severely ill include older adults, those without a spleen, the immunocompromised (e.g., due to cancer, organ transplant, or human immunodeficiency virus [HIV]), and those who have other serious, chronic conditions such as diabetes or liver, cardiac, or renal disease (CDC, 2018a, 2022f). Babesiosis diagnostic testing is reviewed in Table 4.
Table 4
Babesiosis Diagnostic Testing
Diagnostic Test | Probable Results |
Routine laboratory work |
|
Blood-smear microscopy |
|
PCR |
|
Serology |
|
(CDC, 2017, 2022f)
While most asymptomatic individuals do not require treatment, ill patients are treated with a combination of two medications for at least 7 to 10 days. According to the CDC (2019d, 2022f), treatment for adults is typically one of the following regimens (with the combination of atovaquone [Mepron] and azithromycin [Zithromax] being preferred or clindamycin [Cleocin] and quinine [Qualaquin]):
Primary regimen:
- Standard of care for patients with non-hospitalized mild to moderate babesiosis:
- atovaquone (Mepron) 750 mg by mouth twice daily, PLUS
- azithromycin (Zithromax) 500 mg by mouth on the first day, followed by 250 mg daily on subsequent days to complete 7 to 10 days of combined therapy
- Standard of care for patients with hospitalized acute severe babesiosis illness:
- atovaquone (Mepron) 750 mg by mouth twice daily, PLUS
- azithromycin (Zithromax) 500 mg IV daily until symptoms lessen, followed by a transition to all oral step-down therapy
- Standard of care for patients hospitalized on step-down therapy:
- atovaquone (Mepron) 750 mg by mouth twice daily, PLUS
- azithromycin (Zithromax) 250 mg to 500 mg orally daily (consider 500 mg to 1000 mg for immunocompromised adults (CDC, 2019d, 2022f)
Alternative regimen:
- Standard of care for patients with non-hospitalized mild to moderate babesiosis:
- clindamycin (Cleocin) 600 mg orally 3 times daily, PLUS
- quinine (Qualaquin) 650 mg by mouth 3 times daily
- Standard of care for patients with hospitalized acute severe babesiosis illness:
- clindamycin (Cleocin) 600 mg IV every 6 hours until symptoms lessen, then transition to oral step-down therapy, PLUS
- quinine (Qualaquin) 650 mg by mouth 3 times daily
- Standard of care for patients hospitalized on step-down therapy:
- clindamycin (Cleocin) 600 mg orally 3 times daily, PLUS
- quinine (Qualaquin) 650 mg by mouth 3 times daily (CDC, 2019d, 2022f)
In addition to these definitive treatments, acutely ill patients often require supportive care therapies such as antipyretics or blood transfusions. Critically ill patients may require vasopressors, mechanical ventilation, or dialysis. Complications of untreated babesiosis can include hypotension, thrombocytopenia, and disseminated intravascular coagulation (DIC), which can lead to blood clots and bleeding, malfunction of vital organs, and death (CDC, 2018a, 2019d).
Ehrlichiosis
Ehrlichiosis can be caused by the pathogen Ehrlichia muris eauclairensis, most commonly carried by the black-legged tick (Ixodes scapularis), or by Ehrlichia chaffeensis and Ehrlichia ewingii, which are carried by the Lone star tick (Amblyomma americanum). As demonstrated in Figure 6, the Lone star tick (Amblyomma americanum) is widely distributed in the southeastern and eastern US. This aggressive tick acquired its name from the classic white dot on its back. Individuals bitten by a Lone star tick can develop skin irritation, redness, and discomfort at the bite site, but these symptoms are considered a local reaction. They do not necessarily indicate an infection (CDC, 2022e).
Figure 6
Geographic Distribution of the Lone Star Tick
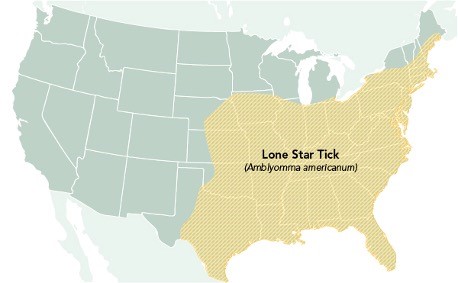
(CDC, 2022e)
While E. muris eauclairensis and E. ewingii have no reported deaths, E. chaffeensis is considered the most serious and is linked to fatalities. The incidence of E. chaffeensis increases with age. However, the highest mortality rates are among children under 10 and adults 70 years and older. The incubation period for ehrlichiosis is 5 to 14 days. The clinical presentation resembles several other types of TBDs, exhibiting the classic early symptomology of fevers, chills, malaise, headaches, myalgias, nausea, vomiting, diarrhea, and anorexia. Other symptoms may include altered mental status, confusion, and skin rash. The rash begins about 5 days after the onset of symptoms and is more common in children, affecting up to 60% of children and under 30% of adults. More commonly associated with E. chaffeensis, the rash is generally non-pruritic, spares the face, and ranges from maculopapular to petechial in presentation. Severe illness can develop if treatment is not initiated promptly, leading to serious manifestations such as meningitis, meningoencephalitis, acute respiratory distress syndrome, liver or renal failure, septic shock-like syndromes, and coagulopathies. Those at increased risk for severe illness include older adults, young children, and immunocompromised patients (CDC, 2019a).
Confirmation of the diagnosis is premised on laboratory testing, as described in Table 5. However, the CDC (2022f) supports the early initiation of antibiotic therapy for patients with high clinical suspicion for the illness (CDC, 2018d, 2019b).
Table 5
Ehrlichiosis Diagnostic Testing
Diagnostic Test | Probable Results |
Routine laboratory work |
|
Blood-smear microscopy |
|
PCR |
|
Serology |
|
IHC and culture |
|
(CDC, 2019a, 2022f)
Doxycycline (Doryx) is the first-line treatment for ehrlichiosis in adults and children. According to the CDC (2019a), patients appropriately treated early in the disease trajectory with doxycycline (Doryx) usually recover rapidly and completely. Adults should be prescribed doxycycline (Doryx) 100 mg by mouth or IV every 12 hours, and children under 100 lbs. (45 kg) require a dosing level of 2.2 mg/kg by mouth or IV twice daily (maximum 100 mg per dose). Treatment should continue until at least 3 days after the fever subsides with evidence of clinical improvement. Typically, patients are treated for 5 to 7 days. While the CDC (2019a) acknowledges that patients who experience a more severe illness trajectory may require IV antibiotics, they do not offer guidelines delineating which IV antibiotics are appropriate.
Tickborne Relapsing Fever
TBRF is caused by the pathogens Borrelia hermsii and Borreilia turicatae and is most commonly transmitted through infected "soft ticks" that belong to the genus Ornithodoros such as O. hermsi, O. parkeri, and O. turicata. These ticks appear within the mountainous regions of the Western parts of the US. O. hermsi is responsible for most cases of TBRF in the US and is most commonly found at high altitudes of 1,500 to 8,000 feet, where they feed on tree squirrels and chipmunks. O. parkeri and O. turicata are generally found at lower altitudes in the Southwest region, where they dwell in caves and the burrows of squirrels, prairie dogs, and owls (CDC, 2015).
TBRF is most common in the summer when people vacation in the mountains and sleep in camp cabins. However, it can occur in the winter when fires are started to warm cabins, activating ticks hiding in the walls and woodwork. The incubation period of TRBF is about 7 days, and the classic triad of symptoms includes a high fever (i.e., 103°F), muscle and joint aches, and a headache. These symptoms follow a distinct pattern of febrile episodes that last about 3 days, followed by a 7-day afebrile period and another 3 days of fever, as demonstrated in Figure 7. If not appropriately treated with antibiotics, this pattern can recur several times. Less commonly, patients may develop nausea, vomiting, a macular or petechial rash, jaundice, hepatosplenomegaly, and photophobia. TBRF can be transmitted transplacentally and is associated with premature birth, spontaneous abortion, and neonatal death (CDC, 2015, 2022f).
Figure 7
Usual Pattern of Symptoms in TBRF
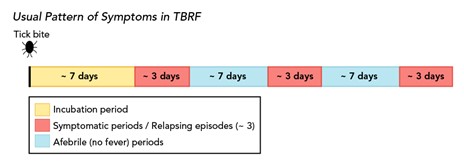
(CDC, 2022g)
For diagnosing TBRF, organisms are best detected in the blood by microscopy or culture obtained during the febrile period of the illness. Spirochetes are more readily detected by microscopy in symptomatic, untreated patients early in the infection (CDC, 2015, 2022f). The details regarding the diagnostic testing of TBRF are outlined in Table 6.
Table 6
TBRF Diagnostic Testing
Diagnostic Test | Probable Results |
Routine laboratory work |
|
Blood-smear microscopy |
|
Serology |
|
(CDC, 2022g)
TBRF is primarily treated with antibiotics such as doxycycline (Doryx), azithromycin (Zithromax), penicillin, or ceftriaxone (Rocephin). Patients treated for TBRF should be monitored closely for Jarisch-Herxheimer reactions during the first 2 to 4 hours of antibiotic therapy. A Jarisch-Herxheimer reaction is a systemic response that may be caused by the release of endotoxin-like substances into the bloodstream due to antibiotic treatment causing cellular lysis. As the bacterial cells are lysed or broken and destroyed, endotoxin-like substances are dispersed throughout the bloodstream. A temporary inflammatory response follows, leading to fevers, chills, rigors, hypotension, tachycardia, and diaphoresis. The reaction usually lasts under 4 hours but may persist for up to 24 hours and is most commonly associated with the administration of tetracycline antibiotics. It is rarely seen with subsequent dosing of the same antibiotic and is most often a self-limiting condition that resolves without significant morbidity. However, rare complications can include cardiovascular collapse, acute respiratory distress syndrome requiring intubation, and death. Treatment is limited to the management of symptoms; however, the use of acetaminophen (Tylenol), corticosteroids, and non-steroidal anti-inflammatory (NSAIDs) do not appear to modify these symptoms or patient outcomes (Butler, 2017). Antibiotic treatment of TBRF includes the following:
- Pharmacological management of adults
- doxycycline (Doryx) 100 mg orally or IV, 2 times daily for 10 days (first-line treatment)
- azithromycin (Zithromax) 500 mg by mouth daily for 10 days
- penicillin G (Bicillin LA) 4,000,000 units every 6 hours IV for 10 days
- ceftriaxone (Rocephin) 2 g IV every 24 hours for 10 days (reserved for patients with central nervous system involvement)
- Pharmacological management of children
- doxycycline (Doryx) 2.2 mg/kg per dose IV or oral, 2 times daily (maximum 100 mg/dose; first-line treatment)
- azithromycin (Zithromax) 10 mg/kg by mouth daily (maximum 500 mg/day) for 10 days
- penicillin G (Bicillin LA) 50,000 to 100,000 units/kg every 6 hours IV (maximum 4,000,000 units/dose) for 10 days
- ceftriaxone (Rocephin) 50 to 75 mg/kg IV daily (maximum 2 g/day) for 10 days (reserved for patients with central nervous system involvement [CDC, 2022f])
Tularemia
Tularemia is caused by the pathogen Francisella tularensis, which can be transmitted by the American dog tick (Dermacentor variabilis), the Lone star tick (Amblyomma americanum), or the Rocky Mountain wood tick (Dermacentor andersoni). It is widely distributed east of the Rocky Mountains and southwestern Canada from elevations of 4,000 to 10,500 feet, as well as in discrete areas on the Pacific Coast. It carries an average incubation period of 3 to 5 days, but the range can extend to 21 days. Tularemia is also referred to as "rabbit fever" or "deer fly fever," as rabbits, hares, and rodents are particularly susceptible to the illness (CDC, 2022f).
Tularemia is challenging to diagnose due to its highly variable clinical presentation; it is not solely a tickborne illness. People can become infected by several modes of transmission, such as skin contact with infected animals, drinking contaminated water, laboratory exposure, or inhaling contaminated agricultural or landscaping dust and aerosols. Symptoms can be mild to life-threatening and vary based on how the bacteria enter the body. General symptoms can include chills, headaches, fatigue, malaise, myalgia, anorexia, a sore throat, chest discomfort, coughing, abdominal pain, nausea, and diarrhea. The six main forms of tularemia and their associated symptomology are outlined in Table 7/Table 2. However, all forms of illness are accompanied by a fever, ranging from mild to severe but not above 104 °F (CDC, 2018c).
Table 7
Forms of Tularemia and Associated Signs and Symptoms
Type | Signs and Symptoms |
Ulceroglandular |
|
Glandular |
|
Oculoglandular |
|
Oropharyngeal |
|
Pneumonic |
|
Typhoidal |
|
(CDC, 2018c, 2022f)
Tularemia is diagnosed through clinical suspicion based on signs and symptoms, travel history, and blood and culture results, as outlined in Table 8.
Table 8
Diagnosis of Tularemia
Diagnostic Test | Probable Results |
Routine laboratory work |
|
Culture |
|
PCR and serology |
|
(CDC, 2018c, 2022b)
Tularemia is treated with antibiotic therapy for 10 to 21 days. Streptomycin is considered first-line therapy due to its efficacy and FDA approval; it is only available in the generic formulation. Gentamicin (Genoptic) is an acceptable alternative; however, it has a lower success rate and is not FDA-approved for this indication. Both streptomycin and gentamicin (Genoptic) require dose adjustments for patients with renal insufficiency. Ciprofloxacin (Cipro) and other fluoroquinolones are also not FDA-approved for treating tularemia but have shown promising efficacy in humans (CDC, 2018c, 2022f). Treatment options include the following:
- Pharmacological management of adults
- streptomycin 1 mg IM twice daily for a minimum of 10 days
- gentamicin (Genoptic) 5 mg/kg IM or IV every 24 hours for a minimum of 10 days
- drug serum levels must be monitored with a desired peak serum level of at least 5 mcg/mL
- ciprofloxacin (Cipro) 400 mg IV twice daily or 500 mg by mouth twice daily for 10 to 14 days
- doxycycline (Doryx) 100 mg IV twice daily or 100 mg by mouth twice daily for 14 to 21 days
- Pharmacological management of children
- streptomycin 15 mg/kg IM twice daily for a minimum of 10 days
- gentamicin (Genoptic) 2.5 mg/kg IM or IV 3 times per day for at least 10 days
- drug serum levels must be monitored, and clinicians are advised to consult with a pediatric infectious disease specialist
- ciprofloxacin (Cipro) 15 mg/kg IV or orally twice daily for 10 days
- maximum daily dose should not exceed 800 mg
- doxycycline (Doryx) 2.2 mg/kg IV or orally twice daily (maximum 100 mg IV or orally twice daily) for 14 to 21 days (CDC, 2022f)
Powassan Virus Disease
POW originates from a family of viruses known as Flavivirdae, and the genus Flavivirus comprises more than 50 viral species worldwide. Tickborne flaviviruses (TBFVs) are more commonly found in subtropical areas of the Northern Hemisphere and less prominently in subtropical regions of Africa and Australia (Kemenesi & Banyai, 2019). POW is the only tickborne flavivirus known in the US, commonly carried by the black-legged tick (Ixodes scapularis). It is related to mosquito-borne viruses, such as the West Nile virus. Although rare, most cases in the US occur in the northeast and the Great Lakes geographic region during the height of the tick season (i.e., between late spring and mid-fall; CDC, 2022e; Fatmi et al., 2017).
The incubation period is 1 to 4 weeks. Initial signs and symptoms of the illness may include fever, headaches, vomiting, and generalized weakness. While some cases may be mild or asymptomatic, others can be severe, including encephalitis or meningitis. Symptoms of encephalitis and meningitis include severe headaches with nausea or vomiting, anorexia, altered mental status/confusion or difficulty concentrating, photophobia, seizures, lethargy, aphasia, paresis, movement disorders, or cranial nerve palsies. The diagnosis is primarily confirmed through the CDC and some state health departments, as commercial testing for POW is limited. Cross-reactions can occur with other flaviviruses, such as West Nile or Dengue (CDC, 2022f). Table 9 outlines the diagnostic process for POW.
Table 9
Diagnosis of POW
Diagnostic Test | Probable Results |
PCR and serology |
|
(CDC, 2022f)
No specific antiviral treatment for POW is currently available, so those affected should receive supportive care therapies for symptom management (CDC, 2022f).
Colorado Tick Fever
CTF is a virus primarily found in the western US, specifically Utah, Colorado, Wyoming, and Montana. The incubation period for CTF is 1 to 14 days. Although rare, it can be spread from person to person through a blood transfusion. Signs and symptoms of CTF include headaches, myalgias, fevers, chills, and lethargy, with 50% of patients displaying a biphasic illness cycle (symptoms remitting after 2 to 4 days but returning 1 to 3 days later). Pharyngeal erythema, conjunctival injection, and lymphadenopathy may be present. A petechial or maculopapular rash is present in under 20% of cases. In addition, prolonged symptoms of weakness and fatigue are common in adults. Although rare, life-threatening complications—including DIC, meningoencephalitis, and death—can occur. General laboratory findings include leukopenia and moderate thrombocytopenia. A preliminary diagnosis of CTF is based on signs and symptoms, places and dates of travel, and history of tick exposure. The preferred CTF test depends on the collection timing since antibody production is delayed 14 to 21 days after the onset of symptoms. For samples collected within 14 days of symptom onset, reverse-transcriptase (RT-PCR) is the preferred test. For samples collected later, antibody testing should be done. Treatment of CTF is supportive (CDC, 2022f).
References
Benzoni, T., & Cooper, J. S. (2022). Tick removal. StatPearls [Internet]. https://www.ncbi.nlm.nih.gov/books/NBK441855
Butler, T. (2017). The Jarisch-Herxheimer reaction after antibiotic treatment of spirochetal infections: A review of recent cases and our understanding of pathogenesis. American Journal of Tropical Medicine and Hygiene, 96(1), 46-52. https://doi.org/10.4269/ajtmh.16-0434
Centers for Disease Control and Prevention. (n.d.). Guidelines for DEET insect repellent use. Retrieved February 7, 2022, from https://www.cdc.gov/malaria/toolkit/DEET.pdf
Centers for Disease Control and Prevention. (2015). Tickborne relapsing fever. https://www.cdc.gov/relapsing-fever/index.html
Centers for Disease Control and Prevention. (2017). Babesiosis. https://www.cdc.gov/dpdx/babesiosis/index.html
Centers for Disease Control and Prevention. (2018a). Babesiosis: Disease. https://www.cdc.gov/parasites/babesiosis/disease.html
Centers for Disease Control and Prevention. (2018b). Record number of tickborne diseases reported in US in 2017. https://www.cdc.gov/media/releases/2018/s1114-record-number-tickborne-diseases.html
Centers for Disease Control and Prevention. (2018c). Tularemia signs and symptoms. https://www.cdc.gov/tularemia/signssymptoms/index.html
Centers for Disease Control and Prevention. (2019a). Ehrlichiosis: Information for healthcare providers. https://www.cdc.gov/ehrlichiosis/healthcare-providers/index.html
Centers for Disease Control and Prevention. (2019b). Preventing ticks in the yard. https://www.cdc.gov/ticks/avoid/in_the_yard.html
Centers for Disease Control and Prevention. (2019c). Preventing ticks on your pets. https://www.cdc.gov/ticks/avoid/on_pets.html
Centers for Disease Control and Prevention. (2019d). Parasites - Babesiosis: Resources for health professionals. https://www.cdc.gov/parasites/babesiosis/health_professionals/index.html
Centers for Disease Control and Prevention. (2019e). Rocky Mountain spotted fever (RMSF). https://www.cdc.gov/rmsf/index.html
Centers for Disease Control and Prevention. (2020). Preventing tick bites. https://www.cdc.gov/ticks/avoid/on_people.html
Centers for Disease Control and Prevention. (2021a). Guidance for clinicians: Caring for patients after a tick bite. https://www.cdc.gov/lyme/resources/FS-Guidance-for-Clinicians-Patients-after-TickBite-508.pdf
Centers for Disease Control and Prevention. (2021b). Tick bite: What to do. https://www.cdc.gov/ticks/pdfs/FS_TickBite-508.pdf
Centers for Disease Control and Prevention. (2022a). Anaplasmosis. https://www.cdc.gov/anaplasmosis/index.html
Centers for Disease Control and Prevention. (2022b). For clinicians: Diagnostic testing for Tularemia. https://www.cdc.gov/tularemia/clinicians/index.html
Centers for Disease Control and Prevention. (2022c). Lyme disease treatment. https://www.cdc.gov/lyme/treatment/index.html
Centers for Disease Control and Prevention. (2022d). Regions where ticks live. https://www.cdc.gov/ticks/geographic_distribution.html
Centers for Disease Control and Prevention. (2022e). Tickborne disease surveillance data summary. https://www.cdc.gov/ticks/data-summary/index.html
Centers for Disease Control and Prevention. (2022f). Tickborne diseases of the United States: A reference manual for healthcare providers (6th ed.). https://www.cdc.gov/ticks/tickbornediseases/TickborneDiseases-P.pdf
Centers for Disease Control and Prevention. (2022g). Tickborne relapsing fever: Information for clinicians. https://www.cdc.gov/relapsing-fever/clinicians/index.html
Cowles, J. (2018). Amazing arachnids. Princeton University Press.
Eickhoff, C., & Blaylock, J. (2017). Tickborne diseases other than Lyme in the United States. Cleveland Clinic Journal of Medicine, 84(7), 555-567. https://doi.org/10.3949/ccjm.84a.16110
Fatmi, S. S., Zehra, R., & Carpenter, D. O. (2017). Powassan virus- A new reemerging tickborne disease. Frontiers in Public Health, 5(342), 1-12. https://doi.org/10.3389/fpubh.2017.00342
Hu, L. (2021). Patient education: Lyme disease treatment (beyond the basics). UpToDate. Retrieved February 5, 2022, from https://www.uptodate.com/contents/lyme-disease-treatment-beyond-the-basics#H6
Kemenesi, G., & Banyai, K. (2019). Tickborne flavivirus, with a focus on Powassan Virus. Clinical Microbiology Reviews, 32(1), 1-29. https://doi.org/10.1128/CMR.00106-17
Mead, P., Petersen, J., & Hinckley, A. (2019). Updated CDC recommendation for serologic diagnosis of Lyme disease. Morbidity and Mortality Weekly Report, 68(32), 703. http://dx.doi.org/10.15585/mmwr.mm6832a4
US Department of Health and Human Services. (2018). Tickborne disease working group: 2018 report to congress. https://www.hhs.gov/sites/default/files/tbdwg-report-to-congress-2018.pdf
US Environmental Protection Agency. (2022). DEET. https://www.epa.gov/insect-repellents/deet#product